Chemistry:AB-CHFUPYCA
 | |
| Legal status | |
|---|---|
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| Chemical and physical data | |
| Formula | C22H29FN4O2 |
| Molar mass | 400.498 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
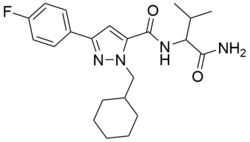
AB-CHFUPYCA (also AB-CHMFUPPYCA)[1] is a compound that was first identified as a component of synthetic cannabis products in Japan in 2015.[2][3] The name "AB-CHFUPYCA" is an acronym of its systematic name N-(1-Amino-3-methyl-1-oxoButan-2-yl)-1-(CycloHexylmethyl)-3-(4-FlUorophenyl)-1H-PYrazole-5-CarboxAmide. There are two known regioisomers of AB-CHFUPYCA: 3,5-AB-CHMFUPPYCA (pictured) and 5,3-AB-CHMFUPPYCA.[4] The article[1] refers to both 3,5-AB-CHMFUPPYCA and 5,3-AB-CHMFUPPYCA as AB-CHMFUPPYCA isomers, so AB-CHMFUPPYCA and AB-CHFUPYCA are not names for a unique chemical structure.
AB-CHFUPYCA contains some similar structural elements to other synthetic cannabinoids such as 5F-AB-FUPPYCA, JWH-307, JWH-030, JWH-147, AB-PINACA. It may be considered an analog of the traditional pyrazole cannabinoid receptor 1 antagonist rimonabant. The pharmacological properties of AB-CHFUPYCA have not been studied.
See also
References
- ↑ "The synthesis and characterization of the 'research chemical' N-(1-amino-3-methyl-1-oxobutan-2-yl)-1-(cyclohexylmethyl)-3-(4-fluorophenyl)-1H-pyrazole-5-carboxamide (3,5-AB-CHMFUPPYCA) and differentiation from its 5,3-regioisomer". Drug Testing and Analysis 8 (9): 920–9. September 2016. doi:10.1002/dta.1864. PMID 26360802. http://researchonline.ljmu.ac.uk/3149/1/DTA-15-0206.R1.pdf.
- ↑ "A new pyrazole-carboxamide type synthetic cannabinoid AB-CHFUPYCA [N-(1-amino-3-methyl-1-oxobutan-2-yl)-1-(cyclohexylmethyl)-3-(4-fluorophenyl)-1H-pyrazole-5-carboxamide] identified in illegal products". Forensic Toxicology 33 (2): 367–373. August 2015. doi:10.1007/s11419-015-0283-8.
- ↑ "In vitro metabolism of the synthetic cannabinoid 3,5-AB-CHMFUPPYCA and its 5,3-regioisomer and investigation of their thermal stability". Drug Testing and Analysis 9 (2): 311–316. February 2017. doi:10.1002/dta.1950. PMID 26888282. http://researchonline.ljmu.ac.uk/id/eprint/3141/3/dta1950-sup-0001-Supplementary.pdf.
- ↑ "5-fluoro-3,5-AB-PFUPPYCA". Cayman Chemical Company. https://www.caymanchem.com/product/18296.
 |
