Chemistry:Phenaridine
 | |
| Clinical data | |
|---|---|
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| Chemical and physical data | |
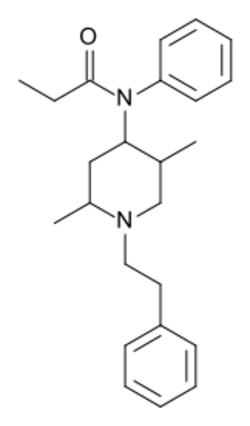
| Formula | C24H32N2O |
| Molar mass | 364.533 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Phenaridine (2,5-dimethylfentanyl) is an opioid analgesic that is an analogue of fentanyl. It was developed in 1972,[2] and is used for surgical anasthesia.[3][4]
Phenaridine has similar effects to fentanyl. It is slightly less potent than fentanyl in rats. Side effects of fentanyl analogs are similar to those of fentanyl itself, which include itching, nausea and potentially serious respiratory depression, which can be life-threatening. Irresponsible use of fentanyl analogues administrated in several times larger doses than recommended, have ended up in a death of hundreds of people throughout Europe and the former Soviet republics since the most recent resurgence in use began in Estonia in the early 2000s, and novel derivatives continue to appear.[5]
See also
- 3-Methylfentanyl
- 4-Fluorofentanyl
- α-Methylfentanyl
- Acetylfentanyl
- Furanylfentanyl
- List of fentanyl analogues
References
- ↑ Drug Enforcement Administration (February 2018). "Schedules of Controlled Substances:Temporary Placement of Fentanyl-Related Substances in Schedule I. Temporary amendment; temporary scheduling order". Federal Register 83 (25): 5188–92. PMID 29932611.
- ↑ "4-Anilidopiperidine analgesics. I. Synthesis and analgesic activity of certain ring-methylated 1-substituted 4-propananilidopiperidines". Journal of Pharmaceutical Sciences 62 (6): 983–6. June 1973. doi:10.1002/jps.2600620627. PMID 4712637.
- ↑ "[The new Soviet narcotic analgesic phenaridine as a component of general anesthesia during cancer surgery]" (in ru). Anesteziologiia I Reanimatologiia (1): 42–6. 1991. PMID 1862963.
- ↑ "[The evaluation of the analgesic action of phenaridine when combined with agents used in anesthesiological practice]" (in ru). Farmakologiia I Toksikologiia 54 (3): 17–20. 1991. PMID 1680749.
- ↑ "Fentanyls: Are we missing the signs? Highly potent and on the rise in Europe". The International Journal on Drug Policy 26 (7): 626–31. July 2015. doi:10.1016/j.drugpo.2015.04.003. PMID 25976511.
 |
