Chemistry:Testosterone enantate benzilic acid hydrazone
 | |
| Clinical data | |
|---|---|
| Trade names | Climacteron, Lactimex, Lactostat |
| Other names | Testosterone enantate benziloylhydrazone; Testosterone 17β-enantate 3-benzilic acid hydrazone; TEBH; TEBAH; TEBaH |
| Routes of administration | Intramuscular injection[1][2] |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| Chemical and physical data | |
| Formula | C40H52N2O4 |
| Molar mass | 624.866 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
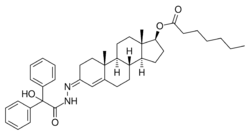
Testosterone enantate benzilic acid hydrazone (TEBH), or testosterone 17β-enantate 3-benzilic acid hydrazone, is a synthetic, injected androgen/anabolic steroid and an androgen ester – specifically, the C17β enantate (heptanoate) ester and C3 benzilic acid hydrazone of testosterone.[1][2] It was previously marketed in combination with estradiol benzoate and estradiol dienantate under the brand names Climacteron, Lactimex, and Lactostat.[2][3][4] Clinical studies have assessed this formulation.[5][6][7][8][9][10][11][2] TEBH was first described in the scientific literature in 1959.[12] It is a very long-lasting prodrug of testosterone when administered in oil via intramuscular injection.[13][14][15]
Chemistry
Chemical synthesis of TEBH has been described.[16]
See also
- List of androgen esters § Testosterone esters
- Hydroxyprogesterone heptanoate benzilic acid hydrazone
References
- ↑ 1.0 1.1 J. Elks (14 November 2014). The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. Springer. pp. 641–. ISBN 978-1-4757-2085-3. https://books.google.com/books?id=0vXTBwAAQBAJ&pg=PA641.
- ↑ 2.0 2.1 2.2 2.3 "The safety of testosterone therapy in women". J Obstet Gynaecol Can 34 (9): 859–865. September 2012. doi:10.1016/S1701-2163(16)35385-3. PMID 22971455.
- ↑ Geburtshilfe und Frauenheilkunde: Ergebnisse der Forschung für die Praxis. Georg Thieme Verlag.. 1969. https://books.google.com/books?id=_-owAAAAIAAJ.
- ↑ Zentralblatt für Gynäkologie. J. A. Barth. 1971. https://books.google.com/books?id=uNkwAAAAIAAJ.
- ↑ "Effects of parenteral administration of estrogen and androgen on plasma hormone levels and hot flushes in the surgical menopause". Am. J. Obstet. Gynecol. 148 (5): 552–7. March 1984. doi:10.1016/0002-9378(84)90746-4. PMID 6367474.
- ↑ "Differential symptom response to parenteral estrogen and/or androgen administration in the surgical menopause". Am. J. Obstet. Gynecol. 151 (2): 153–60. January 1985. doi:10.1016/0002-9378(85)90001-8. PMID 3881960.
- ↑ "Changes in sexual behavior as a function of plasma sex steroid levels in post-menopausal women". Maturitas 7 (3): 225–33. September 1985. doi:10.1016/0378-5122(85)90044-1. PMID 4079822.
- ↑ "Sex steroids and affect in the surgical menopause: a double-blind, cross-over study". Psychoneuroendocrinology 10 (3): 325–35. 1985. doi:10.1016/0306-4530(85)90009-5. PMID 3903822.
- ↑ "Postmenopausal estrogen and androgen replacement and lipoprotein lipid concentrations". Am. J. Obstet. Gynecol. 156 (2): 414–9. February 1987. doi:10.1016/0002-9378(87)90295-X. PMID 3826177.
- ↑ "Affective changes with estrogen and androgen replacement therapy in surgically menopausal women". J Affect Disord 14 (2): 177–87. 1988. doi:10.1016/0165-0327(88)90061-4. PMID 2966832.
- ↑ "Estrogen and/or androgen replacement therapy and cognitive functioning in surgically menopausal women". Psychoneuroendocrinology 13 (4): 345–57. 1988. doi:10.1016/0306-4530(88)90060-1. PMID 3067252.
- ↑ "The duration of activity of the benziloyl hydrazones of testosterone-17-heptanoate, estrone-3-heptanoate and 17 alpha-hydroxy-progesterone-17-heptanoate". Endocrinology 65 (3): 508–11. September 1959. doi:10.1210/endo-65-3-508. PMID 13828402.
- ↑ "The hormonal control of lactation". Clin Obstet Gynecol 3 (4): 1099–108. December 1960. doi:10.1097/00003081-196003040-00022. PMID 13723335.
- ↑ "The treatment of the menopause". Can Med Assoc J 86 (3): 113–4. January 1962. PMID 13901504.
- ↑ "Evaluation of a new preparation for the suppression of lactation". Can Med Assoc J 83 (24): 1240–2. December 1960. PMID 13752392.
- ↑ "Testosterone 17-Heptanoate 3-Benziloylhydrazone". J. Med. Chem. 7 (4): 573. July 1964. doi:10.1021/jm00334a042. PMID 14221154.
{{Navbox
| name = Androgens and antiandrogens | title = Androgens and antiandrogens | state = collapsed | listclass = hlist | groupstyle = text-align:center;
| group1 = Androgens
(incl. AAS)
| list1 =
| group2 = Antiandrogens | list2 = {{Navbox|child | groupstyle = text-align:center; | groupwidth = 9em;
| group1 = AR antagonists | list1 =
- Steroidal: Abiraterone acetate
- Canrenone
- Chlormadinone acetate
- Cyproterone acetate
- Delmadinone acetate
- Dienogest
- Drospirenone
- Medrogestone
- Megestrol acetate
- Nomegestrol acetate
- Osaterone acetate
- Oxendolone
- Potassium canrenoate
- Spironolactone
- Nonsteroidal: Apalutamide
- Bicalutamide
- Cimetidine
- Darolutamide
- Enzalutamide
- Flutamide
- Ketoconazole
- Nilutamide
- Seviteronel†
- Topilutamide (fluridil)
| group2 = Steroidogenesis| list2 =
inhibitors
| 5α-Reductase | |
|---|---|
| Others |
| group3 = Antigonadotropins | list3 =
- D2 receptor antagonists (prolactin releasers) (e.g., domperidone, metoclopramide, risperidone, haloperidol, chlorpromazine, sulpiride)
- Estrogens (e.g., bifluranol, [[diethylstilbestrol, estradiol, estradiol esters, ethinylestradiol, ethinylestradiol sulfonate, paroxypropione)
- GnRH agonists (e.g., leuprorelin)
- GnRH antagonists (e.g., cetrorelix)
- Progestogens (incl., chlormadinone acetate, [[cyproterone acetate, hydroxyprogesterone caproate, gestonorone caproate, [[Chemistry:Medroxyprogesterone medroxyprogesterone acetate, Chemistry:Megestrol acetate|megestrol acetate]])
| group4 = Others | list4 =
- Androstenedione immunogens: Androvax (androstenedione albumin)
- Ovandrotone albumin (Fecundin)
}}
| liststyle = background:#DDDDFF;| list3 =
- #WHO-EM
- ‡Withdrawn from market
- Clinical trials:
- †Phase III
- §Never to phase III
- See also
- Androgen receptor modulators
- Estrogens and antiestrogens
- Progestogens and antiprogestogens
- List of androgens/anabolic steroids
}}
 |

