Chemistry:Paliperidone palmitate
 | |
| Clinical data | |
|---|---|
| Trade names | Invega Hayfera, Invega Sustenna, Invega Trinza, others |
| Other names | PP; PP1M; PP3M; PP6M; JNS-010; RO-92670; RO92670 |
| MedlinePlus | a607005 |
| License data | |
| Routes of administration | Intramuscular |
| Drug class | Atypical antipsychotic |
| Legal status | |
| Legal status | |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| Chemical and physical data | |
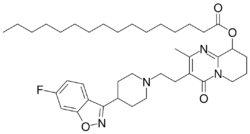
| Formula | C39H57FN4O4 |
| Molar mass | 664.907 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Paliperidone palmitate (PP), sold under the brand name Invega Sustenna among others, is an atypical antipsychotic which is used in the treatment of schizophrenia and schizoaffective disorder.[1][6][7] It is an antipsychotic ester – specifically the palmitate ester of paliperidone – and acts as a long-lasting form of paliperidone.[7] Paliperidone palmitate is formulated as an aqueous suspension, has a strong smell similar to alcohol, and is administered by intramuscular injection into deltoid or gluteal muscle once every 1, 3, or 6 months depending on the formulation.[7][8][6] A formulation for injection once every 6 months is also pending regulatory approval as of September 2021.[6][9]
With the once-monthly formulation of paliperidone palmitate, the time to peak is 13 days and the elimination half-life is 25 to 49 days, and with the 3-month formulation, the time to peak is 30 to 33 days and the half-life is 84 to 95 days via deltoid muscle and 118 to 139 days gluteally.[10] The peak-to-trough ratio of paliperidone palmitate at steady state ranges from 1.56 to 1.70 with the 1- and 3-month formulations.[10] The 3-month formulation of paliperidone palmitate has larger crystal sizes than the 1-month formulation and this allows for its longer duration.[11] No pharmacokinetic data for the 6-month formulation has been released as of January 2021.[11]
| Medication | Brand name | Class | Vehicle | Dosage | Tmax | t1/2 single | t1/2 multiple | logPc | Ref |
|---|---|---|---|---|---|---|---|---|---|
| Aripiprazole lauroxil | Aristada | Atypical | Watera | 441–1064 mg/4–8 weeks | 24–35 days | ? | 54–57 days | 7.9–10.0 | |
| Aripiprazole monohydrate | Abilify Maintena | Atypical | Watera | 300–400 mg/4 weeks | 7 days | ? | 30–47 days | 4.9–5.2 | |
| Bromperidol decanoate | Impromen Decanoas | Typical | Sesame oil | 40–300 mg/4 weeks | 3–9 days | ? | 21–25 days | 7.9 | [12] |
| Clopentixol decanoate | Sordinol Depot | Typical | Viscoleob | 50–600 mg/1–4 weeks | 4–7 days | ? | 19 days | 9.0 | [13] |
| Flupentixol decanoate | Depixol | Typical | Viscoleob | 10–200 mg/2–4 weeks | 4–10 days | 8 days | 17 days | 7.2–9.2 | [13][14] |
| Fluphenazine decanoate | Prolixin Decanoate | Typical | Sesame oil | 12.5–100 mg/2–5 weeks | 1–2 days | 1–10 days | 14–100 days | 7.2–9.0 | [15][16][17] |
| Fluphenazine enanthate | Prolixin Enanthate | Typical | Sesame oil | 12.5–100 mg/1–4 weeks | 2–3 days | 4 days | ? | 6.4–7.4 | [16] |
| Fluspirilene | Imap, Redeptin | Typical | Watera | 2–12 mg/1 week | 1–8 days | 7 days | ? | 5.2–5.8 | [18] |
| Haloperidol decanoate | Haldol Decanoate | Typical | Sesame oil | 20–400 mg/2–4 weeks | 3–9 days | 18–21 days | 7.2–7.9 | [19][20] | |
| Olanzapine pamoate | Zyprexa Relprevv | Atypical | Watera | 150–405 mg/2–4 weeks | 7 days | ? | 30 days | – | |
| Oxyprothepin decanoate | Meclopin | Typical | ? | ? | ? | ? | ? | 8.5–8.7 | |
| Paliperidone palmitate | Invega Sustenna | Atypical | Watera | 39–819 mg/4–12 weeks | 13–33 days | 25–139 days | ? | 8.1–10.1 | |
| Perphenazine decanoate | Trilafon Dekanoat | Typical | Sesame oil | 50–200 mg/2–4 weeks | ? | ? | 27 days | 8.9 | |
| Perphenazine enanthate | Trilafon Enanthate | Typical | Sesame oil | 25–200 mg/2 weeks | 2–3 days | ? | 4–7 days | 6.4–7.2 | [21] |
| Pipotiazine palmitate | Piportil Longum | Typical | Viscoleob | 25–400 mg/4 weeks | 9–10 days | ? | 14–21 days | 8.5–11.6 | [14] |
| Pipotiazine undecylenate | Piportil Medium | Typical | Sesame oil | 100–200 mg/2 weeks | ? | ? | ? | 8.4 | |
| Risperidone | Risperdal Consta | Atypical | Microspheres | 12.5–75 mg/2 weeks | 21 days | ? | 3–6 days | – | |
| Zuclopentixol acetate | Clopixol Acuphase | Typical | Viscoleob | 50–200 mg/1–3 days | 1–2 days | 1–2 days | 4.7–4.9 | ||
| Zuclopentixol decanoate | Clopixol Depot | Typical | Viscoleob | 50–800 mg/2–4 weeks | 4–9 days | ? | 11–21 days | 7.5–9.0 | |
| Note: All by intramuscular injection. Footnotes: a = Microcrystalline or nanocrystalline aqueous suspension. b = Low-viscosity vegetable oil (specifically fractionated coconut oil with medium-chain triglycerides). c = Predicted, from PubChem and DrugBank. Sources: Main: See template. | |||||||||
References
- ↑ 1.0 1.1 "Invega Sustenna- paliperidone palmitate injection". 31 January 2019. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=1af14e42-951d-414d-8564-5d5fce138554.
- ↑ "Invega Trinza- paliperidone palmitate injection, suspension, extended release". 31 January 2019. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=c39e65d7-fa44-4e4c-8b12-a654d3ed0eae.
- ↑ "Invega Hafyera- paliperidone palmitate injection, suspension, extended release". https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=6cd61892-d2cb-434d-83ed-5c1b2c4e7a0b.
- ↑ "Byannli EPAR". 28 April 2020. https://www.ema.europa.eu/en/medicines/human/EPAR/byannli-previously-paliperidone-janssen-cilag-international.
- ↑ "Byannli Product information". https://ec.europa.eu/health/documents/community-register/html/h1453.htm.
- ↑ 6.0 6.1 6.2 "Paliperidone palmitate - Johnson & Johnson - AdisInsight". https://adisinsight.springer.com/drugs/800040526.
- ↑ 7.0 7.1 7.2 "A review of paliperidone palmitate". Expert Review of Neurotherapeutics 12 (12): 1383–1397. December 2012. doi:10.1586/ern.12.137. PMID 23237346.
- ↑ "Paliperidone 3-Month Injection for Treatment of Schizophrenia: A Narrative Review". Frontiers in Psychiatry 12. 2021. doi:10.3389/fpsyt.2021.699748. PMID 34621193.
- ↑ "Paliperidone Palmitate 6-month (PP6M)". Archives of Psychiatry Research 57 (2): 229–232. 1 June 2021. doi:10.20471/dec.2021.57.02.13. ISSN 2671-1079.
- ↑ 10.0 10.1 "Pharmacokinetic Characteristics of Long-Acting Injectable Antipsychotics for Schizophrenia: An Overview". CNS Drugs 35 (1): 39–59. January 2021. doi:10.1007/s40263-020-00779-5. PMID 33507525.
- ↑ 11.0 11.1 "Therapeutic Drug Monitoring of Long-Acting Injectable Antipsychotic Drugs". Therapeutic Drug Monitoring 43 (1): 79–102. February 2021. doi:10.1097/FTD.0000000000000830. PMID 33196621.
- ↑ "Long-term treatment of chronic psychotics with bromperidol decanoate: clinical and pharmacokinetic evaluation.". Current Therapeutic Research 34 (1): 1–6. 1983.
- ↑ 13.0 13.1 "Clopenthixol and flupenthixol depot preparations in outpatient schizophrenics. III. Serum levels". Acta Psychiatrica Scandinavica. Supplementum 279: 41–54. 1980. doi:10.1111/j.1600-0447.1980.tb07082.x. PMID 6931472.
- ↑ 14.0 14.1 "Anxiolytic sedatives, hypnotics and neuroleptics.". Martindale: The Extra Pharmacopoeia (30th ed.). London: Pharmaceutical Press. 1993. pp. 364–623.
- ↑ "Future of depot neuroleptic therapy: pharmacokinetic and pharmacodynamic approaches". The Journal of Clinical Psychiatry 45 (5 Pt 2): 50–9. May 1984. PMID 6143748.
- ↑ 16.0 16.1 "Kinetics of fluphenazine after fluphenazine dihydrochloride, enanthate and decanoate administration to man". British Journal of Clinical Pharmacology 7 (4): 325–31. April 1979. doi:10.1111/j.1365-2125.1979.tb00941.x. PMID 444352.
- ↑ "Explaining the pharmacokinetics of fluphenazine through computer simulations. (Abstract.)". 19th Annual Midyear Clinical Meeting of the American Society of Hospital Pharmacists.. Dallas, Texas. 1984.
- ↑ "The pharmacology of fluspirilene (R 6218), a potent, long-acting and injectable neuroleptic drug". Arzneimittel-Forschung 20 (11): 1689–98. November 1970. PMID 4992598.
- ↑ "Haloperidol decanoate. A preliminary review of its pharmacodynamic and pharmacokinetic properties and therapeutic use in psychosis". Drugs 33 (1): 31–49. January 1987. doi:10.2165/00003495-198733010-00002. PMID 3545764.
- ↑ "Pharmacokinetics of haloperidol decanoate. A 2-year follow-up". International Pharmacopsychiatry 17 (4): 238–46. 1982. doi:10.1159/000468580. PMID 7185768.
- ↑ "On the pharmacokinetics of perphenazine: a clinical study of perphenazine enanthate and decanoate.". Current Therapeutic Research 36 (6): 1071–88. 1984.
External links
- "Paliperidone palmitate". Drug Information Portal. U.S. National Library of Medicine. https://druginfo.nlm.nih.gov/drugportal/name/paliperidone%20palmitate.
 |
