Chemistry:WIN 55,212-2
 | |
| Legal status | |
|---|---|
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| ChemSpider | |
| UNII | |
| ChEBI | |
| ChEMBL | |
| Chemical and physical data | |
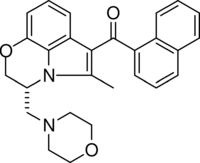
| Formula | C27H26N2O3 |
| Molar mass | 426.516 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |

WIN 55,212-2 is a chemical described as an aminoalkylindole derivative, which produces effects similar to those of cannabinoids such as tetrahydrocannabinol (THC) but has an entirely different chemical structure.[3][4][5]
WIN 55,212-2 is a potent cannabinoid receptor agonist[6] that has been found to be a potent analgesic[7] in a rat model of neuropathic pain.[8] It activates p42 and p44 MAP kinase via receptor-mediated signaling.[9]
At 5 μM WIN 55,212-2 inhibits ATP production in sperm in a CB1 receptor-dependent fashion.[10]
WIN 55,212-2, along with HU-210 and JWH-133, may prevent the inflammation caused by amyloid beta proteins involved in Alzheimer's disease, in addition to preventing cognitive impairment and loss of neuronal markers. This anti-inflammatory action is induced through agonist action at cannabinoid receptors, which prevents microglial activation that elicits the inflammation.
WIN 55,212-2 is a full agonist at the CB1 cannabinoid receptor (Ki = 1.9 nM) and has much higher affinity than THC (Ki = 41 nM) for this receptor.[11] WIN 55,212-2 is also an agonist of the PPARα and PPARγ nuclear receptors.[12]
WIN 55,212-2 reduces voluntary wheel running in laboratory mice, but with effects that depend on both genetic background and sex.[13]
In the United States, all CB1 receptor agonists of the 3-(1-naphthoyl)indole class such as WIN 55,212-2 are Schedule I Controlled Substances.[14] WIN 55,212-2 is illegal in the UK.[15]
WIN 55,212-2 is also a CB2 receptor agonist and thereby, like other cannabinoid CB2 agonists, found to significantly improve cardiac recovery after ischaemia/reperfusion (I/R) in the hearts of diabetic fatty rats, by restoring coronary perfusion pressure and heart rate to pre-ischaemic levels, by the restoration of the inducible nitric oxide synthase (iNOS)/endothelial nitric oxide synthase (eNOS) cardiac equilibrium.[16][17]
See also
- WIN 48,098 (Pravadoline)
- WIN 54,461 (6-Bromopravadoline)
- WIN 55,225 (JWH-200)
- WIN 56,098
References
- ↑ "Controlled Drugs and Substance Act - Schedule II". Justice Laws Website. Government of Canada. 18 March 2021. https://laws-lois.justice.gc.ca/eng/acts/c-38.8/page-14.html#docCont.
- ↑ "Cannabinoids reduce markers of inflammation and fibrosis in pancreatic stellate cells". PLOS ONE 3 (2): e1701. February 2008. doi:10.1371/journal.pone.0001701. PMID 18301776. Bibcode: 2008PLoSO...3.1701M.
- ↑ "Aminoalkylindole analogs: cannabimimetic activity of a class of compounds structurally distinct from delta 9-tetrahydrocannabinol". The Journal of Pharmacology and Experimental Therapeutics 263 (3): 1118–1126. December 1992. PMID 1335057.
- ↑ "The cannabinoid receptor agonist WIN 55,212-2 regulates glutamate transmission in rat cerebral cortex: an in vivo and in vitro study". Cerebral Cortex 11 (8): 728–733. August 2001. doi:10.1093/cercor/11.8.728. PMID 11459762.
- ↑ "In vitro metabolism of R(+)-[2,3-dihydro-5-methyl-3-[(morpholinyl)methyl]pyrrolo [1,2,3-de]1,4-benzoxazinyl]-(1-naphthalenyl) methanone mesylate, a cannabinoid receptor agonist". Drug Metabolism and Disposition 30 (10): 1077–1086. October 2002. doi:10.1124/dmd.30.10.1077. PMID 12228183.
- ↑ "Comparison of the pharmacology and signal transduction of the human cannabinoid CB1 and CB2 receptors". Molecular Pharmacology 48 (3): 443–450. September 1995. PMID 7565624.
- ↑ "An analgesia circuit activated by cannabinoids". Nature 395 (6700): 381–383. September 1998. doi:10.1038/26481. PMID 9759727. Bibcode: 1998Natur.395..381M.
- ↑ "The analgesic effects of R(+)-WIN 55,212-2 mesylate, a high affinity cannabinoid agonist, in a rat model of neuropathic pain". Neuroscience Letters 221 (2–3): 157–160. January 1997. doi:10.1016/S0304-3940(96)13308-5. PMID 9121688.
- ↑ "Activation of mitogen-activated protein kinases by stimulation of the central cannabinoid receptor CB1". The Biochemical Journal 312 ( Pt 2) (Pt 2): 637–641. December 1995. doi:10.1042/bj3120637. PMID 8526880.
- ↑ "Δ9-Tetrahydrocannabinol (Δ9-THC) attenuates mouse sperm motility and male fecundity". British Journal of Pharmacology 165 (8): 2575–2583. April 2012. doi:10.1111/j.1476-5381.2011.01506.x. PMID 21615727.
- ↑ "Aminoalkylindole binding in rat cerebellum: selective displacement by natural and synthetic cannabinoids". The Journal of Pharmacology and Experimental Therapeutics 264 (3): 1352–1363. March 1993. PMID 8450470.
- ↑ "An update on PPAR activation by cannabinoids". British Journal of Pharmacology 173 (12): 1899–1910. June 2016. doi:10.1111/bph.13497. PMID 27077495.
- ↑ "Sex differences in cannabinoid receptor-1 (CB1) pharmacology in mice selectively bred for high voluntary wheel-running behavior". Pharmacology, Biochemistry, and Behavior 101 (4): 528–537. June 2012. doi:10.1016/j.pbb.2012.02.017. PMID 22405775.
- ↑ : Schedules of controlled substances
- ↑ "The Misuse of Drugs Act 1971 (Amendment) Order 2013". legislation.gov.uk. http://www.legislation.gov.uk/uksi/2013/239/article/4/made.
- ↑ "Cannabinoid/agonist WIN 55,212-2 reduces cardiac ischaemia–reperfusion injury in Zucker diabetic fatty rats: role of CB2 receptors and iNOS/eNOS". Diabetes/Metabolism Research and Reviews 27 (4): 331–340. May 2011. doi:10.1002/dmrr.1176. PMID 21309057.
- ↑ "Delta-9-tetrahydrocannabinol protects cardiac cells from hypoxia via CB2 receptor activation and nitric oxide production". Molecular and Cellular Biochemistry 283 (1–2): 75–83. February 2006. doi:10.1007/s11010-006-2346-y. PMID 16444588.
Further reading
- "The cannabinoid WIN 55,212-2 inhibits transient receptor potential vanilloid 1 (TRPV1) and evokes peripheral antihyperalgesia via calcineurin". Proceedings of the National Academy of Sciences of the United States of America 103 (30): 11393–11398. July 2006. doi:10.1073/pnas.0603861103. PMID 16849427. Bibcode: 2006PNAS..10311393P.
- "Prevention of Alzheimer's disease pathology by cannabinoids: neuroprotection mediated by blockade of microglial activation". The Journal of Neuroscience 25 (8): 1904–1913. February 2005. doi:10.1523/JNEUROSCI.4540-04.2005. PMID 15728830.
External links
- "Win 55,212-2 Data Sheet". Enzo Life Sciences. http://www.enzolifesciences.com/BML-CR105/win-55-212-2/.
 |
