Chemistry:Eplivanserin
Eplivanserin, also known by its former developmental code names SR-46349 and SR-46615 and by its former tentative brand names Ciltyri and Sliwens, is a serotonin 5-HT2A receptor antagonist which was under development by Sanofi Aventis for the treatment of a variety of medical conditions but was never marketed.[1][2] It is taken orally.[1]
Pharmacology
Pharmacodynamics
Eplivanserin is an inverse agonist on the serotonin receptor subtype 5-HT2A. In contrast to older sedating drugs acting on 5-HT2A receptors (e.g., mirtazapine, clozapine, risperidone), eplivanserin has practically no affinity to dopamine, histamine and adrenergic receptors.[3]
Eplivanserin blocks the head-twitch response produced by the serotonin precursor 5-hydroxytryptophan (5-HTP), by serotonergic psychedelics (serotonin 5-HT2A receptor agonists) like DOI, and by serotonin 5-HT1A receptor antagonists like WAY-100635 and (S)-UH-301.[4][5][6][7][8]
The drug has been shown to upregulate serotonin 5-HT2A receptor expression, unlike many known serotonin 5-HT2A receptor antagonists, which paradoxically induce receptor downregulation.[9][10] Relatedly, although eplivanserin given acutely can block the head-twitch response induced by various serotonergic drugs, it can also enhance the head-twitch response induced by 5-HTP and psychedelics like DOI when given on a sub-acute basis (given continuously and then withdrawn).[11][12]
Pharmacokinetics
Eplivanserin is well-absorbed, with an absorption of more than 70%.[13] The time to peak levels of eplivanserin is 2 to 6 hours.[13] Its elimination half-life is relatively long, with an average value of 50 hours.[13]
Chemistry
Synthesis
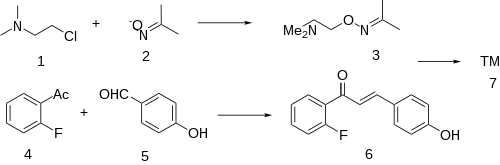
The condensation between 2'-Fluoroacetophenone [445-27-2] (5) & 4-hydroxybenzaldehyde [123-08-0] (6) give a chalcone intermediate (also an enone), i.e. CID:53982926 (7).
(2-chloroethyl)dimethylamine (CDMA) & acetone oxime are reacted together to give dimethylaminoacetoxime (DMA acetoxime), CID:16641114 (3).
Convergent synthesis gives the product as a mixture of isomers.
History
Sanofi Aventis announced in December 2009 that it was withdrawing its application for approval of eplivanserin from both the U.S. Food and Drug Administration and the European Medicines Agency.[15]
Research
Eplivanserin was under development for the treatment of anxiety disorders, insomnia, major depressive disorder, myocardial infarction (heart attack), and sleep apnea, but development for all indications was discontinued.[1] It reached preregistration for insomnia, but the New Drug Application (NDA) was declined by the Food and Drug Administration (FDA), which requested additional evidence regarding benefit–risk ratio.[1][16][17] Eplivanserin completed a clinical development program for insomnia that included two phase 3 clinical trials and almost 3,000 patients.[16] In a placebo controlled phase 2 trial with 351 subjects, eplivanserin reduced the sleep latency by 39 minutes (versus 26 minutes under placebo).[3]
See also
- Serotonin 5-HT2A receptor antagonist
- List of investigational insomnia drugs
- Eplivanserin/volinanserin
- Pimavanserin
- Volinanserin
References
- ↑ 1.0 1.1 1.2 1.3 "Eplivanserin". 5 November 2023. https://adisinsight.springer.com/drugs/800001948.
- ↑ "Future Treatments for Depression, Anxiety, Sleep Disorders, Psychosis, and ADHD". Neurotransmitter.net. http://www.neurotransmitter.net/newdrugs.html.
- ↑ 3.0 3.1 "5-HT(2A) inverse-agonists for the treatment of insomnia". Current Topics in Medicinal Chemistry 8 (11): 969–76. 2008. doi:10.2174/156802608784936700. PMID 18673166.
- ↑ "Biochemical and pharmacological properties of SR 46349B, a new potent and selective 5-hydroxytryptamine2 receptor antagonist". J Pharmacol Exp Ther 262 (2): 759–768. August 1992. doi:10.1016/S0022-3565(25)10821-5. PMID 1501121.
- ↑ "Enhancement of latent inhibition by two 5-HT2A receptor antagonists only when given at both pre-exposure and conditioning". Psychopharmacology (Berl) 169 (3–4): 321–331. September 2003. doi:10.1007/s00213-002-1173-4. PMID 14530903.
- ↑ "(1-(2,5-dimethoxy-4 iodophenyl)-2-aminopropane)-induced head-twitches in the rat are mediated by 5-hydroxytryptamine (5-HT) 2A receptors: modulation by novel 5-HT2A/2C antagonists, D1 antagonists and 5-HT1A agonists". J Pharmacol Exp Ther 273 (1): 101–112. April 1995. doi:10.1016/S0022-3565(25)09485-6. PMID 7714755.
- ↑ "The mechanism by which the selective 5-HT1A receptor antagonist S-(-) UH 301 produces head-twitches in mice". Pharmacol Biochem Behav 55 (1): 1–10. September 1996. doi:10.1016/0091-3057(96)00072-x. PMID 8870031.
- ↑ "The silent and selective 5-HT1A antagonist, WAY 100635, produces via an indirect mechanism, a 5-HT2A receptor-mediated behaviour in mice during the day but not at night. Short communication". J Neural Transm (Vienna) 105 (6–7): 635–643. 1998. doi:10.1007/s007020050085. PMID 9826108.
- ↑ "Repeated administration of SR 46349B, a selective 5-hydroxytryptamine2 antagonist, up-regulates 5-hydroxytryptamine2 receptors in mouse brain". Molecular Pharmacology 43 (1): 84–89. January 1993. doi:10.1016/S0026-895X(25)13451-2. PMID 8423772.
- ↑ "Antagonist functional selectivity: 5-HT2A serotonin receptor antagonists differentially regulate 5-HT2A receptor protein level in vivo". The Journal of Pharmacology and Experimental Therapeutics 339 (1): 99–105. October 2011. doi:10.1124/jpet.111.183780. PMID 21737536.
- ↑ "Repeated administration of SR 46349B, a selective 5-hydroxytryptamine2 antagonist, up-regulates 5-hydroxytryptamine2 receptors in mouse brain". Mol Pharmacol 43 (1): 84–89. January 1993. doi:10.1016/S0026-895X(25)13451-2. PMID 8423772.
- ↑ "Up-regulation of 5-HT2 receptors in the rat brain by repeated administration of SR 46349B, a selective 5-HT2 receptor antagonist". Eur J Pharmacol 246 (1): 73–80. June 1993. doi:10.1016/0922-4106(93)90012-x. PMID 8354344.
- ↑ 13.0 13.1 13.2 "Method of treating sleep disorders using eplivanserin". 10 November 2009. https://patents.google.com/patent/EP2365805A1/en. "Eplivanserin or pharmaceutically acceptable salts or esters thereof, in particular hemifumarate salt, is an antagonist of 5HT2A receptors (Journal of Pharmacological Experiment in Therapeutics, (1992), vol. 262 (2), pp. 759-68). Eplivanserin is well absorbed (>70%). Conventional dosage, between 1 and 10 mg, leads to a maximal plasma concentration that is reached between 2 and 6 hours; the half-life time of eplivanserin or pharmaceutically acceptable salts or esters thereof is relatively long, with an average value of 50 hours. Eplivanserin or pharmaceutically acceptable salts or esters thereof is also known to enhance slow wave sleep (SWS) (Neuropsychopharmacology (1999), vol.21 (3), pp. 455-466)."
- ↑ Eric Garcia, Christian Hoff, U.S. Patent 20,120,022,292 (2012 to Sanofi-Aventis).
- ↑ "Sanofi-Aventis Discontinues Eplivanserin For Insomnia". Dow Jones & Co. 21 December 2009. http://english.capital.gr/news.asp?id=876133.
- ↑ 16.0 16.1 "Drug Repurposing Patent Applications April-June 2017". Assay Drug Dev Technol 15 (8): 372–377. December 2017. doi:10.1089/adt.2017.29068.pq2. PMID 29235896.
- ↑ "Role of 5-HT2A receptor antagonists in the treatment of insomnia". Nat Sci Sleep 2: 139–150. 2010. doi:10.2147/nss.s6849. PMID 23616706. "Eplivanserin (Ciltyri®, SR 46349B; trans, 4-[(3Z) 3-(2- dimethylamino-ethyl) oxyimino-3 (2-fluorophenyl) propen-1 -yl] phenol hemifumarate), was previously in development by Sanofi-aventis which coined the term “ASTAR” (Antagonist of Serotonin Two A Receptors), in an effort to educate the public regarding this new mechanism of action for sleep aids. Also originally designated as a 5-HT2 receptor antagonist,31 eplivanserin is even more selective than ritanserin for 5-HT2A over 5-HT2C 14 (Table 1). Eplivanserin was reviewed by the FDA as a potential treatment for patients with chronic insomnia, but the FDA requested additional information regarding benefitrisk (sanofi-aventis press release, September 16, 2009) and development of the drug has been discontinued (sanofi-aventis press release, December 21, 2009).".
 |
