Chemistry:Ohmefentanyl
 | |
 | |
| Clinical data | |
|---|---|
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| Chemical and physical data | |
| Formula | C23H30N2O2 |
| Molar mass | 366.505 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
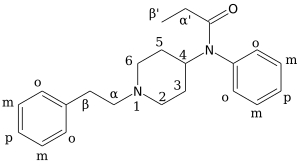
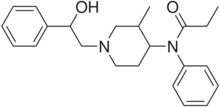
Ohmefentanyl (also known as β-hydroxy-3-methylfentanyl, OMF and RTI-4614-4)[1] is an extremely potent opioid analgesic drug which selectively binds to the µ-opioid receptor.[2][3]
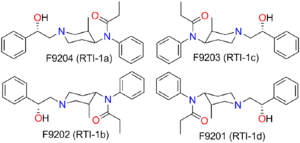
There are eight possible stereoisomers of ohmefentanyl. These stereoisomers are among the most potent μ-opioid receptor agonists known, comparable to super-potent opioids such as carfentanil and etorphine which are only legally used for tranquilizing large animals such as elephants in veterinary medicine. In mouse studies, the most active stereoisomer, 3R,4S,βS-ohmefentanyl, was 28 times more powerful as a painkiller than fentanyl, the chemical from which it is derived, and 6300 times more powerful than morphine.[4][5][6][7] Ohmefentanyl has three stereogenic centers and eight stereoisomers, which are named F9201–F9208. Researchers are studying the different pharmaceutical properties of these isomers.[8]
The 4″-fluoro analogue (i.e., substituted on the phenethyl ring) of the 3R,4S,βS isomer of ohmefentanyl is one of the most potent opioid agonists yet discovered, possessing an analgesic potency approximately 18,000 times that of morphine.[9] Other analogues with potency higher than that of ohmefentanyl itself include the 2′-fluoro derivative (i.e., substituted on the aniline phenyl ring), and derivatives where the N-propionyl group was replaced by N-methoxyacetyl or 2-furamide groups, or a carboethoxy group is added to the 4-position of the piperidine ring. The latter is listed as being up to 30,000 times more potent than morphine.[10]
Side effects of fentanyl analogues are similar to those of fentanyl itself, which include itching, nausea and potentially serious respiratory depression, which can be life-threatening. Illicitly used fentanyl analogues have killed hundreds of people throughout Europe and the former Soviet republics since the most recent resurgence in use began in Estonia in the early 2000s, and novel derivatives continue to appear.[11]
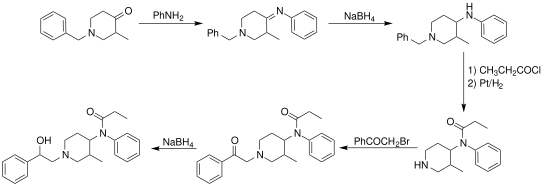
Synthesis
See also
References
- ↑ "RTI-4614-4: an analog of (+)-cis-3-methylfentanyl with a 27,000-fold binding selectivity for mu versus delta opioid binding sites". Life Sciences 48 (23): PL111–PL116. April 1991. doi:10.1016/0024-3205(91)90346-D. PMID 1646357.
- ↑ "Enantiomers of diastereomeric cis-N-[1-(2-hydroxy-2-phenylethyl)- 3-methyl-4-piperidyl]-N-phenylpropanamides: synthesis, X-ray analysis, and biological activities". Journal of Medicinal Chemistry 38 (9): 1547–1557. April 1995. doi:10.1021/jm00009a015. PMID 7739013.
- ↑ "Stereoisomers of N-[1-hydroxy-(2-phenylethyl)-3-methyl-4-piperidyl]- N-phenylpropanamide: synthesis, stereochemistry, analgesic activity, and opioid receptor binding characteristics". Journal of Medicinal Chemistry 38 (18): 3652–3659. September 1995. doi:10.1021/jm00018a026. PMID 7658453.
- ↑ H. D. Banks, C. P. Ferguson (September 1988). "The Metabolites of Fentanyl and its Derivatives". U.S. Army Chemical Research, Development and Engineering Center, Aberdeen Proving Ground, MD. http://apps.dtic.mil/dtic/tr/fulltext/u2/a199595.pdf.
- ↑ "Studies on synthesis and relationship between analgesic activity and receptor affinity for 3-methyl fentanyl derivatives". Scientia Sinica 24 (5): 710–720. May 1981. PMID 6264594.
- ↑ "[Studies on potent analgesics. VII. Synthesis and analgesic activity of diastereoisomers of 1-beta-hydroxy-3-methylfentanyl (7302) and related compounds]". Yao Xue Xue Bao = Acta Pharmaceutica Sinica 18 (12): 900–904. December 1983. PMID 6679170.
- ↑ "Comparison of physical dependence of ohmefentanyl stereoisomers in mice". Life Sciences 67 (2): 113–120. June 2000. doi:10.1016/S0024-3205(00)00617-2. PMID 10901279.
- ↑ "Effect of chronic treatment of ohmefentanyl stereoisomers on cyclic AMP formation in Sf9 insect cells expressing human mu-opioid receptors". Life Sciences 74 (24): 3001–3008. April 2004. doi:10.1016/j.lfs.2003.10.027. PMID 15051423.
- ↑ "Synthesis and analgesic activity of stereoisomers of cis-fluoro-ohmefentanyl". Die Pharmazie 58 (5): 300–302. May 2003. PMID 12779044. http://www.ingentaconnect.com/contentone/govi/pharmaz/2003/00000058/00000005/art00002.
- ↑ "Ohmefentanyl and its stereoisomers: Chemistry and pharmacology". Current Medicinal Chemistry 4 (4): 247–270. August 1997. doi:10.2174/0929867304666220313115017. ISSN 0929-8673.
- ↑ "Fentanyls: Are we missing the signs? Highly potent and on the rise in Europe". The International Journal on Drug Policy 26 (7): 626–631. July 2015. doi:10.1016/j.drugpo.2015.04.003. PMID 25976511.
External links
- Ohmefentanyl at the US National Library of Medicine Medical Subject Headings (MeSH)
 |