Chemistry:Orestrate
From HandWiki
Short description: Chemical compound
 | |
| Clinical data | |
|---|---|
| Other names | Estradiol 3-propionate 17β-(1-cyclohexenyl) ether; 17β-(Cyclohexen-1-yloxy)-estra-1,3,5(10)-trien-3-ol propionate |
| Drug class | Estrogen; Estrogen ester |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| Chemical and physical data | |
| Formula | C27H36O3 |
| Molar mass | 408.582 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
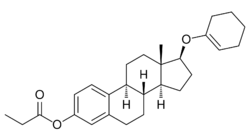
Orestrate (INN), also known as estradiol 3-propionate 17β-(1-cyclohexenyl) ether, is an estrogen medication and estrogen ester which was never marketed.[1][2][3][4][5] It is the C3 propionate ester and C17β-(1-cyclohexenyl) ether of estradiol.[1][5]
See also
References
- ↑ 1.0 1.1 The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. Springer. 14 November 2014. pp. 898, 905. ISBN 978-1-4757-2085-3. https://books.google.com/books?id=0vXTBwAAQBAJ&pg=PA905.
- ↑ "1,3,5(10)-Estratrien-17 -yl enol ethers and acetals. New classes of orally and parenterally active estrogenic derivatives". Journal of Medicinal Chemistry 16 (2): 123–127. February 1973. doi:10.1021/jm00260a009. PMID 4683106.
- ↑ "Effect of two orally active estradiol derivatives on sulfobromphthalein retention in rats". Pharmacological Research Communications 6 (2): 135–145. April 1974. doi:10.1016/s0031-6989(74)80021-4. PMID 4438394.
- ↑ "Orestratum". Chemikalien und Drogen Teil A: N-Q. Springer-Verlag. 12 March 2013. pp. 331–. ISBN 978-3-642-65035-2. https://books.google.com/books?id=4TWnBgAAQBAJ&pg=PA331.
- ↑ 5.0 5.1 Design of biopharmaceutical properties through prodrugs and analogs: a symposium. The Academy. 1977. p. 7. ISBN 978-0-917330-16-2. https://books.google.com/books?id=T7zwAAAAMAAJ.
 |
