Chemistry:Tetrahydrocannabihexol
From HandWiki
Short description: Chemical compound
 | |
 | |
| Identifiers | |
|---|---|
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| Chemical and physical data | |
| Formula | C22H32O2 |
| Molar mass | 328.496 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
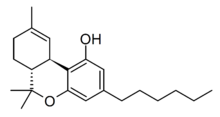
Tetrahydrocannabihexol (Δ9-THCH, Δ9-Parahexyl, n-Hexyl-Δ9-THC) is a phytocannabinoid, the hexyl homologue of tetrahydrocannabinol (THC) which was first isolated from Cannabis plant material in 2020 along with the corresponding hexyl homologue of cannabidiol,[1][2] though it had been known for several decades prior to this as an isomer of the synthetic cannabinoid parahexyl.[3] Another isomer Δ8-THCH is also known as a synthetic cannabinoid under the code number JWH-124,[4][5] though it is unclear whether this occurs naturally in Cannabis, but likely is due to Delta-8-THC itself being a degraded form of Delta-9-THC. THC-Hexyl can be synthesized from 4-Hexylresorcinol and was studied by Roger Adams as early as 1942.[6]
See also
- Cannabidiphorol
- Hexahydrocannabihexol
- Tetrahydrocannabiorcol
- Tetrahydrocannabivarin
- Tetrahydrocannabutol
- Tetrahydrocannabiphorol
References
- ↑ "Identification of a new cannabidiol n-hexyl homolog in a medicinal cannabis variety with an antinociceptive activity in mice: cannabidihexol". Scientific Reports 10 (1): 22019. December 2020. doi:10.1038/s41598-020-79042-2. PMID 33328530. Bibcode: 2020NatSR..1022019L.
- ↑ "Cannabinoids, Phenolics, Terpenes and Alkaloids of Cannabis". Molecules 26 (9): 2774. May 2021. doi:10.3390/molecules26092774. PMID 34066753.
- ↑ "Metabolism of n-hexyl-homologues of delta-8-tetrahydrocannabinol and delta-9-tetrahydrocannabinol in the mouse". European Journal of Drug Metabolism and Pharmacokinetics 13 (3): 165–76. 1988. doi:10.1007/BF03189936. PMID 2853671.
- ↑ "Manipulation of the tetrahydrocannabinol side chain delineates agonists, partial agonists, and antagonists". The Journal of Pharmacology and Experimental Therapeutics 290 (3): 1065–79. September 1999. PMID 10454479.
- ↑ "The Structure-Function Relationships of Classical Cannabinoids: CB1/CB2 Modulation". Perspectives in Medicinal Chemistry 8: 17–39. 2016. doi:10.4137/PMC.S32171. PMID 27398024.
- ↑ https://pubs.acs.org/doi/abs/10.1021/ja01255a061
 |
