Chemistry:Nitrogen trichloride
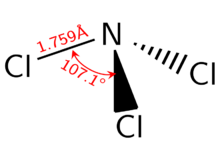
| |
 | |

| |
| Names | |
|---|---|
| Other names
Trichloramine
Agene Nitrogen(III) chloride Trichloroazane Trichlorine nitride | |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| EC Number |
|
| 1840 | |
PubChem CID
|
|
| RTECS number |
|
| UNII | |
| |
| |
| Properties | |
| NCl 3 | |
| Molar mass | 120.36 g·mol−1 |
| Appearance | yellow oily liquid |
| Odor | chlorine-like |
| Density | 1.653 g/mL |
| Melting point | −40 °C (−40 °F; 233 K) |
| Boiling point | 71 °C (160 °F; 344 K) |
| immiscible slowly decomposes | |
| Solubility | soluble in benzene, chloroform, CCl 4, CS 2, PCl 3 |
| Structure | |
| orthorhombic (below −40 °C) | |
| trigonal pyramidal | |
| 0.6 D | |
| Thermochemistry | |
Std enthalpy of
formation (ΔfH⦵298) |
232 kJ/mol |
| Hazards | |
| NFPA 704 (fire diamond) | |
| 93 °C (199 °F; 366 K) | |
| Related compounds | |
Other anions
|
Nitrogen trifluoride Nitrogen tribromide Nitrogen triiodide |
Other cations
|
Phosphorus trichloride Arsenic trichloride |
Related chloramines
|
Monochloramine Dichloramine |
Related compounds
|
Nitrosyl chloride |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Nitrogen trichloride, also known as trichloramine, is the chemical compound with the formula NCl
3. This yellow, oily, pungent-smelling and explosive liquid is most commonly encountered as a byproduct of chemical reactions between ammonia-derivatives and chlorine (for example, in swimming pools). Alongside monochloramine and dichloramine, trichloramine is responsible for the distinctive 'chlorine smell' associated with swimming pools, where the compound is readily formed as a product from hypochlorous acid reacting with ammonia and other nitrogenous substances in the water, such as urea from urine.[1]
Preparation and structure
The compound is prepared by treatment of ammonium salts, such as sal ammoniac with a chlorine source.
Intermediates in this conversion include monochloramine and dichloramine, NH
2Cl and NHCl
2, respectively.
Like ammonia, NCl
3 is a pyramidal molecule. The N-Cl distances are 1.76 Å, and the Cl-N-Cl angles are 107°.[2]
Reactions and uses
The chemistry of NCl
3 has been well explored.[3] It is moderately polar with a dipole moment of 0.6 D. The nitrogen center is basic but much less so than ammonia. It is hydrolyzed by hot water to release ammonia and hypochlorous acid.
- NCl
3 + 3 H
2O → NH
3 + 3 HOCl
NCl
3 explodes to give N
2 and chlorine gas.
- 2 NCl
3 → N
2 + 3 Cl
2
This reaction is inhibited for dilute gases.
Nitrogen trichloride can form in small amounts when public water supplies are disinfected with monochloramine, and in swimming pools by disinfecting chlorine reacting with urea in urine and sweat from bathers.
Nitrogen trichloride, trademarked as Agene, was at one time used to bleach flour,[4] but this practice was banned in the United States in 1949 due to safety concerns.
Safety
Nitrogen trichloride can irritate mucous membranes—it is a lachrymatory agent, but has never been used as such.[5][6] The pure substance (rarely encountered) is a dangerous explosive, being sensitive to light, heat, even moderate shock, and organic compounds. Pierre Louis Dulong first prepared it in 1812, and lost several fingers and an eye in two explosions.[7] In 1813, an NCl
3 explosion blinded Sir Humphry Davy temporarily, inducing him to hire Michael Faraday as a co-worker. They were both injured in another NCl
3 explosion shortly thereafter.[8]
See also
- List of food contamination incidents
References
- ↑ "Chlorine Chemistry - Chlorine Compound of the Month: Chloramines: Understanding "Pool Smell"". https://chlorine.americanchemistry.com/Science-Center/Chlorine-Compound-of-the-Month-Library/Chloramines-Understanding-Pool-Smell/.
- ↑ Holleman, A. F.; Wiberg, E. (2001). Inorganic Chemistry. San Diego: Academic Press. ISBN 978-0-12-352651-9.
- ↑ Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. ISBN 978-0-08-037941-8.
- ↑ Hawthorn, J.; Todd, J. P. (1955). "Some effects of oxygen on the mixing of bread doughs". Journal of the Science of Food and Agriculture 6 (9): 501–511. doi:10.1002/jsfa.2740060906. Bibcode: 1955JSFA....6..501H.
- ↑ White, G. C. (1999). The Handbook of Chlorination and Alternative Disinfectants (4th ed.). Wiley. p. 322. ISBN 978-0-471-29207-4.
- ↑ "Health Hazard Evaluation Report: Investigation of Employee Symptoms at an Indoor Water Park". NIOSH ENews 6 (4). August 2008. HETA 2007-0163-3062. https://www.cdc.gov/niosh/hhe/reports/pdfs/2007-0163-3062.pdf.
- ↑ Thénard J. L.; Berthollet C. L. (1813). "Report on the work of Pierre Louis Dulong". Annales de Chimie et de Physique 86 (6): 37–43.
- ↑ Thomas, J.M. (1991). Michael Faraday and The Royal Institution: The Genius of Man and Place (PBK). CRC Press. p. 17. ISBN 978-0-7503-0145-9. https://books.google.com/books?id=GN70U1tTe_EC&pg=PA17.
Further reading
- Jander, J. (1976). Recent Chemistry and Structure Investigation of Nitrogen Triiodide, Tribromide, Trichloride, and Related Compounds. Advances in Inorganic Chemistry and Radiochemistry. 19. 1–63. doi:10.1016/S0065-2792(08)60070-9. ISBN 9780120236190.
- Kovacic, P.; Lowery, M. K.; Field, K. W. (1970). "Chemistry of N-Bromamines and N-Chloramines". Chemical Reviews 70 (6): 639–665. doi:10.1021/cr60268a002.
- Hartl, H.; Schöner, J.; Jander, J.; Schulz, H. (1975). "Die Struktur des Festen Stickstofftrichlorids (−125 °C)". Zeitschrift für Anorganische und Allgemeine Chemie 413 (1): 61–71. doi:10.1002/zaac.19754130108.
- Cazzoli, G.; Favero, P. G.; Dal Borgo, A. (1974). "Molecular Structure, Nuclear Quadrupole Coupling Constant and Dipole Moment of Nitrogen Trichloride from Microwave Spectroscopy". Journal of Molecular Spectroscopy 50 (1–3): 82–89. doi:10.1016/0022-2852(74)90219-7. Bibcode: 1974JMoSp..50...82C.
- Bayersdorfer, L.; Engelhardt, U.; Fischer, J.; Höhne, K.; Jander, J. (1969). "Untersuchungen an Stickstoff–Chlor-Verbindungen. V. Infrarot- und RAMAN-Spektren von Stickstofftrichlorid". Zeitschrift für Anorganische und Allgemeine Chemie 366 (3–4): 169–179. doi:10.1002/zaac.19693660308.
External links
| NH3 | He(N2)11 | ||||||||||||||||
| Li3N | Be3N2 | BN | β-C3N4 g-C3N4 |
N2 | NxOy | NF3 | Ne | ||||||||||
| Na3N | Mg3N2 | AlN | Si3N4 | PN P3N5 |
SxNy SN S4N4 |
NCl3 | Ar | ||||||||||
| K3N | Ca3N2 | ScN | TiN | VN | CrN Cr2N |
MnxNy | FexNy | CoN | Ni3N | CuN | Zn3N2 | GaN | Ge3N4 | As | Se | NBr3 | Kr |
| Rb3N | Sr3N2 | YN | ZrN | NbN | β-Mo2N | Tc | Ru | Rh | PdN | Ag3N | CdN | InN | Sn | Sb | Te | NI3 | Xe |
| Cs3N | Ba3N2 | Hf3N4 | TaN | WN | Re | Os | Ir | Pt | Au | Hg3N2 | TlN | Pb | BiN | Po | At | Rn | |
| Fr3N | Ra3N | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Nh | Fl | Mc | Lv | Ts | Og | |
| ↓ | |||||||||||||||||
| La | CeN | Pr | Nd | Pm | Sm | Eu | GdN | Tb | Dy | Ho | Er | Tm | Yb | Lu | |||
| Ac | Th | Pa | UN | Np | Pu | Am | Cm | Bk | Cf | Es | Fm | Md | No | Lr | |||
 |


