Chemistry:Allenolic acid
 | |
| Identifiers | |
|---|---|
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| ChEBI | |
| Chemical and physical data | |
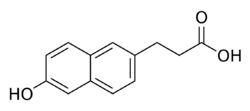
| Formula | C13H12O3 |
| Molar mass | 216.236 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Allenolic acid, or allenoic acid, is a synthetic,[1] nonsteroidal estrogen discovered in 1947 or 1948 that, although studied clinically,[2] was never marketed.[3][4][5] It is an open-ring or seco-analogue of steroidal estrogens like estrone and equilenin.[6][7][8] The compound was named after Edgar Allen, one of the pioneers in estrogen research.[9][10] Although described as an estrogen, allenolic acid probably is totally inactive at the receptor, whereas a derivative, allenestrol (α,α-dimethyl-β-ethylallenolic acid), is reported to be a potent estrogen.[11] Another derivative of allenolic acid (specifically 6-methoxy-allenestrol), methallenestril (brand name Vallestril), is also a potent estrogen and, in contrast to allenolic acid and allenestrol, has been marketed.[12][13][14][15]
See also
References
- ↑ The Menopause and Postmenopause: The Proceedings of an International Symposium held in Rome, June 1979. Springer Science & Business Media. 6 December 2012. pp. 110–. ISBN 978-94-011-7230-1. https://books.google.com/books?id=wZF9CAAAQBAJ&pg=PA110.
- ↑ American Practitioner and Digest of Treatment. Lippincott.. January 1951. p. 443. https://books.google.com/books?id=px8gAQAAMAAJ.
- ↑ Reversibility of Chronic Degenerative Disease and Hypersensitivity, Volume 1: Regulating Mechanisms of Chemical Sensitivity. CRC Press. 18 June 2010. pp. 464–. ISBN 978-1-4398-1345-4. https://books.google.com/books?id=--U8r_WYVIcC&pg=PA464.
- ↑ "Estrogens and antiestrogens". Gynecologic Investigation 3 (1): 2–29. 1972. doi:10.1159/000301742. PMID 4347198.
- ↑ "Experimental and clinical studies on a new synthetic estrogen, an allenolic acid derivative, vallestril". American Journal of Obstetrics and Gynecology 74 (3): 635–50. September 1957. doi:10.1016/0002-9378(57)90519-7. PMID 13458265.
- ↑ Indian Journal of Chemistry: Organic including medicinal. Council of Scientific & Industrial Research.. 1980. p. 886. https://books.google.com/books?id=7ZdVAAAAMAAJ.
- ↑ Endocrines, Vitamins, and Some Common Metabolic Disorders. Dar al-Maaref. 1963. p. 194. https://books.google.com/books?id=xXhFAAAAYAAJ.
- ↑ Morrison, James D. (November 1983). Stereodifferentiating addition reactions. Academic Press. p. v. ISBN 978-0-12-507702-6. https://books.google.com/books?id=Iw3wAAAAMAAJ.
- ↑ Thompson, Willard Owen (1953). The Year Book of Endocrinology. Year Book Medical Publishers. p. 292. https://books.google.com/books?id=vUtEAQAAIAAJ.
- ↑ American Practitioner and Digest of Treatment. Lippincott.. January 1956. https://books.google.com/books?id=DiggAQAAMAAJ.
- ↑ "753. Oestrogenic carboxylic acids. Part II. Open-chain analogues of doisynolic acid". Journal of the Chemical Society (Resumed): 3714. 1959. doi:10.1039/jr9590003714. ISSN 0368-1769.
- ↑ Journal of the Japanese Obstetrical & Gynecological Society. 1958. p. 83. https://books.google.com/books?id=tRcHAQAAIAAJ.
- ↑ Steroid Biochemistry. Academic Press. 1970. p. 144. ISBN 9780123366504. https://books.google.com/books?id=gAlrAAAAMAAJ.
- ↑ The Effects of the Sulfonylureas and Related Compounds in Experimental and Clinical Diabetes. The Academy. 1957. p. 681. https://books.google.com/books?id=b64PAAAAIAAJ.
- ↑ Antineoplastic and Immunosuppressive Agents. Springer Science & Business Media. 27 November 2013. pp. 106–. ISBN 978-3-642-65806-8. https://books.google.com/books?id=aU_oCAAAQBAJ&pg=PA106.
 |
