Chemistry:CP 55,940
 | |
| Legal status | |
|---|---|
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| ChEMBL | |
| Chemical and physical data | |
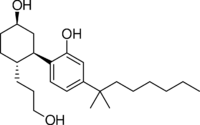
| Formula | C24H40O3 |
| Molar mass | 376.581 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
CP 55,940 is a synthetic cannabinoid which mimics the effects of naturally occurring THC (one of the psychoactive compounds found in cannabis). CP 55,940 was created by Pfizer in 1974 but was never marketed. It is currently used as a research tool to study the endocannabinoid system.[1]
Pharmacology
CP 55,940 is 45 times more potent than Δ9-THC, and fully antagonized by rimonabant (SR141716A).[2] It is considered a full agonist at both CB1 and CB2 receptors and has Ki values of 0.58 nM and 0.68 nM respectively, but is an antagonist at GPR55, the putative "CB3" receptor.[3] CP 55,940 binding has been detected in the cytosol of rat brain cerebral cortex.[4] It can upregulate 5-HT2A receptors in mice.[5]
In vitro studies
CP 55,940 induced cell death in NG 108-15 Mouse neuroblastoma x Rat glioma hybrid brain cancer (genetically engineered mouse x rat brain cancer) cells.[6][7]
In vivo studies
CP 55,940 showed protective effects on rat brain mitochondria upon paraquat exposure.[8]
It also showed neuroprotective effects by reducing intracellular calcium release and reducing hippocampal cell death in cultured neurons subjected to high levels of NMDA.[9]
See also
References
- ↑ "Cannabinoid receptors in the human brain: a detailed anatomical and quantitative autoradiographic study in the fetal, neonatal and adult human brain". Neuroscience 77 (2): 299–318. March 1997. doi:10.1016/s0306-4522(96)00428-9. PMID 9472392.
- ↑ "Characterization and distribution of binding sites for [3H]-SR 141716A, a selective brain (CB1) cannabinoid receptor antagonist, in rodent brain". Life Sciences 58 (15): 1239–47. 1996. doi:10.1016/0024-3205(96)00085-9. PMID 8614277.
- ↑ "Atypical responsiveness of the orphan receptor GPR55 to cannabinoid ligands". The Journal of Biological Chemistry 284 (43): 29817–27. October 2009. doi:10.1074/jbc.M109.050187. PMID 19723626.
- ↑ "Identification and characterization of a novel synthetic cannabinoid CP 55,940 binder in rat brain cytosol". Molecular and Cellular Biochemistry 181 (1–2): 21–27. April 1998. doi:10.1023/A:1006855504094. PMID 9562238.
- ↑ "Cannabinoid receptor agonists upregulate and enhance serotonin 2A (5-HT(2A)) receptor activity via ERK1/2 signaling". Synapse 67 (3): 145–59. March 2013. doi:10.1002/syn.21626. PMID 23151877.
- ↑ "Cytotoxicity of synthetic cannabinoids found in "Spice" products: the role of cannabinoid receptors and the caspase cascade in the NG 108-15 cell line". Toxicology Letters 207 (1): 12–7. November 2011. doi:10.1016/j.toxlet.2011.08.021. PMID 21907772.
- ↑ "General Cell Collection: NG108-15". Public Health England Culture Collections. http://www.hpacultures.org.uk/products/celllines/generalcell/detail.jsp?refId=88112302&collection=ecacc_gc.
- ↑ "Protective effects of the synthetic cannabinoids CP55,940 and JWH-015 on rat brain mitochondria upon paraquat exposure". Neurochemical Research 35 (9): 1323–32. September 2010. doi:10.1007/s11064-010-0188-1. PMID 20514518.
- ↑ "Cannabinoids produce neuroprotection by reducing intracellular calcium release from ryanodine-sensitive stores". Neuropharmacology 48 (8): 1086–96. June 2005. doi:10.1016/j.neuropharm.2005.01.005. PMID 15910885.
 |
