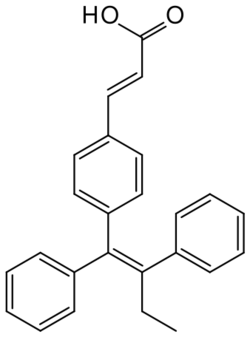
Chemistry:Etacstil
 | |
| Identifiers | |
|---|---|
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| ChEMBL | |
| Chemical and physical data | |
| Formula | C25H22O2 |
| Molar mass | 354.449 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Etacstil (developmental code names GW-5638, DPC974) is an orally active, nonsteroidal, combined selective estrogen receptor modulator (SERM) and selective estrogen receptor degrader (SERD) that was developed for the treatment of estrogen receptor-positive breast cancer.[1][2][3] It was shown to overcome antiestrogen (tamoxifen, aromatase inhibitor, fulvestrant) resistance in breast cancer by altering the shape of the estrogen receptor, thus exhibiting SERD properties.[4][5][6][7][8] Etacstil is a tamoxifen derivative and one of the first drugs to overcome tamoxifen-resistance. It is the predecessor of GW-7604,[3][9][10] of which etacstil is a prodrug (GW-7604 being the 4-hydroxy metabolite of etacstil).[11] This is analogous to the case of tamoxifen being a prodrug of afimoxifene (4-hydroxytamoxifen).[11]
Etacstil was developed in the early 1990s by Duke University, Glaxo Wellcome, and later, Dupont.[12][13] In 2001, Bristol Myers-Squibb (BMS) acquired Dupont, and for non-scientific, corporate reasons, closed the trial and abandoned the release of etacstil and its metabolite GW-7604.[6][9][12]
After many dormant years, a recent resurgence of interest in SERDs has led to the development of brilanestrant, a structural analogue of etacstil.[9]
See also
References
- ↑ Cancer Chemoprevention: Volume 2: Strategies for Cancer Chemoprevention. 2008-08-17. ISBN 9781592597680. https://books.google.com/books?id=ldyg4-cem9UC&pg=PA251.
- ↑ "GW 5638 Profile". AdisInsight. Springer Nature Switzerland AG. http://adisinsight.springer.com/drugs/800010109.
- ↑ 3.0 3.1 "GW 5638". Nuclear Receptor Signaling Atlas.. doi:10.1621/B4A9CIQ78V.
- ↑ Antiestrogen GW5638 induces a unique structural change in the ER. The biological significance of this conformational change was revealed in studies that demonstrated that tamoxifen-resistant breast tumor explants are not cross-resistant to GW5638. Because of these properties, this drug is currently being developed as a potential therapeutic for tamoxifen-resistant breast cancers."Circumventing tamoxifen resistance in breast cancers using antiestrogens that induce unique conformational changes in the estrogen receptor". Cancer Research 61 (7): 2917–2922. April 2001. PMID 11306468.
- ↑ "GW5638 uniquely alters the shape of the estrogen receptor". The Ben May Department for Cancer Research. The University of Chicago. 2015. http://benmay.uchicago.edu/page/news-archive-year-2005-GW5638.
- ↑ 6.0 6.1 "Tamoxifen-like drug suggests new ways to selectively block estrogen.". The University of Chicago Medical Center. 12 May 2005. http://www.uchospitals.edu/news/2005/20050512-gw5638.html.
- ↑ "Effects of a new clinically relevant antiestrogen (GW5638) related to tamoxifen on breast and endometrial cancer growth in vivo". Clinical Cancer Research 8 (6): 1995–2001. June 2002. PMID 12060645.
- ↑ "Quantitative comparison of the inhibitory effects of GW5638 and tamoxifen on angiogenesis in the cornea pocket assay". Angiogenesis 9 (2): 53–58. 2006. doi:10.1007/s10456-006-9029-x. PMID 16622786.
- ↑ 9.0 9.1 9.2 "Evaluation of the pharmacological activities of RAD1901, a selective estrogen receptor degrader". Endocrine-Related Cancer 22 (5): 713–724. October 2015. doi:10.1530/ERC-15-0287. PMID 26162914.
- ↑ "Molecular mechanism of action at estrogen receptor alpha of a new clinically relevant antiestrogen (GW7604) related to tamoxifen". Endocrinology 142 (2): 838–846. February 2001. doi:10.1210/endo.142.2.7932. PMID 11159857.
- ↑ 11.0 11.1 "Molecular mechanism of action at estrogen receptor alpha of a new clinically relevant antiestrogen (GW7604) related to tamoxifen". Endocrinology 142 (2): 838–846. February 2001. doi:10.1210/endo.142.2.7932. PMID 11159857.
- ↑ 12.0 12.1 "Osteoporosis Drug Bazedoxifene Stops Growth Of Breast Cancer Cells". Medical News Today. 17 June 2013. http://www.medicalnewstoday.com/articles/262039.php.
- ↑ "3-[4-(1,2-Diphenylbut-1-enyl)phenyl]acrylic acid: a non-steroidal estrogen with functional selectivity for bone over uterus in rats". Journal of Medicinal Chemistry 37 (11): 1550–1552. May 1994. doi:10.1021/jm00037a002. PMID 8201587.
 |
