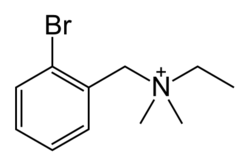
Chemistry:Bretylium
 | |
 | |
| Clinical data | |
|---|---|
| MedlinePlus | a682861 |
| Pregnancy category |
|
| Routes of administration | IV, IM |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | NA |
| Protein binding | NA |
| Metabolism | None |
| Elimination half-life | 7-8 hours |
| Excretion | Renal |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| Chemical and physical data | |
| Formula | C11H17BrN+ |
| Molar mass | 243.168 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Bretylium (also bretylium tosylate) is an antiarrhythmic agent.[1] It blocks the release of noradrenaline from nerve terminals. In effect, it decreases output from the peripheral sympathetic nervous system. It also acts by blocking K+ channels and is considered a class III antiarrhythmic. The dose is 5–10 mg/kg and side effects are high blood pressure followed by low blood pressure and ventricular ectopy.
Originally introduced in 1959 for the treatment of hypertension.[2] Its use as an antiarrhythmic for ventricular fibrillation was discovered and patented by Marvin Bacaner in 1969 at the University of Minnesota.[3]
The American Heart Association removed bretylium from their 2000 ECC/ACC guidelines due to its unproven efficacy and ongoing supply problems. Many have cited these supply problems as an issue of raw materials needed in the production of Bretylium. By the release of the AHA 2005 ECC/ACC guidelines there is no mention of Bretylium and it is virtually unavailable throughout most of the world.[4][5]
On June 8, 2011 bretylium tosylate was announced as unavailable in the US after request of Hospira Inc. to withdraw its NDA from the market. Bretylium will remain on the FDA's discontinued drug list since its withdrawal was not the result of a safety or effectiveness concern.[6] In mid 2019, it was reintroduced. [citation needed]
Uses
The drug was used in emergency medicine, cardiology, and other specialties throughout the 1980s-1990s for the acute management of ventricular tachycardia and ventricular fibrillation refractory to other first line treatments such as defibrillation or lidocaine.[7]
It is contraindicated in patients with AV (atrioventricular) heart block or digoxin toxicity.
Bretylium should be used only in an ICU or emergency department setting and should not be used elsewhere due to its dramatic actions and its predominant side effect of hypotension.[citation needed]
Experimental uses
It is used in physiological and pharmacological research as an inhibitor of sympathetic transmission. Its mechanism of action is the inhibition of neurotransmitter release from sympathetic nerve terminals, both by the inhibition of action potentials in the nerve terminals and by other mechanisms.[8] Its specificity for sympathetic nerves is achieved because it is a substrate for the noradrenaline transporter;[9] hence, it accumulates inside nerve terminals which have this transporter.
References
- ↑ "Selective inhibition of K+-stimulation of Na,K-ATPase by bretylium". British Journal of Pharmacology 104 (4): 895–900. December 1991. doi:10.1111/j.1476-5381.1991.tb12523.x. PMID 1667290.
- ↑ "The drug treatment of hypertension. The results of drug treatment". Proceedings of the Royal Society of Medicine 55: 283–6. April 1962. PMID 13904707.
- ↑ Marvin B Bacaner, "Suppression of cardiac ventricular fibrillation and cardiac arrhythmias with bretylium tosylate", US patent 3441649, assigned to University of Minnesota
- ↑ Khan, M. Gabriel (December 14, 2005). Encyclopedia of Heart Diseases. Academic Press. p. 221. ISBN 978-0-12-406061-6. https://books.google.com/books?id=xco9aJ_Y9XIC&q=bretylium+raw+materials&pg=PA221. Retrieved 2015-07-01.
- ↑ Hypothermia~treatment at eMedicine
- ↑ "Determination that Bretylium Tosylate Injection, 50 Milligrams/Milliliter, Was Not Withdrawn From Sale for Reasons of Safety or Effectiveness". Food and Drug Administration. December 19, 2011. pp. 78669–70. https://www.federalregister.gov/articles/2011/12/19/2011-32367/determination-that-bretylium-tosylate-injection-50-milligramsmilliliter-was-not-withdrawn-from-sale. 76 FR 78669
- ↑ "ACS". kumc.edu. Kansas University Medical Center. http://www2.kumc.edu/wichita/meded/intro/modules/ami/reading/RAMI-09.htm.
- ↑ "Bretylium abolishes neurotransmitter release without necessarily abolishing the nerve terminal action potential in sympathetic terminals". British Journal of Pharmacology 153 (4): 831–9. February 2008. doi:10.1038/sj.bjp.0707623. PMID 18071295.
- ↑ "The selective accumulation of bretylium in sympathetic ganglia and their postganglionic nerves". British Journal of Pharmacology and Chemotherapy 15 (2): 265–70. June 1960. doi:10.1111/j.1476-5381.1960.tb01242.x. PMID 13803289.
 |

