Chemistry:Quinestrol
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Estrovis, others |
| Other names | Quinoestrol; Quinestrenol; Quinoestrenol; Ethinylestradiol 3-cyclopentyl ether; EECPE; EE2CPE; W-3566; 3-(Cyclopentyloxy)-17α-ethynylestra-1,3,5(10)-trien-17β-ol |
| AHFS/Drugs.com | Micromedex Detailed Consumer Information |
| Routes of administration | By mouth |
| Drug class | Estrogen; Estrogen ether |
| ATC code |
|
| Pharmacokinetic data | |
| Elimination half-life | >120 hours (>5 days)[1] |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| Chemical and physical data | |
| Formula | C25H32O2 |
| Molar mass | 364.529 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Quinestrol, also known as ethinylestradiol cyclopentyl ether (EECPE), sold under the brand name Estrovis among others, is an estrogen medication which has been used in menopausal hormone therapy, hormonal birth control, and to treat breast cancer and prostate cancer.[2][3] It is taken once per week to once per month by mouth.[4][5][6][7]
Medical uses
Quinestrol has been used as the estrogen component in menopausal hormone therapy and in combined hormonal birth control.[2][3] It has also occasionally been used in the treatment of breast cancer and prostate cancer, as well as to suppress lactation.[2][3][8] On its own as an estrogen, quinestrol was taken once per week by mouth.[4] As a combined birth control pill, it was used together with quingestanol acetate and was taken once per month by mouth.[5][6][7]
Pharmacology
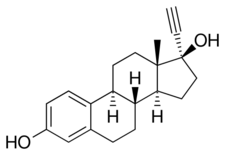
Quinestrol is a prodrug of ethinylestradiol (EE), with no estrogenic activity of its own.[3][9][10] It is taken orally and has prolonged activity following a single dose,[9][10] with a very long biological half-life of more than 120 hours (5 days) due to enhanced lipophilicity and storage in fat.[3][1] Because of its much longer half-life, quinestrol is two to three times as potent as EE.[3] Also because of its long half-life, quinestrol can be taken once a week or once a month.[3][4][5][6][7]
Following administration, quinestrol is absorbed via the lymphatic system, is stored in adipose tissue, and is gradually released from adipose tissue.[11]
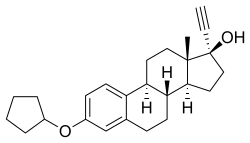
Chemistry
Quinestrol, also known as ethinylestradiol 3-cyclopentyl ether (EE2CPE), is a synthetic estrane steroid and a derivative of estradiol.[12][13] It is an estrogen ether, specifically the C3 cyclopentyl ether of ethinylestradiol (17α-ethynylestradiol).[12][13] Closely related estrogens include mestranol (ethinylestradiol 3-methyl ether) and ethinylestradiol sulfonate (EES; Turisteron; ethinylestradiol 3-isopropylsulfonate).[12][13]
History
Quinestrol was developed and introduced for medical use in the 1960s.[14]
Society and culture
Generic names
Quinestrol is the generic name of the drug and its INN, USAN, and BAN.[12][13][15][16] It is also known by its former developmental code name W-3566.[12][13][15][16]
Brand names
Quinestrol has been marketed under brand names including Agalacto-Quilea, Basaquines, Eston, Estrovis, Estrovister, Plestrovis, Qui-Lea, Soluna, and Yueketing, among others.[12][13][15][16]
Availability
Quinestrol was marketed as Estrovis in the United States by Parke-Davis and as Qui-Lea in Argentina ,[13] but is reportedly not currently marketed.[3] However, it does appear to still be available as an oral contraceptive in combination with progestins in Argentina and China .[16]
One tablet form available in China consists of 6 mg levonorgestrel and 3 mg quinestrol; it is used as a prescription "long-term" oral contraceptive, with one dose taken each month.[16][17] It is sold under various brand names including Yuèkětíng (Chinese: 悦可婷) and Àiyuè (Chinese: 艾悦). A version with the racemic norgestrel in place of levonorgestrel also appears to be available.[16]
Veterinary use
Rodents
The Chinese levonorgestrel/quinestrol 2:1 formula is known as EP-1 in veterinary practice. It is known to have some organ-specific effects on the Mongolian gerbil as measured by receptor mRNA expression.[18] Incorporated into baits at a concentration of 50 ppm, EP-1 has been used to control wild Mongolian gerbil populations with some success.[19]
References
- ↑ 1.0 1.1 "Pharmacology of Different Administration Routes-Oral vs Transdermal.". Estrogens and Antiestrogens II: Pharmacology and Clinical Application of Estrogens and Antiestrogen. Handbook of Experimental Pharmacology. 135 / 2. Springer Science & Business Media. 6 December 2012. pp. 248–. doi:10.1007/978-3-642-60107-1_14. ISBN 978-3-642-60107-1. https://books.google.com/books?id=wBvyCAAAQBAJ&pg=PA248.
- ↑ 2.0 2.1 2.2 "Quinestrol". Dictionary of Obstetrics and Gynecology. Walter de Gruyter. 1 January 1988. pp. 204–. ISBN 978-3-11-085727-6. https://books.google.com/books?id=EQlvzV9V7xIC&pg=PA204.
- ↑ 3.0 3.1 3.2 3.3 3.4 3.5 3.6 3.7 "Primary and secondary hypogonadism in women.". Hormone Replacement Therapy. Springer Science & Business Media. 1 June 1999. pp. 381–. ISBN 978-1-59259-700-0. https://books.google.com/books?id=ja2nBgAAQBAJ&pg=PA381.
- ↑ 4.0 4.1 4.2 "Biologic effects of natural and synthetic estrogens.". The Menopause. Springer Science & Business Media. 6 December 2012. pp. 60–. ISBN 978-1-4612-5525-3. https://books.google.com/books?id=z0LuBwAAQBAJ&pg=PA60.
- ↑ 5.0 5.1 5.2 "Contraception". Ovarian Function and its Disorders: Diagnosis and Therapy. Springer Science & Business Media. 6 December 2012. pp. 85,358,367. ISBN 978-94-009-8195-9. https://books.google.com/books?id=7IrpCAAAQBAJ&pg=PA367.
- ↑ 6.0 6.1 6.2 "Other Hormal Contraception Procedures". Human Fertility Control: Theory and Practice. Elsevier Science. 22 October 2013. pp. 92–94. ISBN 978-1-4831-6361-1. https://books.google.com/books?id=1lL0AgAAQBAJ&pg=PA92.
- ↑ 7.0 7.1 7.2 Chemical Contraception. Macmillan International Higher Education. 18 June 1974. pp. 61–. ISBN 978-1-349-02287-8. https://books.google.com/books?id=4FVdDwAAQBAJ&pg=PA61.
- ↑ The Breast: Morphology, Physiology, and Lactation. Elsevier Science. 2 December 2012. pp. 201–203. ISBN 978-0-323-15726-1. https://books.google.com/books?id=wYxirvD2X2IC&pg=PA201.
- ↑ 9.0 9.1 "Prolonged menstrual response of patients with gonadal failure following quinestrol administration". International Journal of Fertility 12 (2): 181–186. 1967. PMID 6033895.
- ↑ 10.0 10.1 "Prolonged oestrogenic activity in rats after single oral administration of ethinyloestradiol-3-cyclopentyl ether". The Journal of Pharmacy and Pharmacology 21 (4): 271–272. April 1969. doi:10.1111/j.2042-7158.1969.tb08247.x. PMID 4390151.
- ↑ "Current status of estrogen therapy for the menopause". Fertility and Sterility 37 (1): 5–25. January 1982. doi:10.1016/S0015-0282(16)45970-4. PMID 6277697.
- ↑ 12.0 12.1 12.2 12.3 12.4 12.5 The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. Springer. 14 November 2014. pp. 522–. ISBN 978-1-4757-2085-3. https://books.google.com/books?id=0vXTBwAAQBAJ&pg=PA522.
- ↑ 13.0 13.1 13.2 13.3 13.4 13.5 13.6 "Quinestrol". Index Nominum 2000: International Drug Directory. Taylor & Francis. January 2000. pp. 905–. ISBN 978-3-88763-075-1. https://books.google.com/books?id=5GpcTQD_L2oC&pg=PA905.
- ↑ Medical Gynaecology and Sociology. Medical and Scientific Services Limited. 1967. https://books.google.com/books?id=WfxxAAAAIAAJ. "[...] J. Fertil., 1967, 12, 2) contains 23 papers presented at a symposium on QUINESTROL. Quinestrol is a newly-developed synthetic steroid, and is the cyclo-pentyl ether of a ethinyl oestradiol."
- ↑ 15.0 15.1 15.2 Concise Dictionary of Pharmacological Agents: Properties and Synonyms. Springer Science & Business Media. 6 December 2012. pp. 243–. ISBN 978-94-011-4439-1. https://books.google.com/books?id=tsjrCAAAQBAJ&pg=PA243.
- ↑ 16.0 16.1 16.2 16.3 16.4 16.5 "Quinestrol". Drugs.com. https://www.drugs.com/international/quinestrol.html.
- ↑ "悦可婷 左炔诺孕酮炔雌醚片 6片/盒" (in Chinese). https://detail.yao.95095.com/item.htm?id=528371420852. "When taking the medicine for the first time, take the medicine once after lunch on the fifth day counting from the day of menstrual cramps, and take the second medicine at an interval of 20 days. Afterwards, take the second medicine taking day as the monthly medicine taking date, and take one tablet every month. When changing from short-acting oral contraceptives to long-acting contraceptives, you can take one long-acting contraceptive the next day after taking 22 tablets, and then take one tablet every month on the same day you started taking long-acting contraceptives."
- ↑ "Combined effects of levonorgestrel and quinestrol on reproductive hormone levels and receptor expression in females of the Mongolian gerbil (Meriones unguiculatus)". Zoological Science 29 (1): 37–42. January 2012. doi:10.2108/zsj.29.37. PMID 22233494.
- ↑ "Effects of levonorgestrel-quinestrol (EP-1) treatment on Mongolian gerbil wild populations: a case study". Integrative Zoology 8 (3): 277–284. September 2013. doi:10.1111/1749-4877.12018. PMID 24020466.
 |
