Chemistry:ADB-FUBHQUCA
From HandWiki
Short description: Chemical compound
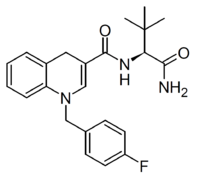 | |
| Identifiers | |
|---|---|
| |
| PubChem CID | |
| Chemical and physical data | |
| Formula | C23H26FN3O2 |
| Molar mass | 395.478 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
ADB-FUBHQUCA is a synthetic cannabinoid receptor agonist that has been sold as a designer drug, first reported in 2022.[1] It is related to the previously reported compound ADB-FUBICA but with the central indole ring system expanded to a 1,4-dihydroquinoline structure. This breaks the aromaticity of the ring system, and ADB-FUBHQUCA is relatively low in potency compared to related compounds where the aromatic core is retained.[2][3][4][5][6][7][8][9][10][11][12][13]
See also
References
- ↑ "New Substance Report. 118. ADB-FUBHQUCA" (in ru). AIPSIN monitoring. 18 February 2022. https://aipsin.com/newsubstance/806/.
- ↑ "Cumyl-PeGaClone and other recently encountered synthetic cannabinoid receptor agonists. A review of the evidence on their use and harms.". Advisory Council on the Misuse of Drugs. Government Digital Service, UK Government. 2022. https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/1159142/Cumyl-pegalone_and_other_uncontrolled_SCRA_report-FINAL.pdf.
- ↑ "From JWH-018 to OXIZIDS: Structural evolution of synthetic cannabinoids in the European Union from 2008 to present day". Drug Testing and Analysis 15 (4): 378–387. April 2023. doi:10.1002/dta.3422. PMID 36507715.
- ↑ "Synthesis and biological evaluation of 1,8-naphthyridin-4(1H)-on-3-carboxamide derivatives as new ligands of cannabinoid receptors". Bioorganic & Medicinal Chemistry 12 (8): 1921–1933. April 2004. doi:10.1016/j.bmc.2004.01.035. PMID 15051060.
- ↑ "Novel 4-oxo-1,4-dihydroquinoline-3-carboxamide derivatives as new CB2 cannabinoid receptors agonists: synthesis, pharmacological properties and molecular modeling". Journal of Medicinal Chemistry 49 (1): 70–79. January 2006. doi:10.1021/jm050467q. PMID 16392793.
- ↑ "Design, synthesis, and biological evaluation of new 1,8-naphthyridin-4(1H)-on-3-carboxamide and quinolin-4(1H)-on-3-carboxamide derivatives as CB2 selective agonists". Journal of Medicinal Chemistry 49 (20): 5947–5957. October 2006. doi:10.1021/jm0603466. PMID 17004710.
- ↑ "Pharmacomodulations around the 4-oxo-1,4-dihydroquinoline-3-carboxamides, a class of potent CB2-selective cannabinoid receptor ligands: consequences in receptor affinity and functionality". Journal of Medicinal Chemistry 50 (22): 5471–5484. November 2007. doi:10.1021/jm070387h. PMID 17915849.
- ↑ "New 1,8-naphthyridine and quinoline derivatives as CB2 selective agonists". Bioorganic & Medicinal Chemistry Letters 17 (23): 6505–6510. December 2007. doi:10.1016/j.bmcl.2007.09.089. PMID 17942307.
- ↑ "Investigations on the 4-quinolone-3-carboxylic acid motif. 2. Synthesis and structure-activity relationship of potent and selective cannabinoid-2 receptor agonists endowed with analgesic activity in vivo". Journal of Medicinal Chemistry 51 (16): 5075–5084. August 2008. doi:10.1021/jm800552f. PMID 18680276.
- ↑ "Rational design, synthesis, and pharmacological properties of new 1,8-naphthyridin-2(1H)-on-3-carboxamide derivatives as highly selective cannabinoid-2 receptor agonists". Journal of Medicinal Chemistry 52 (12): 3644–3651. June 2009. doi:10.1021/jm801563d. PMID 19435366.
- ↑ "Investigations on the 4-quinolone-3-carboxylic acid motif. 3. Synthesis, structure-affinity relationships, and pharmacological characterization of 6-substituted 4-quinolone-3-carboxamides as highly selective cannabinoid-2 receptor ligands". Journal of Medicinal Chemistry 53 (16): 5915–5928. August 2010. doi:10.1021/jm100123x. PMID 20718492.
- ↑ "Investigations on the 4-quinolone-3-carboxylic acid motif. 4. Identification of new potent and selective ligands for the cannabinoid type 2 receptor with diverse substitution patterns and antihyperalgesic effects in mice". Journal of Medicinal Chemistry 54 (15): 5444–5453. August 2011. doi:10.1021/jm200476p. PMID 21702498.
- ↑ "Investigations on the 4-quinolone-3-carboxylic acid motif. 6. Synthesis and pharmacological evaluation of 7-substituted quinolone-3-carboxamide derivatives as high affinity ligands for cannabinoid receptors". European Journal of Medicinal Chemistry 58: 30–43. December 2012. doi:10.1016/j.ejmech.2012.09.035. PMID 23085772.
 |

