Chemistry:N-Phenethylnormorphine
From HandWiki
Short description: Chemical compound
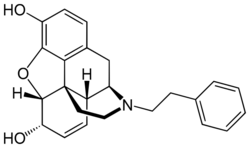 | |
| Identifiers | |
|---|---|
| |
| PubChem CID | |
| ChemSpider | |
| ChEMBL | |
| Chemical and physical data | |
| Formula | C24H25NO3 |
| Molar mass | 375.468 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
N-Phenethylnormorphine is an opioid analgesic drug derived from morphine by replacing the N-methyl group with β-phenethyl.[1] It is around eight to fourteen times more potent than morphine as a result of this modification,[2] in contrast to most other N-substituted derivatives of morphine, which are substantially less active, or act as antagonists.[3] Binding studies have helped to explain the increased potency of N-phenethylnormorphine, showing that the phenethyl group extends out to reach an additional binding point deeper inside the μ-opioid receptor cleft, analogous to the binding of the phenethyl group on fentanyl.[4][5]
See also
- 14-Cinnamoyloxycodeinone
- 14-Phenylpropoxymetopon
- 7-PET
- MR-2096
- N-Phenethyl-14-ethoxymetopon
- N-Phenethylnordesomorphine
- Phenomorphan
- RAM-378
- Ro4-1539
References
- ↑ "Notes: An Improved Synthesis of N-Phenethylnormorphine and Analogs". Journal of Organic Chemistry 23 (9): 1387–1388. 1958. doi:10.1021/jo01103a615.
- ↑ "The search for new analgesics". Journal of Chronic Diseases 4 (1): 59–71. July 1956. doi:10.1016/0021-9681(56)90007-8. PMID 13332043.
- ↑ "Analgesics. 1. Synthesis and analgesic properties of N-sec-alkyl- and N-tert-alkylnormorphines". Journal of Medicinal Chemistry 21 (5): 415–22. May 1978. doi:10.1021/jm00203a002. PMID 207868.
- ↑ "Molecular docking reveals a novel binding site model for fentanyl at the mu-opioid receptor". Journal of Medicinal Chemistry 43 (3): 381–91. February 2000. doi:10.1021/jm9903702. PMID 10669565.
- ↑ Molecular modeling of opioid receptor-ligand complexes. Progress in Medicinal Chemistry. 40. 2002. pp. 107–35. doi:10.1016/S0079-6468(08)70083-3. ISBN 9780444510549.
 |

