Biology:Ryanodine receptor
| RyR domain | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Identifiers | |||||||||
| Symbol | RyR | ||||||||
| Pfam | PF02026 | ||||||||
| InterPro | IPR003032 | ||||||||
| TCDB | 1.A.3 | ||||||||
| OPM superfamily | 8 | ||||||||
| OPM protein | 5gl0 | ||||||||
| |||||||||
Ryanodine receptors (RyR for short) form a class of intracellular calcium channels in various forms of excitable animal tissue like muscles and neurons.[1] There are three major isoforms of the ryanodine receptor, which are found in different tissues and participate in different signaling pathways involving calcium release from intracellular organelles. The RYR2 ryanodine receptor isoform is the major cellular mediator of calcium-induced calcium release (CICR) in animal cells.
Etymology
The ryanodine receptors are named after the plant alkaloid ryanodine which shows a high affinity to them.
Isoforms
There are multiple isoforms of ryanodine receptors:
- RyR1 is primarily expressed in skeletal muscle
- RyR2 is primarily expressed in myocardium (heart muscle)
- RyR3 is expressed more widely, but especially in the brain.[2]
- Non-mammalian vertebrates typically express two RyR isoforms, referred to as RyR-alpha and RyR-beta.
- Many invertebrates, including the model organisms Drosophila melanogaster (fruitfly) and Caenorhabditis elegans, only have a single isoform. In non-metazoan species, calcium-release channels with sequence homology to RyRs can be found, but they are shorter than the mammalian ones and may be closer to IP3 Receptors.
|
|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Physiology
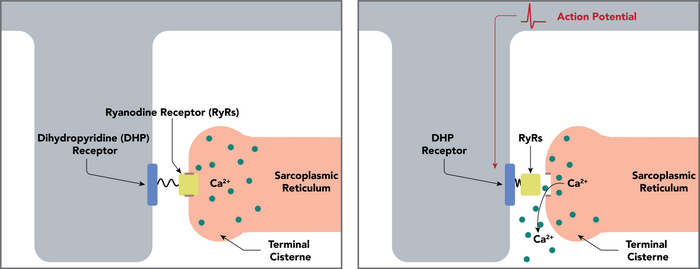
Ryanodine receptors mediate the release of calcium ions from the sarcoplasmic reticulum and endoplasmic reticulum, an essential step in muscle contraction.[1] In skeletal muscle, activation of ryanodine receptors occurs via a physical coupling to the dihydropyridine receptor (a voltage-dependent, L-type calcium channel), whereas, in cardiac muscle, the primary mechanism of activation is calcium-induced calcium release, which causes calcium outflow from the sarcoplasmic reticulum.[3]
It has been shown that calcium release from a number of ryanodine receptors in a ryanodine receptor cluster results in a spatiotemporally restricted rise in cytosolic calcium that can be visualised as a calcium spark.[4] Ryanodine receptors are very close to mitochondria and calcium release from RyR has been shown to regulate ATP production in heart and pancreas cells.[5][6][7]
Ryanodine receptors are similar to the inositol trisphosphate (IP3 or InsP3) receptor, and stimulated to transport Ca2+ into the cytosol by recognizing Ca2+ on its cytosolic side, thus establishing a positive feedback mechanism; a small amount of Ca2+ in the cytosol near the receptor will cause it to release even more Ca2+ (calcium-induced calcium release/CICR).[1] However, as the concentration of intracellular Ca2+ rises, this can trigger closing of RyR, preventing the total depletion of SR. This finding therefore indicates that a plot of opening probability for RyR as a function of Ca2+ concentration is a bell-curve.[8] Furthermore, RyR can sense the Ca2+ concentration inside the ER/SR and spontaneously open in a process known as store overload-induced calcium release (SOICR).[9]
RyRs are especially important in neurons and muscle cells. In heart and pancreas cells, another second messenger (cyclic ADP-ribose) takes part in the receptor activation.
The localized and time-limited activity of Ca2+ in the cytosol is also called a Ca2+ wave. The building of the wave is done by
- the feedback mechanism of the ryanodine receptor
- the activation of phospholipase C by GPCR or RTK, which leads to the production of inositol trisphosphate, which in turn activates the InsP3 receptor.
Associated proteins
RyRs form docking platforms for a multitude of proteins and small molecule ligands.[1] The cardiac-specific isoform of the receptor (RyR2) is known to form a quaternary complex with luminal calsequestrin, junctin, and triadin.[10] Calsequestrin has multiple Ca2+ binding sites and binds Ca2+ ions with very low affinity so they can be easily released.
Pharmacology
- Antagonists:[11]
- Ryanodine locks the RyRs at half-open state at nanomolar concentrations, yet fully closes them at micromolar concentration.
- Dantrolene the clinically used antagonist
- Ruthenium red
- procaine, tetracaine, etc. (local anesthetics)
- Activators:[12]
- Agonist: 4-chloro-m-cresol and suramin are direct agonists, i.e., direct activators.
- Xanthines like caffeine and pentifylline activate it by potentiating sensitivity to native ligand Ca.
- Physiological agonist: Cyclic ADP-ribose can act as a physiological gating agent. It has been suggested that it may act by making FKBP12.6 (12.6 kilodalton FK506 binding protein, as opposed to 12 kDa FKBP12 which binds to RyR1) which normally bind (and blocks) RyR2 channel tetramer in an average stoichiometry of 3.6, to fall off RyR2 (which is the predominant RyR in pancreatic beta cells, cardiomyocytes and smooth muscles).[13]
A variety of other molecules may interact with and regulate ryanodine receptor. For example: dimerized Homer physical tether linking inositol trisphosphate receptors (IP3R) and ryanodine receptors on the intracellular calcium stores with cell surface group 1 metabotropic glutamate receptors and the Alpha-1D adrenergic receptor[14]
Ryanodine
The plant alkaloid ryanodine, for which this receptor was named, has become an invaluable investigative tool. It can block the phasic release of calcium, but at low doses may not block the tonic cumulative calcium release. The binding of ryanodine to RyRs is use-dependent, that is the channels have to be in the activated state. At low (<10 micromolar, works even at nanomolar) concentrations, ryanodine binding locks the RyRs into a long-lived subconductance (half-open) state and eventually depletes the store, while higher (~100 micromolar) concentrations irreversibly inhibit channel-opening.
Caffeine
RyRs are activated by millimolar caffeine concentrations. High (greater than 5 mmol/L) caffeine concentrations cause a pronounced increase (from micromolar to picomolar) in the sensitivity of RyRs to Ca2+ in the presence of caffeine, such that basal Ca2+ concentrations become activatory. At low millimolar caffeine concentrations, the receptor opens in a quantal way, but has complicated behavior in terms of repeated use of caffeine or dependence on cytosolic or luminal calcium concentrations.
Role in disease
RyR1 mutations are associated with malignant hyperthermia and central core disease.[15] Mutant-type RyR1 receptors exposed to volatile anesthetics or other triggering agents can display an increased affinity for cytoplasmic Ca2+ at activating sites as well as a decreased cytoplasmic Ca2+ affinity at inhibitory sites.[16] The breakdown of this feedback mechanism causes uncontrolled release of Ca2+ into the cytoplasm, and increased ATP hydrolysis resulting from ATPase enzymes shuttling Ca2+ back into the sarcoplasmic reticulum leads to excessive heat generation.[17]
RyR2 mutations play a role in stress-induced polymorphic ventricular tachycardia (a form of cardiac arrhythmia) and ARVD.[2] It has also been shown that levels of type RyR3 are greatly increased in PC12 cells overexpressing mutant human Presenilin 1, and in brain tissue in knockin mice that express mutant Presenilin 1 at normal levels,[18] and thus may play a role in the pathogenesis of neurodegenerative diseases, like Alzheimer's disease.[19]
The presence of antibodies against ryanodine receptors in blood serum has also been associated with myasthenia gravis.[1]
Sudden cardiac death in several young individuals in the Amish community (four of which were from the same family) was traced to homozygous duplication of a mutant RyR2 (Ryanodine Receptor) gene.[20] Normal (wild type) ryanodine receptors are involved in CICR in heart and other muscles, and RyR2 functions primarily in the myocardium (heart muscle).
Structure

Ryanodine receptors are multidomain homotetramers which regulate intracellular calcium ion release from the sarcoplasmic and endoplasmic reticula.[21] They are the largest known ion channels, with weights exceeding 2 megadaltons, and their structural complexity enables a wide variety of allosteric regulation mechanisms.[22][23]
RyR1 cryo-EM structure revealed a large cytosolic assembly built on an extended α-solenoid scaffold connecting key regulatory domains to the pore. The RyR1 pore architecture shares the general structure of the six-transmembrane ion channel superfamily. A unique domain inserted between the second and third transmembrane helices interacts intimately with paired EF-hands originating from the α-solenoid scaffold, suggesting a mechanism for channel gating by Ca2+.[1][24]
See also
- Ryanoid, a class of insecticide that act through ryanodine receptors
- Neuromuscular junction
- Dihydropyridine receptor
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 "Essential Roles of Intracellular Calcium Release Channels in Muscle, Brain, Metabolism, and Aging". Current Molecular Pharmacology 8 (2): 206–222. 2015. doi:10.2174/1874467208666150507105105. PMID 25966694.
- ↑ 2.0 2.1 "The sarcoplasmic reticulum Ca2+ channel/ryanodine receptor: modulation by endogenous effectors, drugs and disease states". Pharmacological Reviews 49 (1): 1–51. March 1997. PMID 9085308.
- ↑ "Calcium-induced release of calcium from the cardiac sarcoplasmic reticulum". The American Journal of Physiology 245 (1): C1-14. July 1983. doi:10.1152/ajpcell.1983.245.1.C1. PMID 6346892.
- ↑ "Calcium sparks: elementary events underlying excitation-contraction coupling in heart muscle". Science 262 (5134): 740–744. October 1993. doi:10.1126/science.8235594. PMID 8235594. Bibcode: 1993Sci...262..740C.
- ↑ "Cardiomyocyte ATP production, metabolic flexibility, and survival require calcium flux through cardiac ryanodine receptors in vivo". The Journal of Biological Chemistry 288 (26): 18975–18986. June 2013. doi:10.1074/jbc.M112.427062. PMID 23678000.
- ↑ "Glucagon-like peptide-1 mobilizes intracellular Ca2+ and stimulates mitochondrial ATP synthesis in pancreatic MIN6 beta-cells". The Biochemical Journal 369 (Pt 2): 287–299. January 2003. doi:10.1042/BJ20021288. PMID 12410638.
- ↑ "Glucose and endoplasmic reticulum calcium channels regulate HIF-1beta via presenilin in pancreatic beta-cells". The Journal of Biological Chemistry 283 (15): 9909–9916. April 2008. doi:10.1074/jbc.M710601200. PMID 18174159.
- ↑ "Kinetics of rapid Ca2+ release by sarcoplasmic reticulum. Effects of Ca2+, Mg2+, and adenine nucleotides". Biochemistry 25 (1): 236–244. January 1986. doi:10.1021/bi00349a033. PMID 3754147.
- ↑ "Ryanodine receptors: structure and function". The Journal of Biological Chemistry 287 (38): 31624–31632. September 2012. doi:10.1074/jbc.r112.349068. PMID 22822064.
- ↑ "Calsequestrin interacts directly with the cardiac ryanodine receptor luminal domain". Journal of Cell Science 129 (21): 3983–3988. November 2016. doi:10.1242/jcs.191643. PMID 27609834.
- ↑ "Distinct modes of inhibition by ruthenium red and ryanodine of calcium-induced calcium release in avian atrium". The Journal of Pharmacology and Experimental Therapeutics 268 (3): 1476–1484. March 1994. PMID 7511166.
- ↑ "Potential for pharmacology of ryanodine receptor/calcium release channels". Annals of the New York Academy of Sciences 853 (1): 130–148. September 1998. doi:10.1111/j.1749-6632.1998.tb08262.x. PMID 10603942. Bibcode: 1998NYASA.853..130T.
- ↑ "FKBP12.6 and cADPR regulation of Ca2+ release in smooth muscle cells". American Journal of Physiology. Cell Physiology 286 (3): C538–C546. March 2004. doi:10.1152/ajpcell.00106.2003. PMID 14592808.
- ↑ "Homer binds a novel proline-rich motif and links group 1 metabotropic glutamate receptors with IP3 receptors". Neuron 21 (4): 717–726. October 1998. doi:10.1016/S0896-6273(00)80589-9. PMID 9808459.
- ↑ "RYR1 mutations causing central core disease are associated with more severe malignant hyperthermia in vitro contracture test phenotypes". Human Mutation 20 (2): 88–97. August 2002. doi:10.1002/humu.10098. PMID 12124989.
- ↑ "Functional defects in six ryanodine receptor isoform-1 (RyR1) mutations associated with malignant hyperthermia and their impact on skeletal excitation-contraction coupling". The Journal of Biological Chemistry 278 (28): 25722–25730. July 2003. doi:10.1074/jbc.m302165200. PMID 12732639.
- ↑ "Thermogenesis and energy expenditure: control of heat production by the Ca(2+)-ATPase of fast and slow muscle". Molecular Membrane Biology 19 (4): 301–310. 2002-01-01. doi:10.1080/09687680210166217. PMID 12512777.
- ↑ "Presenilin-1 mutations increase levels of ryanodine receptors and calcium release in PC12 cells and cortical neurons". The Journal of Biological Chemistry 275 (24): 18195–18200. June 2000. doi:10.1074/jbc.M000040200. PMID 10764737.
- ↑ "Polymorphisms Within RYR3 Gene Are Associated With Risk and Age at Onset of Hypertension, Diabetes, and Alzheimer's Disease". American Journal of Hypertension 31 (7): 818–826. June 2018. doi:10.1093/ajh/hpy046. PMID 29590321.
- ↑ "Identification of a Novel Homozygous Multi-Exon Duplication in RYR2 Among Children With Exertion-Related Unexplained Sudden Deaths in the Amish Community". JAMA Cardiology 5 (3): 13–18. March 2020. doi:10.1001/jamacardio.2019.5400. PMID 31913406.
- ↑ "Ryanodine Receptor Structure and Function in Health and Disease". Membrane Protein Complexes: Structure and Function. 87. Singapore: Springer Singapore. 2018. 329–352. doi:10.1007/978-981-10-7757-9_11. ISBN 978-981-10-7756-2.
- ↑ "Ryanodine receptors: structure, expression, molecular details, and function in calcium release". Cold Spring Harbor Perspectives in Biology 2 (11): a003996. November 2010. doi:10.1101/cshperspect.a003996. PMID 20961976.
- ↑ "Ryanodine receptors: allosteric ion channel giants". Journal of Molecular Biology 427 (1): 31–53. January 2015. doi:10.1016/j.jmb.2014.08.004. PMID 25134758.
- ↑ "Structure of a mammalian ryanodine receptor". Nature 517 (7532): 44–49. January 2015. doi:10.1038/nature13950. PMID 25470061. Bibcode: 2015Natur.517...44Z.
External links
- Ryanodine+Receptor at the US National Library of Medicine Medical Subject Headings (MeSH)
 |