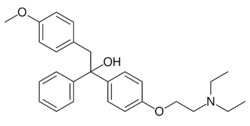
Chemistry:Ethamoxytriphetol
 | |
| Clinical data | |
|---|---|
| Other names | MER-25; NSC-19857 |
| Routes of administration | By mouth |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| Chemical and physical data | |
| Formula | C27H33NO3 |
| Molar mass | 419.565 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Ethamoxytriphetol (developmental code name MER-25) is a synthetic nonsteroidal antiestrogen that was studied clinically in the late 1950s and early 1960s but was never marketed.[1] MER-25 was first reported in 1958, and was the first antiestrogen to be discovered.[2][3][4] It has been described as "essentially devoid of estrogenic activity" and as having "very low estrogenic activity in all species tested".[1][2] However, some estrogenic effects in the uterus have been observed,[2] so it is not a pure antiestrogen (that is, a silent antagonist of the estrogen receptor (ER)) but is, instead, technically a selective estrogen receptor modulator (SERM).[5] For all intents and purposes, it is a nearly pure antiestrogen, however.[6]
MER-25 produces antifertility effects in animals, and garnered interest as a potential hormonal contraceptive.[3][4] However, clinical development was discontinued due to its low potency and the incidence of unacceptable central nervous system side effects,[3][4][7] including hallucinations and psychotic episodes, with higher doses.[8][9] Undesirable gastrointestinal side effects were also described.[7] Prior to being discontinued, the drug was also administered by Roy Hertz to three patients with metastatic breast cancer and was found to provide relief from bone pain, presumably due to dissolution of bone metastases.[10][8] This was the first such study of its kind of antiestrogen therapy for the treatment of breast cancer, and it led to the development of tamoxifen for this indication a decade later.[8] The drug was also evaluated for the purpose of ovulation induction and as a treatment of chronic mastitis and endometrial cancer before clinical development was stopped.[9]
MER-25, a simple triphenylethanol derivative,[6][4] is closely related structurally to the triphenylethylene (TPE) group of SERMs, which includes clomifene and tamoxifen.[2] The drug, a derivative of the cholesterol-lowering agent triparanol (MER-29) (which itself was derived from the estrogen chlorotrianisene (also known as TACE)),[9][11] was originally being studied in animals at Merrell Dow as a treatment for coronary artery disease.[4] Its antiestrogenic properties were discovered serendipitously when Leonard Lerner, a research endocrinologist at the company who was employed to study nonsteroidal estrogen pharmacology, noted the structural similarity of MER-25 to estrogenic TPE derivatives and decided to test the compound for estrogenicity.[4] Instead of the expected estrogenic effects however, Lerner found that MER-25 blocked the effects of estrogens.[4] Lerner subsequently went on to be involved in the discovery of clomifene, the first potently antiestrogenic TPE derivative to be characterized.[4] The structure of clomifene is similar to that of its predecessor, MER-25.[4][7] Clomifene is about 10-fold more potent as an antiestrogen than MER-25.[7]
The affinity of ethamoxytriphetol for the rat ER is approximately 0.06% relative to estradiol.[12][13] For comparison, the affinities of tamoxifen and afimoxifene (4-hydroxytamoxifen) for the rat ER relative to estradiol were 1% and 252%, respectively.[12][13]
See also
References
- ↑ 1.0 1.1 Analogue-based Drug Discovery III. John Wiley & Sons. 15 October 2012. pp. 5–. ISBN 978-3-527-65110-8. https://books.google.com/books?id=BP2Bo11gTOMC&pg=SA5-PA56.
- ↑ 2.0 2.1 2.2 2.3 "Discovery and Pharmacology of Nonsteroidal Estrogens and Antiestrogens". Tamoxifen: Pioneering Medicine in Breast Cancer. Springer Science & Business Media. 23 July 2013. pp. 7–. ISBN 978-3-0348-0664-0. https://books.google.com/books?id=p-W5BAAAQBAJ&pg=PA7.
- ↑ 3.0 3.1 3.2 "Structure-Activity Relationships of Nonsteroidal Estrogens and Antiestrogens". Estrogen/antiestrogen Action and Breast Cancer Therapy. Univ of Wisconsin Press. 1986. pp. 28,154. ISBN 978-0-299-10480-1. https://books.google.com/books?id=7WmLZfGXST0C&pg=PA28.
- ↑ 4.0 4.1 4.2 4.3 4.4 4.5 4.6 4.7 4.8 "A Century of Deciphering the Control Mechanisms of Estrogen Action in Breast Cancer: The Origins of Targeted Therapy and Chemoprevention". Estrogen Action, Selective Estrogen Receptor Modulators and Women's Health: Progress and Promise. World Scientific. 27 May 2013. pp. 7,112. ISBN 978-1-84816-959-3. https://books.google.com/books?id=ZM26CgAAQBAJ&pg=PA7.
- ↑ "The Aromatization Hypothesis 1970-1990". Brain Aromatase, Estrogens, and Behavior. OUP USA. 15 November 2012. pp. 161–. ISBN 978-0-19-984119-6. https://books.google.com/books?id=VPpMAgAAQBAJ&pg=PA161.
- ↑ 6.0 6.1 "Pure Antiestrogen". Hormone Therapy in Breast and Prostate Cancer. Springer Science & Business Media. 5 February 2010. pp. 4,161. ISBN 978-1-59259-152-7. https://books.google.com/books?id=dM0uvBnxiN0C&pg=PA161.
- ↑ 7.0 7.1 7.2 7.3 "Pharmacological Control of Reproduction in Women". Progress in Drug Research. Fortschritte der Arzneimittelforschung. Progres des Recherches Pharmaceutiques 12: 47–136. 1968. doi:10.1007/978-3-0348-7065-8_2. ISBN 978-3-0348-7067-2. PMID 4892528.
- ↑ 8.0 8.1 8.2 "Determinants of Drug Responsiveness". The Anticancer Drugs. Oxford University Press. 1994. pp. 21–. ISBN 978-0-19-506739-2. https://books.google.com/books?id=nPR1L4K5HuEC&pg=PA21.
- ↑ 9.0 9.1 9.2 "Pharmacology and Use of Antiestrogens in Treatment and Chemoprevention of Breast Cancer". Endocrinology of Breast Cancer. Springer Science & Business Media. 15 January 1999. pp. 286–287. ISBN 978-1-59259-699-7. https://books.google.com/books?id=7DSYBwAAQBAJ&pg=PA286.
- ↑ "Introduction". Estrogens, Estrogen Receptor, and Breast Cancer. IOS Press. 1 January 2000. pp. 13–. ISBN 978-0-9673355-4-4. https://books.google.com/books?id=v7ai5Mz9TZQC&pg=PA13.
- ↑ The Evolution of Drug Discovery: From Traditional Medicines to Modern Drugs. John Wiley & Sons. 11 January 2011. pp. 178–. ISBN 978-3-527-32669-3. https://books.google.com/books?id=iDNy0XxGqT8C&pg=PA178.
- ↑ 12.0 12.1 "The biological evaluation of novel antioestrogens for the treatment of breast cancer". Crit Rev Oncol Hematol 15 (3): 243–69. December 1993. doi:10.1016/1040-8428(93)90044-5. PMID 8142059.
- ↑ 13.0 13.1 "Endocrine Disruptors: Effects on Sex Steroid Hormone Receptors and Sex Development". Drug Toxicity in Embryonic Development II: Advances in Understanding Mechanisms of Birth Defects: Mechanistics Understanding of Human Development Toxicants. Springer Science & Business Media. 6 December 2012. pp. 437–. ISBN 978-3-642-60447-8. https://books.google.com/books?id=vcHsCAAAQBAJ&pg=PA437.
 |
