Chemistry:THC hemisuccinate
From HandWiki
Short description: Synthetic derivative of tetrahydrocannabinol
 | |
| Identifiers | |
|---|---|
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| Chemical and physical data | |
| Formula | C25H34O5 |
| Molar mass | 414.542 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
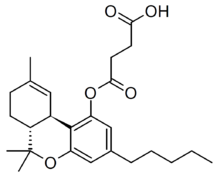
THC hemisuccinate (Δ9-THC-O-hemisuccinate, Dronabinol hemisuccinate) is a synthetic derivative of tetrahydrocannabinol, developed in the 1990s. It is a water-soluble prodrug ester which is converted into THC inside the body, and was developed to overcome the poor bioavailability of THC when taken by non-inhaled routes of administration.[1][2][3] In medical applications it has mainly been formulated as rectal suppositories.[4][5][6]
See also
References
- ↑ "Rectal bioavailability of delta-9-tetrahydrocannabinol from various esters". Pharmacology, Biochemistry, and Behavior 40 (3): 497–502. November 1991. doi:10.1016/0091-3057(91)90353-4. PMID 1666913.
- ↑ "Rectal bioavailability of delta-9-tetrahydrocannabinol from the hemisuccinate ester in monkeys". Journal of Pharmaceutical Sciences 80 (10): 942–5. October 1991. doi:10.1002/jps.2600801008. PMID 1664466.
- ↑ "Preparation and characterization of inclusion complexes of a hemisuccinate ester prodrug of delta9-tetrahydrocannabinol with modified beta-cyclodextrins". AAPS PharmSciTech 11 (2): 509–17. June 2010. doi:10.1208/s12249-010-9401-4. PMID 20333489.
- ↑ "Δ9-THC Hemisuccinate in Suppository Form as an Alternative to Oral and Smoked THC". Marihuana and Medicine. Totowa, NJ: Humana Press. 1999. doi:10.1007/978-1-59259-710-9_13.
- ↑ "Analgesic and Reinforcing Properties of Δ9-THC-Hemisuccinate in Adjuvant-Arthritic Rats.". Journal of Cannabis Therapeutics 1 (3–4): 171–182. 2001. doi:10.1300/J175v01n03_11.
- ↑ "Pharmacokinetics and Tolerability of Δ9-THC-Hemisuccinate in a Suppository Formulation as an Alternative to Capsules for the Systemic Delivery of Δ9-THC". Medical Cannabis and Cannabinoids 1 (1): 44–53. June 2018. doi:10.1159/000489037. PMID 34676321.
 |
