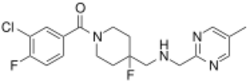
Chemistry:F-15599
 | |
| Clinical data | |
|---|---|
| Other names | F-15599; F15599; NLX-101; NLX101 |
| Routes of administration | Oral |
| Drug class | Serotonin 5-HT1A receptor agonist |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Metabolism | DB16936DB16936 |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| ChEMBL | |
| Chemical and physical data | |
| Formula | C19H22ClF2N4O |
| Molar mass | 395.86 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
F-15,599, also known as NLX-101, is a potent and selective 5-HT1A receptor full agonist.[1][2] In addition, it displays functional selectivity, or biased agonism, by preferentially activating postsynaptic serotonin 5-HT1A receptors over somatodendritic serotonin 5-HT1A autoreceptors.[3][1][2] The drug has been investigated for potential use as a pharmaceutical drug in the treatment of conditions including depression, schizophrenia, cognitive disorders, Rett syndrome, and fragile X syndrome.[4]
Pharmacology
Pharmacodynamics
In terms of its functional selectivity, the drug preferentially activates and phosphorylates ERK1/2 over receptor internalization or inhibition of adenylyl cyclase.[3] In addition, it preferentially activates the Gαi G protein subtype over the Gαo subtype.[3] As a result of its biased agonism for postsynaptic 5-HT1A receptors, F-15,599 shows regional selectivity in its central effects.[3] It mainly activates the prefrontal cortex, cingulate cortex, retrosplenial cortex, septum, and colliculi.[3] Conversely, the drug does not significantly alter cerebral blood flow in areas characterized by abundance of presynaptic serotonin 5-HT1A receptors, such as the raphe nucleus.[3][1][2]
F-15,599 has shown antidepressant-like, anxiolytic-like, antidyskinetic, procognitive, and antiaggressive effects in animals.[3][1][2][5] In cognitive tests in rodents, F-15,599 attenuates memory deficits elicited by the NMDA receptor antagonist phencyclidine (PCP), suggesting that it may improve cognitive function in disorders such as schizophrenia.[6] Another study found that F-15,599 reduces breathing irregularity and apneas observed in mice with mutations of the MeCP2 gene, a mouse model of Rett syndrome.[3][7]
History
F-15,599 was first described in the scientific literature by 2006.[8]
Clinical trials
F-15,599 was discovered and initially developed by Pierre Fabre Médicament, a French pharmaceuticals company. In September 2013, F-15,599 was out-licensed to Neurolixis, a California-based biotechnology company. Neurolixis announced that it intends to re-purpose F-15,599 for the treatment of Rett syndrome.[9] and obtained orphan drug designation from the United States Food and Drug Administration (FDA)[10] and from the European Commission for this indication.[11]
Researchers at the University of Bristol are investigating the activity of F-15599 in animal models of Rett Syndrome, with support from the International Rett Syndrome Foundation.[12] In June 2015, the Rett Syndrome Research Trust awarded a grant to Neurolixis to advance F-15599 to clinical development.[13]
As of September 2024, F-15,599 is in phase 1 clinical trials for fragile X syndrome.[4] Conversely, no recent development has been reported for depressive disorders or Rett syndrome and development has been discontinued for cognition disorders, mood disorders, and schizophrenia.[4]
See also
- Befiradol (F-13640; NLX-112)
- Eptapirone (F-11440)
- NLX-266
- TMU4142
References
- ↑ 1.0 1.1 1.2 1.3 "High-efficacy 5-HT1A agonists for antidepressant treatment: a renewed opportunity". Journal of Medicinal Chemistry 50 (20): 5024–33. October 2007. doi:10.1021/jm070714l. PMID 17803293.
- ↑ 2.0 2.1 2.2 2.3 "Signal transduction and functional selectivity of F15599, a preferential post-synaptic 5-HT1A receptor agonist". British Journal of Pharmacology 156 (2): 338–53. January 2009. doi:10.1111/j.1476-5381.2008.00001.x. PMID 19154445.
- ↑ 3.0 3.1 3.2 3.3 3.4 3.5 3.6 3.7 "Biased agonism in drug discovery: Is there a future for biased 5-HT1A receptor agonists in the treatment of neuropsychiatric diseases?". Pharmacol Ther 227. November 2021. doi:10.1016/j.pharmthera.2021.107872. PMID 33905796.
- ↑ 4.0 4.1 4.2 "NLX 101". 28 September 2024. https://adisinsight.springer.com/drugs/800041224.
- ↑ "F15599, a highly selective post-synaptic 5-HT(1A) receptor agonist: in-vivo profile in behavioural models of antidepressant and serotonergic activity". The International Journal of Neuropsychopharmacology 13 (10): 1285–1298. November 2010. doi:10.1017/S1461145709991222. PMID 20059805.
- ↑ "F15599, a preferential post-synaptic 5-HT1A receptor agonist: activity in models of cognition in comparison with reference 5-HT1A receptor agonists". European Neuropsychopharmacology 20 (9): 641–654. September 2010. doi:10.1016/j.euroneuro.2010.04.005. PMID 20488670.
- ↑ "A selective 5-HT1a receptor agonist improves respiration in a mouse model of Rett syndrome". Journal of Applied Physiology 115 (11): 1626–33. December 2013. doi:10.1152/japplphysiol.00889.2013. PMID 24092697.
- ↑ Assié, M. B., Cosi, C., Slot, L. B., Cussac, D., Martel, J. C., Depoortere, R., ... & Newman-Tancredi, A. (2006, October). Pharmacological profile of F15599, a highly selective serotonin 5-HT1A receptor agonist. In Society for Neuroscience 36th Annual Meeting, Atlanta, Georgia, USA (pp. 14–18). https://scholar.google.com/scholar?cluster=13780243967428028494
- ↑ http://neurolixis.com/images/stories/nlx_pf_license_23sept13.pdf
- ↑ "Enforcement Reports". http://www.accessdata.fda.gov/scripts/opdlisting/oopd/OOPD_Results_2.cfm?Index_Number=410613.
- ↑ "Public Health - European Commission". http://ec.europa.eu/health/documents/community-register/html/o1242.htm.
- ↑ "April: Rett syndrome research | News and features | University of Bristol". http://www.bristol.ac.uk/news/2014/april/rett-syndrome-research.html.
- ↑ "RSRT Awards $530,000 to Neurolixis for Clinical Development of NLX-101". 24 June 2015. https://rettsyndrome.wordpress.com/2015/06/24/rsrt-awards-530000-to-neurolixis-for-clinical-development-of-nlx-101/.
 |
