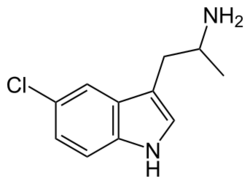
Chemistry:5-Chloro-αMT
 | |
 | |
| Clinical data | |
|---|---|
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| ChEMBL | |
| Chemical and physical data | |
| Formula | C11H13ClN2 |
| Molar mass | 208.69 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
5-Chloro-α-methyltryptamine (5-Chloro-αMT), also known as PAL-542, is a tryptamine derivative related to α-methyltryptamine (αMT) and one of only a few known specific serotonin-dopamine releasing agents (SDRAs).[1][2] It has been investigated in animals as a potential treatment for cocaine dependence.[2] The EC50 values of 5-chloro-αMT in evoking the in vitro release of serotonin (5-HT), dopamine (DA), and norepinephrine (NE) in rat synaptosomes were reported as 16 nM, 54 nM, and 3434 nM, with an NE/DA ratio of 63.6 and a DA/5-HT ratio of 3.38, indicating that it is a highly specific and well-balanced SDRA.[2] However, 5-chloro-αMT has also been found to act as a potent full agonist of the 5-HT2A receptor, with an EC50 value of 6.27 nM and an efficacy of 105%, and almost assuredly acts as a potent agonist of other serotonin receptors as well.[1]
5-Chloro-αMT was found to not reliably produce intracranial self-administration in rats or substitute for cocaine in rats or monkeys.[2] It was found through study of 5-chloro-αMT in rhesus monkeys that norepinephrine release has minimal influence on the abuse potential of monoamine releasing agents and that loss of norepinephrine release activity does not affect efficacy in reducing cocaine self-administration in SDRAs relative to serotonin-norepinephrine-dopamine releasing agents (SNDRAs) such as naphthylisopropylamine (PAL-287).[2] However, SDRAs like PAL-542 would, in theory, be expected to produce fewer side effects (including sympathomimetic/cardiovascular effects, insomnia, hyperthermia, and anxiety) relative to SNDRAs, and thus would likely be comparatively more favorable in the treatment of cocaine dependence and other conditions.[2]
A related agent, 5-fluoro-α-methyltryptamine (5-fluoro-αMT), also known as PAL-544, is a potent MAO-A inhibitor in addition to SNDRA and 5-HT2A receptor agonist.[1][2][3] Due to their close structural similarity, 5-chloro-αMT might also possess activity as an MAO-A inhibitor, a property which, in combination with its activity as monoamine releasing agent, could potentially render it dangerous in an analogous manner to the phenethylamine derivative para-methoxyamphetamine (PMA).[4] In addition, α-ethyltryptamine (αET), an SNDRA and close structural analogue of αMT and 5-chloro-αMT, like many other releasing agents of both 5-HT and DA such as MDMA, has been found to produce long-lasting serotonergic neurotoxicity in rats, and the same could also prove true for 5-chloro-αMT and other monoamine-releasing tryptamines.[5]
As a 5-HT2A receptor agonist, 5-chloro-αMT very likely possesses the capacity for psychedelic effects.[1][6] Indeed, its close analog 5-fluoro-αMT produces a strong head-twitch response in rats,[7] a property which is highly correlated with psychedelic effects in humans,[6][8] and αMT is well-established as a psychedelic drug in humans.[9]
Legality
5-Chloro-AMT is illegal in Singapore.[10]
See also
- 3-Chloromethamphetamine
- 4-Methyl-αMT
- 4-Hydroxy-αMT
- 4-Methyl-αET
- 5-Chloro-DMT
- 5-Cl-bk-MPA
- 5-EtO-AMT
- 5-MeO-αMT
- 5-MeO-αET
- 6-Fluoro-αMT
- 7-Methyl-αET
- 7-Chloro-AMT
- 5-API (PAL-571)
- α,N,N-TMT
- N-t-Butyltryptamine
- ST-1936
References
- ↑ 1.0 1.1 1.2 1.3 "Alpha-ethyltryptamines as dual dopamine-serotonin releasers". Bioorganic & Medicinal Chemistry Letters 24 (19): 4754–8. October 2014. doi:10.1016/j.bmcl.2014.07.062. PMID 25193229.
- ↑ 2.0 2.1 2.2 2.3 2.4 2.5 2.6 "Abuse-related effects of dual dopamine/serotonin releasers with varying potency to release norepinephrine in male rats and rhesus monkeys". Exp Clin Psychopharmacol 22 (3): 274–84. June 2014. doi:10.1037/a0036595. PMID 24796848. PMC 4067459. http://content.apa.org/journals/pha/22/3/274.
- ↑ "Selective inhibition of monoamine oxidase A and B by two substrate-analogues, 5-fluoro-alpha-methyltryptamine and p-chloro-beta-methylphenethylamine". Res. Commun. Chem. Pathol. Pharmacol. 54 (1): 125–8. October 1986. doi:10.1016/0028-3908(91)90057-i. PMID 3797802.
- ↑ "Differential behavioural and neurochemical effects of para-methoxyamphetamine and 3,4-methylenedioxymethamphetamine in the rat". Progress in Neuro-psychopharmacology & Biological Psychiatry 24 (6): 955–77. August 2000. doi:10.1016/S0278-5846(00)00113-5. PMID 11041537.
- ↑ "Reduction in brain serotonin markers by alpha-ethyltryptamine (Monase)". European Journal of Pharmacology 200 (1): 187–90. July 1991. doi:10.1016/0014-2999(91)90686-K. PMID 1722753.
- ↑ 6.0 6.1 "The role of central serotonergic mechanisms on head-twitch and backward locomotion induced by hallucinogenic drugs". Pharmacol. Biochem. Behav. 14 (1): 89–95. January 1981. doi:10.1016/0091-3057(81)90108-8. PMID 6258178.
- ↑ "Effect of gamma-mangostin through the inhibition of 5-hydroxy-tryptamine2A receptors in 5-fluoro-alpha-methyltryptamine-induced head-twitch responses of mice". Br. J. Pharmacol. 123 (5): 855–62. March 1998. doi:10.1038/sj.bjp.0701695. PMID 9535013.
- ↑ "A possible correlation between drug-induced hallucinations in man and a behavioural response in mice". Psychopharmacologia 11 (1): 65–78. 1967. doi:10.1007/bf00401509. PMID 5302272.
- ↑ "The hallucinogenic world of tryptamines: an updated review". Archives of Toxicology 89 (8): 1151–1173. August 2015. doi:10.1007/s00204-015-1513-x. PMID 25877327.
- ↑ "Misuse of Drugs Act - Singapore Statutes Online". https://sso.agc.gov.sg/Act/MDA1973.
| Phenylalkyl- amines (other than cathinones) |
|
|---|---|
| Cyclized phenyl- alkylamines | |
| Cathinones | |
| Tryptamines | |
| Chemical classes | |
| Adamantanes | |
|---|---|
| Adenosine antagonists | |
| Alkylamines | |
| Ampakines | |
| Arylcyclohexylamines | |
| Benzazepines | |
| Cholinergics |
|
| Convulsants | |
| Eugeroics | |
| Oxazolines | |
| Phenethylamines |
|
| Phenylmorpholines | |
| Piperazines | |
| Piperidines |
|
| Pyrrolidines | |
| Racetams | |
| Tropanes |
|
| Tryptamines | |
| Others |
|
| D1-like |
| ||||
|---|---|---|---|---|---|
| D2-like |
| ||||
| 5-HT1 |
| ||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 5-HT2 |
| ||||||||||||||||||||||||||||||||||||||
| 5-HT3–7 |
| ||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||
|
 | 0.00      (0 votes) (0 votes) |
