Chemistry:Fenestrel
From HandWiki
Short description: Chemical compound
 | |
| Clinical data | |
|---|---|
| Other names | Demethoxycarbestrol; NSC-86465; 2-methyl-3-ethyl-4-phenyl-δ4-cyclohexenecarboxylic acid |
| ATC code |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| Chemical and physical data | |
| Formula | C16H20O2 |
| Molar mass | 244.334 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
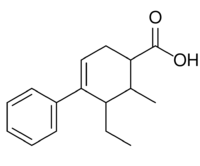
Fenestrel (INN, USAN) (developmental code name ORF-3858) is a synthetic, nonsteroidal estrogen that was developed as a postcoital contraceptive in the 1960s but was never marketed.[1][2][3][4] Synthesized by Ortho Pharmaceutical in 1961 and studied extensively, it was coined the "morning-after-pill" or "postcoital antifertility agent".[5] Fenestrel is a seco analogue of doisynolic acid, and a member of the cyclohexenecarboxylic acid series of estrogens.[6][7]
See also
References
- ↑ The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. Springer. 14 November 2014. pp. 517–. ISBN 978-1-4757-2085-3. https://books.google.com/books?id=0vXTBwAAQBAJ&pg=PA517.
- ↑ Drugs: Synonyms and Properties: Synonyms and Properties. Taylor & Francis. 8 May 2018. pp. 1407–. ISBN 978-1-351-78989-9. https://books.google.com/books?id=xUlaDwAAQBAJ&pg=PT1407.
- ↑ "[Critical study of new contraceptive methods]" (in fr). Schweizerische Medizinische Wochenschrift 101 (3): 92–96. January 1971. PMID 5544232.
- ↑ "Pioneering studies of the "morning-after" pill". The Yale Journal of Biology and Medicine 84 (2): 109–111. June 2011. PMID 21698041.
- ↑ "The Use of Pharmacological Agents to Study Implantation". Cellular and Molecular Aspects of Implantation. Springer Science & Business Media. 6 December 2012. pp. 478–. ISBN 978-1-4613-3180-3. https://books.google.com/books?id=iknTBwAAQBAJ&pg=PA478.
- ↑ Encyclopedia of chemical technology. Wiley. 1980. p. 672. ISBN 978-0-471-02065-3. https://books.google.com/books?id=yb9TAAAAMAAJ.
- ↑ Acta europaea fertilitatis. Piccin Medical Books.. 1969. https://books.google.com/books?id=KP-qAAAAIAAJ.
 |
