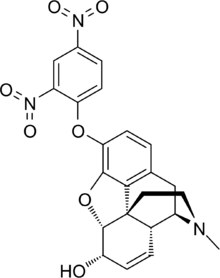
Chemistry:2,4-Dinitrophenylmorphine
From HandWiki
| |
| Names | |
|---|---|
| IUPAC name
3-(2,4-Dinitrophenoxy)-17-methyl-7,8-didehydro-4,5α-epoxymorphinan-6α-ol
| |
| Systematic IUPAC name
(4R,4aR,7S,7aR,12bS)-9-(2,4-Dinitrophenoxy)-3-methyl-2,3,4,4a,7,7a-hexahydro-1H-4,12-methano[1]benzofuro[3,2-e]isoquinolin-7-ol | |
| Identifiers | |
| |
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C23H21N3O7 | |
| Molar mass | 451.43 g/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
2,4-Dinitrophenylmorphine is an analog of morphine in which a hydroxyl group is substituted with a dinitro phenoxy group.[1][2]
Properties
Being an analog of morphine, it would be expected to have the same effects on the body as a typical opioid. Also, as dinitrophenol is a metabolic and respiratory stimulant, this morphine derivative was invented in Austria in 1931 as a narcotic analgesic with less potential to depress respiration.[3]
References
- ↑ Eddy, N. B.; Sumwalt, M. (1939). "Studies of Morphine, Codeine, and Their Derivatives. XV. 2,4-Dinitrophenylmorphine". Journal of Pharmacology and Experimental Therapeutics 67 (2): 127–141. http://jpet.aspetjournals.org/cgi/reprint/67/2/127.pdf.
- ↑ Huggins, R. A.; Bryan, A. R. (1951). "Some cardiovascular actions of dinitrophenylmorphine hydrochloride". Texas Reports on Biology and Medicine 9 (2): 314–318. PMID 14835466.
- ↑ Reynolds and Randall, "Morphine and Allied Drugs", U of Toronto, 1957 pp 203 et al.
 |

