Chemistry:Volinanserin
Volinanserin (INN; developmental code MDL-100,907) is a highly selective 5-HT2A receptor antagonist that is frequently used in scientific research to investigate the function of the 5-HT2A receptor.[1][2][3][4][5] It was also tested in clinical trials as a potential antipsychotic,[6][7] antidepressant,[8] and treatment for insomnia but was never marketed.[1][9] The drug reached phase 3 trials for schizophrenia and insomnia prior to the discontinuation of its development in the late 2000s.[1] It is taken orally.[1]
Pharmacology
Pharmacokinetics
The time to peak levels of volinanserin is 1 to 2.5 hours.[10][11] The elimination half-life of volinanserin is 6.6 hours, with a range of 4.5 to 9.8 hours.[12][13][14][15][10][11] However, cortical serotonin 5-HT2A receptor occupancy with volinanserin measured by positron emission tomography (PET) imaging lasts much longer than its circulating elimination half-life would imply.[12][13][14][15][10]
Chemistry
Synthesis
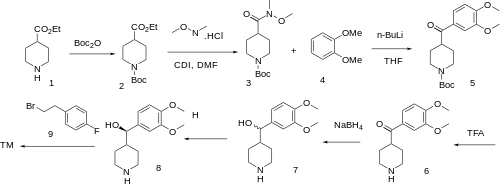
The synthesis of volinanserin has been reported.[16][17][18][19] Beginning with protection of ethyl isonipecotate (1) with Boc anhydride gives ethyl N-Boc-4-piperidinecarboxylate (2). Ester-amide interchange with N-methoxymethylamine HCl in the presence of carbonyldiimidazole (CDI) coupling agent gives 1-Boc-4-[methoxy(methyl)carbamoyl]piperidine (3). Weinreb ketone synthesis occurs upon benzoylation with 1,2-dimethoxybenzene (4) to give 1-Boc-4-(2,3-dimethoxybenzoyl)piperidine (5). Acid removal of the urethane protecting group gives (2,3-dimethoxyphenyl)-piperidin-4-ylmethanone (6). The reduction of the ketone with sodium borohydride leads to (2,3-dimethoxyphenyl)-piperidin-4-ylmethanol (7). Resolution of the alcohol gives (8). SN2 alkylation of the secondary nitrogen with 4-fluorophenethyl bromide (9) completes the synthesis of volinanserin (10).
See also
- Serotonin 5-HT2A receptor antagonist
- List of investigational insomnia drugs
- Eplivanserin/volinanserin
- Glemanserin
- Pruvanserin
- Roluperidone
- Lenperone
- Lidanserin
- Ketanserin
- Ritanserin
- Eplivanserin
- Pimavanserin
References
- ↑ 1.0 1.1 1.2 1.3 "Volinanserin". 13 March 2009. https://adisinsight.springer.com/drugs/800001847.
- ↑ "MDL 100,907: A Selective 5-HT 2A Receptor Antagonist for the Treatment of Schizophrenia". CNS Drug Reviews 3 (1): 49–67. March 1997. doi:10.1111/j.1527-3458.1997.tb00316.x.
- ↑ "5-HT2 receptors exert a state-dependent regulation of dopaminergic function: studies with MDL 100,907 and the amphetamine analogue, 3,4-methylenedioxymethamphetamine". European Journal of Pharmacology 223 (1): 65–74. November 1992. doi:10.1016/0014-2999(92)90819-P. PMID 1362159.
- ↑ "Synthesis and in vitro affinities of various MDL 100907 derivatives as potential 18F-radioligands for 5-HT2A receptor imaging with PET". Bioorganic & Medicinal Chemistry 17 (8): 2989–3002. April 2009. doi:10.1016/j.bmc.2009.03.021. PMID 19329329.
- ↑ "Blockade of the serotonin 5-HT2A receptor suppresses cue-evoked reinstatement of cocaine-seeking behavior in a rat self-administration model". Behavioral Neuroscience 123 (2): 382–96. April 2009. doi:10.1037/a0014592. PMID 19331461.
- ↑ "The role of positron emission tomography in the drug development of M100907, a putative antipsychotic with a novel mechanism of action". Journal of Clinical Pharmacology 39 (S1): 17S–24S. August 1999. doi:10.1002/j.1552-4604.1999.tb05933.x. PMID 10434243.
- ↑ Neurobiology of Mental Illness. OUP USA. July 2013. p. 767. ISBN 978-0-19-993495-9.
- ↑ "The selective 5-HT2A receptor antagonist M100907 enhances antidepressant-like behavioral effects of the SSRI fluoxetine". Neuropsychopharmacology 30 (12): 2205–15. December 2005. doi:10.1038/sj.npp.1300762. PMID 15886717.
- ↑ "5-HT(2A) inverse-agonists for the treatment of insomnia". Current Topics in Medicinal Chemistry 8 (11): 969–76. 2008. doi:10.2174/156802608784936700. PMID 18673166.
- ↑ 10.0 10.1 10.2 "Time course of 5-HT2A receptor occupancy in the human brain after a single oral dose of the putative antipsychotic drug MDL 100,907 measured by positron emission tomography". Neuropsychopharmacology 17 (3): 175–185. September 1997. doi:10.1016/S0893-133X(97)00044-4. PMID 9272484. "MDL 100,907 is rapidly absorbed following oral administration with the tmax occurring at 1–2.5 hours post dose. The plasma half-life is in the range of 4.5–9.8 hours with a mean half-life of 6.6 hours. Taking into account the half-life of 6.6 hours, it can be concluded that the receptor occupancy half-life of MDL 100,907 is considerably longer than the plasma half-life.".
- ↑ 11.0 11.1 Cite error: Invalid
<ref>tag; no text was provided for refs namedAndréeNybergIto1998 - ↑ 12.0 12.1 Cite error: Invalid
<ref>tag; no text was provided for refs namedTeegardenAlShammaXiong2008 - ↑ 13.0 13.1 Cite error: Invalid
<ref>tag; no text was provided for refs namedLandoltWehrle2009 - ↑ 14.0 14.1 Cite error: Invalid
<ref>tag; no text was provided for refs namedCaseyCuiBooth2022 - ↑ 15.0 15.1 Cite error: Invalid
<ref>tag; no text was provided for refs namedPielVernalekenRösch2014 - ↑ "Development of novel chiral capillary electrophoresis methods for the serotonin receptor (5-HT2A) antagonist MDL 100,907 (volinanserin) and for its key intermediate compound". Journal of Pharmaceutical and Biomedical Analysis 88: 579–583. January 2014. doi:10.1016/j.jpba.2013.10.017. PMID 24216279.
- ↑ Carr AA, Kane JM, Hay DA, "(+)-α-(2,3-Dimethoxyphenyl)-1-[2-(4-fluorophenyl)ethyl]-4-piperidinemethanol", WO patent 1991018602, published 12 December 1991
- ↑ "An efficient synthesis of the precursors of [11C]MDL 100907 labeled in two specific positions". Journal of Labelled Compounds and Radiopharmaceuticals 42 (10): 949–957. 1999. doi:10.1002/(SICI)1099-1344(199910)42:10<949::AID-JLCR253>3.0.CO;2-S.
- ↑ Blackburn TP, "Pharmaceutical composition containing a 5HT2c antagonist and a D2 antagonist", WO patent 1998004289, published 5 February 1998
 |
