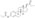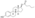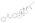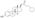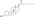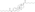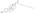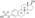Chemistry:Estrogen ester
An estrogen ester is an ester of an estrogen, most typically of estradiol but also of other estrogens such as estrone, estriol, and even nonsteroidal estrogens like diethylstilbestrol.[1][2][3] Esterification renders estradiol into a prodrug of estradiol with increased resistance to first-pass metabolism, slightly improving its oral bioavailability.[1][2][4] In addition, estrogen esters have increased lipophilicity, which results in a longer duration when given by intramuscular or subcutaneous injection due to the formation of a long-lasting local depot in muscle and fat.[1][2][3] Conversely, this is not the case with intravenous injection or oral administration.[1][5] Estrogen esters are rapidly hydrolyzed into their parent estrogen by esterases once they have been released from the depot.[1][2] Because estradiol esters are prodrugs of estradiol, they are considered to be natural and bioidentical forms of estrogen.[2][1][6]
Medical uses
Estrogen esters are used in hormone therapy, hormonal contraception, and high-dose estrogen therapy (e.g., for prostate cancer and breast cancer), among other indications.[1][2] The first estrogen ester to be marketed was estradiol benzoate in 1933, which was followed by many more.[7][8] One of the most widely used estradiol esters is estradiol valerate, which was first introduced in 1954.[9] Other major estradiol esters that are or have been used in medicine include estradiol acetate, estradiol cypionate, estradiol dipropionate, estradiol enantate, estradiol undecylate, and polyestradiol phosphate (an estrogen ester polymer), as well as the nitrogen mustard alkylating antineoplastic agent estramustine phosphate (estradiol normustine phosphate).[2][10]
The most common vehicles for injections of steroids and steroid esters are oil solutions, but aqueous solutions, aqueous suspensions, and emulsions have also been used.[11][additional citation(s) needed] The durations of estrogen esters are not prolonged if they are given orally, vaginally, or by intravenous injection.[11]
Pharmacology
Estrogen esters are essentially inactive themselves, with esters such as estradiol valerate and estradiol sulfate having about 2% of the affinity of estradiol for the estrogen receptor.[12] Likewise, the estrogen ether mestranol (ethinylestradiol 3-methyl ether) has about 1% of the affinity of estradiol for the estrogen receptor.[12] Estrone sulfate has less than 1% of the affinity of estradiol for the estrogen receptor.[13] As such, estrogen esters do not bind to the estrogen receptor except at extremely high concentrations.[14] The residual affinity of estrogen esters for the estrogen receptor in bioassays may actually be due to conversion into the parent estrogen, as attempts to prevent or limit this conversion have been found to abolish binding to the estrogen receptor and estrogenicity.[15][16][17]
| Ligand | Other names | Relative binding affinities (RBA, %)a | Absolute binding affinities (Ki, nM)a | Action | ||
|---|---|---|---|---|---|---|
| ERα | ERβ | ERα | ERβ | |||
| Estradiol | E2; 17β-Estradiol | 100 | 100 | Script error: The function "median" does not exist. (0.04–0.24) | Script error: The function "median" does not exist. (0.10–2.08) | Estrogen |
| Estrone | E1; 17-Ketoestradiol | Script error: The function "median" does not exist. (0.7–60) | Script error: The function "median" does not exist. (1.36–52) | Script error: The function "median" does not exist. (0.3–1.01) | Script error: The function "median" does not exist. (0.35–9.24) | Estrogen |
| Estriol | E3; 16α-OH-17β-E2 | Script error: The function "median" does not exist. (4.03–56) | Script error: The function "median" does not exist. (14.0–44.6) | Script error: The function "median" does not exist. (0.35–1.4) | Script error: The function "median" does not exist. (0.63–0.7) | Estrogen |
| Estetrol | E4; 15α,16α-Di-OH-17β-E2 | 4.0 | 3.0 | 4.9 | 19 | Estrogen |
| Alfatradiol | 17α-Estradiol | Script error: The function "median" does not exist. (7–80.1) | Script error: The function "median" does not exist. (2–42) | 0.2–0.52 | 0.43–1.2 | Metabolite |
| 16-Epiestriol | 16β-Hydroxy-17β-estradiol | Script error: The function "median" does not exist. (4.94–63) | 50 | ? | ? | Metabolite |
| 17-Epiestriol | 16α-Hydroxy-17α-estradiol | Script error: The function "median" does not exist. (29–103) | 79–80 | ? | ? | Metabolite |
| 16,17-Epiestriol | 16β-Hydroxy-17α-estradiol | 1.0 | 13 | ? | ? | Metabolite |
| 2-Hydroxyestradiol | 2-OH-E2 | Script error: The function "median" does not exist. (7–81) | 11–35 | 2.5 | 1.3 | Metabolite |
| 2-Methoxyestradiol | 2-MeO-E2 | 0.0027–2.0 | 1.0 | ? | ? | Metabolite |
| 4-Hydroxyestradiol | 4-OH-E2 | Script error: The function "median" does not exist. (8–70) | 7–56 | 1.0 | 1.9 | Metabolite |
| 4-Methoxyestradiol | 4-MeO-E2 | 2.0 | 1.0 | ? | ? | Metabolite |
| 2-Hydroxyestrone | 2-OH-E1 | 2.0–4.0 | 0.2–0.4 | ? | ? | Metabolite |
| 2-Methoxyestrone | 2-MeO-E1 | <0.001–<1 | <1 | ? | ? | Metabolite |
| 4-Hydroxyestrone | 4-OH-E1 | 1.0–2.0 | 1.0 | ? | ? | Metabolite |
| 4-Methoxyestrone | 4-MeO-E1 | <1 | <1 | ? | ? | Metabolite |
| 16α-Hydroxyestrone | 16α-OH-E1; 17-Ketoestriol | 2.0–6.5 | 35 | ? | ? | Metabolite |
| 2-Hydroxyestriol | 2-OH-E3 | 2.0 | 1.0 | ? | ? | Metabolite |
| 4-Methoxyestriol | 4-MeO-E3 | 1.0 | 1.0 | ? | ? | Metabolite |
| Estradiol sulfate | E2S; Estradiol 3-sulfate | <1 | <1 | ? | ? | Metabolite |
| Estradiol disulfate | Estradiol 3,17β-disulfate | 0.0004 | ? | ? | ? | Metabolite |
| Estradiol 3-glucuronide | E2-3G | 0.0079 | ? | ? | ? | Metabolite |
| Estradiol 17β-glucuronide | E2-17G | 0.0015 | ? | ? | ? | Metabolite |
| Estradiol 3-gluc. 17β-sulfate | E2-3G-17S | 0.0001 | ? | ? | ? | Metabolite |
| Estrone sulfate | E1S; Estrone 3-sulfate | <1 | <1 | >10 | >10 | Metabolite |
| Estradiol benzoate | EB; Estradiol 3-benzoate | 10 | ? | ? | ? | Estrogen |
| Estradiol 17β-benzoate | E2-17B | 11.3 | 32.6 | ? | ? | Estrogen |
| Estrone methyl ether | Estrone 3-methyl ether | 0.145 | ? | ? | ? | Estrogen |
| ent-Estradiol | 1-Estradiol | 1.31–12.34 | 9.44–80.07 | ? | ? | Estrogen |
| Equilin | 7-Dehydroestrone | Script error: The function "median" does not exist. (4.0–28.9) | 13.0–49 | 0.79 | 0.36 | Estrogen |
| Equilenin | 6,8-Didehydroestrone | 2.0–15 | 7.0–20 | 0.64 | 0.62 | Estrogen |
| 17β-Dihydroequilin | 7-Dehydro-17β-estradiol | 7.9–113 | 7.9–108 | 0.09 | 0.17 | Estrogen |
| 17α-Dihydroequilin | 7-Dehydro-17α-estradiol | Script error: The function "median" does not exist. (18–41) | 14–32 | 0.24 | 0.57 | Estrogen |
| 17β-Dihydroequilenin | 6,8-Didehydro-17β-estradiol | 35–68 | 90–100 | 0.15 | 0.20 | Estrogen |
| 17α-Dihydroequilenin | 6,8-Didehydro-17α-estradiol | 20 | 49 | 0.50 | 0.37 | Estrogen |
| Δ8-Estradiol | 8,9-Dehydro-17β-estradiol | 68 | 72 | 0.15 | 0.25 | Estrogen |
| Δ8-Estrone | 8,9-Dehydroestrone | 19 | 32 | 0.52 | 0.57 | Estrogen |
| Ethinylestradiol | EE; 17α-Ethynyl-17β-E2 | Script error: The function "median" does not exist. (68.8–480) | Script error: The function "median" does not exist. (2.0–144) | 0.02–0.05 | 0.29–0.81 | Estrogen |
| Mestranol | EE 3-methyl ether | ? | 2.5 | ? | ? | Estrogen |
| Moxestrol | RU-2858; 11β-Methoxy-EE | 35–43 | 5–20 | 0.5 | 2.6 | Estrogen |
| Methylestradiol | 17α-Methyl-17β-estradiol | 70 | 44 | ? | ? | Estrogen |
| Diethylstilbestrol | DES; Stilbestrol | Script error: The function "median" does not exist. (89.1–468) | Script error: The function "median" does not exist. (61.2–295) | 0.04 | 0.05 | Estrogen |
| Hexestrol | Dihydrodiethylstilbestrol | Script error: The function "median" does not exist. (31–302) | 60–234 | 0.06 | 0.06 | Estrogen |
| Dienestrol | Dehydrostilbestrol | Script error: The function "median" does not exist. (20.4–223) | 56–404 | 0.05 | 0.03 | Estrogen |
| Benzestrol (B2) | – | 114 | ? | ? | ? | Estrogen |
| Chlorotrianisene | TACE | 1.74 | ? | 15.30 | ? | Estrogen |
| Triphenylethylene | TPE | 0.074 | ? | ? | ? | Estrogen |
| Triphenylbromoethylene | TPBE | 2.69 | ? | ? | ? | Estrogen |
| Tamoxifen | ICI-46,474 | Script error: The function "median" does not exist. (0.1–47) | Script error: The function "median" does not exist. (0.28–6) | 3.4–9.69 | 2.5 | SERM |
| Afimoxifene | 4-Hydroxytamoxifen; 4-OHT | Script error: The function "median" does not exist. (1.7–257) | Script error: The function "median" does not exist. (0.98–339) | Script error: The function "median" does not exist. (0.1–3.61) | 0.04–4.8 | SERM |
| Toremifene | 4-Chlorotamoxifen; 4-CT | ? | ? | 7.14–20.3 | 15.4 | SERM |
| Clomifene | MRL-41 | Script error: The function "median" does not exist. (19.2–37.2) | 12 | 0.9 | 1.2 | SERM |
| Cyclofenil | F-6066; Sexovid | 151–152 | 243 | ? | ? | SERM |
| Nafoxidine | U-11,000A | 30.9–44 | 16 | 0.3 | 0.8 | SERM |
| Raloxifene | – | Script error: The function "median" does not exist. (7.8–69) | Script error: The function "median" does not exist. (0.54–16) | 0.188–0.52 | 20.2 | SERM |
| Arzoxifene | LY-353,381 | ? | ? | 0.179 | ? | SERM |
| Lasofoxifene | CP-336,156 | 10.2–166 | 19.0 | 0.229 | ? | SERM |
| Ormeloxifene | Centchroman | ? | ? | 0.313 | ? | SERM |
| Levormeloxifene | 6720-CDRI; NNC-460,020 | 1.55 | 1.88 | ? | ? | SERM |
| Ospemifene | Deaminohydroxytoremifene | 2.63 | 1.22 | ? | ? | SERM |
| Bazedoxifene | – | ? | ? | 0.053 | ? | SERM |
| Etacstil | GW-5638 | 4.30 | 11.5 | ? | ? | SERM |
| ICI-164,384 | – | Script error: The function "median" does not exist. (3.70–97.7) | 166 | 0.2 | 0.08 | Antiestrogen |
| Fulvestrant | ICI-182,780 | Script error: The function "median" does not exist. (9.4–325) | Script error: The function "median" does not exist. (2.05–40.5) | 0.42 | 1.3 | Antiestrogen |
| Propylpyrazoletriol | PPT | Script error: The function "median" does not exist. (10.0–89.1) | 0.12 | 0.40 | 92.8 | ERα agonist |
| 16α-LE2 | 16α-Lactone-17β-estradiol | 14.6–57 | 0.089 | 0.27 | 131 | ERα agonist |
| 16α-Iodo-E2 | 16α-Iodo-17β-estradiol | 30.2 | 2.30 | ? | ? | ERα agonist |
| Methylpiperidinopyrazole | MPP | 11 | 0.05 | ? | ? | ERα antagonist |
| Diarylpropionitrile | DPN | 0.12–0.25 | 6.6–18 | 32.4 | 1.7 | ERβ agonist |
| 8β-VE2 | 8β-Vinyl-17β-estradiol | 0.35 | 22.0–83 | 12.9 | 0.50 | ERβ agonist |
| Prinaberel | ERB-041; WAY-202,041 | 0.27 | 67–72 | ? | ? | ERβ agonist |
| ERB-196 | WAY-202,196 | ? | 180 | ? | ? | ERβ agonist |
| Erteberel | SERBA-1; LY-500,307 | ? | ? | 2.68 | 0.19 | ERβ agonist |
| SERBA-2 | – | ? | ? | 14.5 | 1.54 | ERβ agonist |
| Coumestrol | – | Script error: The function "median" does not exist. (0.0117–94) | Script error: The function "median" does not exist. (0.41–185) | 0.14–80.0 | 0.07–27.0 | Xenoestrogen |
| Genistein | – | Script error: The function "median" does not exist. (0.0012–16) | Script error: The function "median" does not exist. (0.86–87) | 2.6–126 | 0.3–12.8 | Xenoestrogen |
| Equol | – | 0.2–0.287 | Script error: The function "median" does not exist. (0.10–2.85) | ? | ? | Xenoestrogen |
| Daidzein | – | Script error: The function "median" does not exist. (0.0018–9.3) | Script error: The function "median" does not exist. (0.04–17.1) | 2.0 | 85.3 | Xenoestrogen |
| Biochanin A | – | Script error: The function "median" does not exist. (0.022–0.15) | Script error: The function "median" does not exist. (0.010–1.2) | 174 | 8.9 | Xenoestrogen |
| Kaempferol | – | Script error: The function "median" does not exist. (0.029–0.10) | Script error: The function "median" does not exist. (0.002–3.00) | ? | ? | Xenoestrogen |
| Naringenin | – | Script error: The function "median" does not exist. (<0.001–0.01) | Script error: The function "median" does not exist. (0.11–0.33) | ? | ? | Xenoestrogen |
| 8-Prenylnaringenin | 8-PN | 4.4 | ? | ? | ? | Xenoestrogen |
| Quercetin | – | <0.001–0.01 | 0.002–0.040 | ? | ? | Xenoestrogen |
| Ipriflavone | – | <0.01 | <0.01 | ? | ? | Xenoestrogen |
| Miroestrol | – | 0.39 | ? | ? | ? | Xenoestrogen |
| Deoxymiroestrol | – | 2.0 | ? | ? | ? | Xenoestrogen |
| β-Sitosterol | – | <0.001–0.0875 | <0.001–0.016 | ? | ? | Xenoestrogen |
| Resveratrol | – | <0.001–0.0032 | ? | ? | ? | Xenoestrogen |
| α-Zearalenol | – | Script error: The function "median" does not exist. (13–52.5) | ? | ? | ? | Xenoestrogen |
| β-Zearalenol | – | Script error: The function "median" does not exist. (0.032–13) | ? | ? | ? | Xenoestrogen |
| Zeranol | α-Zearalanol | 48–111 | ? | ? | ? | Xenoestrogen |
| Taleranol | β-Zearalanol | Script error: The function "median" does not exist. (13–17.8) | 14 | 0.8 | 0.9 | Xenoestrogen |
| Zearalenone | ZEN | Script error: The function "median" does not exist. (2.04–28) | Script error: The function "median" does not exist. (2.43–31.5) | ? | ? | Xenoestrogen |
| Zearalanone | ZAN | 0.51 | ? | ? | ? | Xenoestrogen |
| Bisphenol A | BPA | Script error: The function "median" does not exist. (0.008–1.0) | Script error: The function "median" does not exist. (0.002–4.23) | 195 | 35 | Xenoestrogen |
| Endosulfan | EDS | <0.001–<0.01 | <0.01 | ? | ? | Xenoestrogen |
| Kepone | Chlordecone | 0.0069–0.2 | ? | ? | ? | Xenoestrogen |
| o,p'-DDT | – | 0.0073–0.4 | ? | ? | ? | Xenoestrogen |
| p,p'-DDT | – | Script error: The function "median" does not exist. | ? | ? | ? | Xenoestrogen |
| Methoxychlor | p,p'-Dimethoxy-DDT | Script error: The function "median" does not exist. (<0.001–0.02) | 0.01–0.13 | ? | ? | Xenoestrogen |
| HPTE | Hydroxychlor; p,p'-OH-DDT | 1.2–1.7 | ? | ? | ? | Xenoestrogen |
| Testosterone | T; 4-Androstenolone | <0.0001–<0.01 | <0.002–0.040 | >5000 | >5000 | Androgen |
| Dihydrotestosterone | DHT; 5α-Androstanolone | Script error: The function "median" does not exist. (<0.001–0.05) | 0.0059–0.17 | 221–>5000 | 73–1688 | Androgen |
| Nandrolone | 19-Nortestosterone; 19-NT | 0.01 | 0.23 | 765 | 53 | Androgen |
| Dehydroepiandrosterone | DHEA; Prasterone | Script error: The function "median" does not exist. (<0.001–0.04) | 0.019–0.07 | 245–1053 | 163–515 | Androgen |
| 5-Androstenediol | A5; Androstenediol | 6 | 17 | 3.6 | 0.9 | Androgen |
| 4-Androstenediol | – | 0.5 | 0.6 | 23 | 19 | Androgen |
| 4-Androstenedione | A4; Androstenedione | <0.01 | <0.01 | >10000 | >10000 | Androgen |
| 3α-Androstanediol | 3α-Adiol | 0.07 | 0.3 | 260 | 48 | Androgen |
| 3β-Androstanediol | 3β-Adiol | 3 | 7 | 6 | 2 | Androgen |
| Androstanedione | 5α-Androstanedione | <0.01 | <0.01 | >10000 | >10000 | Androgen |
| Etiocholanedione | 5β-Androstanedione | <0.01 | <0.01 | >10000 | >10000 | Androgen |
| Methyltestosterone | 17α-Methyltestosterone | <0.0001 | ? | ? | ? | Androgen |
| Ethinyl-3α-androstanediol | 17α-Ethynyl-3α-adiol | 4.0 | <0.07 | ? | ? | Estrogen |
| Ethinyl-3β-androstanediol | 17α-Ethynyl-3β-adiol | 50 | 5.6 | ? | ? | Estrogen |
| Progesterone | P4; 4-Pregnenedione | <0.001–0.6 | <0.001–0.010 | ? | ? | Progestogen |
| Norethisterone | NET; 17α-Ethynyl-19-NT | Script error: The function "median" does not exist. (0.0015–<0.1) | Script error: The function "median" does not exist. (0.01–0.3) | 152 | 1084 | Progestogen |
| Norethynodrel | 5(10)-Norethisterone | Script error: The function "median" does not exist. (0.3–0.7) | <0.1–0.22 | 14 | 53 | Progestogen |
| Tibolone | 7α-Methylnorethynodrel | Script error: The function "median" does not exist. (0.45–2.0) | 0.2–0.076 | ? | ? | Progestogen |
| Δ4-Tibolone | 7α-Methylnorethisterone | 0.069–<0.1 | 0.027–<0.1 | ? | ? | Progestogen |
| 3α-Hydroxytibolone | – | Script error: The function "median" does not exist. (1.06–5.0) | 0.6–0.8 | ? | ? | Progestogen |
| 3β-Hydroxytibolone | – | Script error: The function "median" does not exist. (0.75–1.9) | 0.070–0.1 | ? | ? | Progestogen |
| Footnotes: a = (1) Binding affinity values are of the format "median (range)" (# (#–#)), "range" (#–#), or "value" (#) depending on the values available. The full sets of values within the ranges can be found in the Wiki code. (2) Binding affinities were determined via displacement studies in a variety of in-vitro systems with labeled estradiol and human ERα and ERβ proteins (except the ERβ values from Kuiper et al. (1997), which are rat ERβ). Sources: See template page. | ||||||
Template:Affinities and estrogenic potencies of estrogen esters and ethers at the estrogen receptors
In general, the longer the fatty acid ester chain of an estrogen ester, the greater its lipophilicity, and the longer the duration of the estrogen ester with intramuscular injection.[1][10] It has been said that, via intramuscular injection, the duration of estradiol benzoate (with an ester of length 1 carbon plus a benzene ring) is 2 to 3 days, of estradiol dipropionate (with two esters each of length 2 carbons) is 1 to 2 weeks, of estradiol valerate (ester of 5 carbons) is 1 to 3 weeks, and of estradiol cypionate (ester of 3 carbons plus a cyclopentane ring) is 3 to 4 weeks.[18] Estradiol enantate (ester of 7 carbons) has a duration of around 20 days.[2][19][20] Likewise, estradiol undecylate (ester of 10 carbons) has a very extended duration, which is longer than that of all of the aforementioned esters.[10][21][22]
| Estrogen | Dose | Peak levels | Time to peak | Duration |
|---|---|---|---|---|
| Estradiol benzoate | 5 mg | E2: 940 pg/mL E1: 343 pg/mL |
E2: 1.8 days E1: 2.4 days |
4–5 days |
| Estradiol valerate | 5 mg | E2: 667 pg/mL E1: 324 pg/mL |
E2: 2.2 days E1: 2.7 days |
7–8 days |
| Estradiol cypionate | 5 mg | E2: 338 pg/mL E1: 145 pg/mL |
E2: 3.9 days E1: 5.1 days |
11 days |
| Notes: All via i.m. injection of oil solution. Determinations via radioimmunoassay with chromatographic separation. Sources: See template. | ||||
Template:Parenteral potencies and durations of estrogens
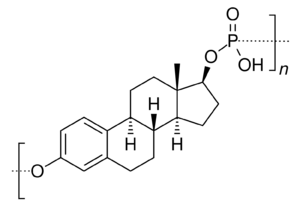
Polyestradiol phosphate is an atypical estradiol ester.[23][24] It is a phosphoric acid ester of estradiol in the form of a polymer, with an average polymer chain length of approximately 13 repeat units of estradiol phosphate.[23] It is slowly cleaved into estradiol and phosphoric acid by phosphatases.[23] Compared to conventional estradiol esters, polyestradiol phosphate has an extremely long duration; its elimination half-life is approximately 70 days.[24] Whereas conventional estradiol esters form a long-lasting depot in muscle and fat at the site of injection,[1] this is not the case with polyestradiol phosphate.[25] Instead, polyestradiol phosphate is taken up rapidly into the bloodstream following injection (by 90% within 24 hours), where it circulates, and is accumulated in the reticuloendothelial system.[25] Unlike other estradiol esters, polyestradiol phosphate is resistant to hydrolysis, which may be because it is a phosphatase inhibitor and may inhibit its own metabolism.[23]
Estrogen esters also occur naturally in the body, for instance estrogen conjugates like estrone sulfate and estrone glucuronide and the very long-lived lipoidal estradiol, which is constituted by ultra-long-chain esters like estradiol palmitate (ester of 16 carbons) and estradiol stearate (ester of 18 carbons).[1][2][26]
Chemistry
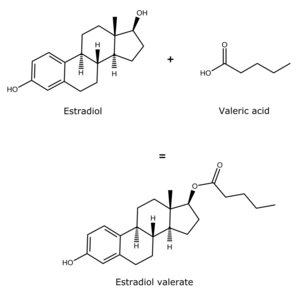
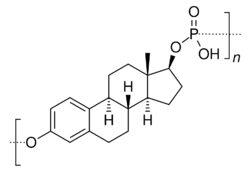
Estradiol esters have an ester moiety, usually a straight-chain fatty acid (e.g., valeric acid) or an aromatic fatty acid (e.g., benzoic acid), attached at the C3 and/or C17β positions of the steroid nucleus. These alkoxy moieties are substituted in place of the hydroxyl groups present in the unesterified estradiol molecule. Fatty acid esters serve to increase the lipophilicity of estradiol, increasing its solubility in fat. This causes them to form a depot with intramuscular or subcutaneous injection and gives them a long duration when administered by these routes.
Some estradiol esters have other moieties instead of fatty acids as the esters. Such esters include sulfuric acid (as in estradiol sulfate), sulfamic acid (as in estradiol sulfamate), phosphoric acid (as in estradiol phosphate), glucuronic acid (as in estradiol glucuronide, and others (e.g., estramustine phosphate (estradiol 3-normustine 17β-phosphate)). These esters are all hydrophilic, and have greater water solubility than estradiol or fatty acid estradiol esters. Unlike fatty acid estradiol esters, water-soluble estradiol esters can be administered by intravenous injection.
A few estrogen esters are polymers. These include polyestradiol phosphate and polyestriol phosphate, which are polymers of estradiol phosphate and estriol phosphate monomers, respectively. The monomers are connected in both cases by phosphate groups via the C3 and C17β positions. Polyestradiol phosphate has an average polymer chain length of approximately 13 repeat units of estradiol phosphate.[23] That is, each polyestradiol phosphate molecule is a polymer consisting on average of 13 estradiol phosphate molecules bonded together.[23] These polymeric estrogen esters are hydrophilic and water-soluble. Upon intramuscular injection, they do not form a depot and instead are rapidly absorbed into the circulation. However, they are only slowly cleaved into monomers, and as a result, have a very long duration in the body even outlasting that of many longer-chain fatty-acid estrogen esters.
| Estrogen | Structure | Ester(s) | Relative mol. weight |
Relative E2 contentb |
logPc | ||||
|---|---|---|---|---|---|---|---|---|---|
| Position(s) | Moiet(ies) | Type | Lengtha | ||||||
| Estradiol | – | – | – | – | 1.00 | 1.00 | 4.0 | ||
| Estradiol acetate | C3 | Ethanoic acid | Straight-chain fatty acid | 2 | 1.15 | 0.87 | 4.2 | ||
| Estradiol benzoate | C3 | Benzenecarboxylic acid | Aromatic fatty acid | – (~4–5) | 1.38 | 0.72 | 4.7 | ||
| Estradiol dipropionate | C3, C17β | Propanoic acid (×2) | Straight-chain fatty acid | 3 (×2) | 1.41 | 0.71 | 4.9 | ||
| Estradiol valerate | C17β | Pentanoic acid | Straight-chain fatty acid | 5 | 1.31 | 0.76 | 5.6–6.3 | ||
| Estradiol benzoate butyrate | C3, C17β | Benzoic acid, butyric acid | Mixed fatty acid | – (~6, 2) | 1.64 | 0.61 | 6.3 | ||
| Estradiol cypionate | C17β | Cyclopentylpropanoic acid | Aromatic fatty acid | – (~6) | 1.46 | 0.69 | 6.9 | ||
| Estradiol enanthate | C17β | Heptanoic acid | Straight-chain fatty acid | 7 | 1.41 | 0.71 | 6.7–7.3 | ||
| Estradiol dienanthate | C3, C17β | Heptanoic acid (×2) | Straight-chain fatty acid | 7 (×2) | 1.82 | 0.55 | 8.1–10.4 | ||
| Estradiol undecylate | C17β | Undecanoic acid | Straight-chain fatty acid | 11 | 1.62 | 0.62 | 9.2–9.8 | ||
| Estradiol stearate | C17β | Octadecanoic acid | Straight-chain fatty acid | 18 | 1.98 | 0.51 | 12.2–12.4 | ||
| Estradiol distearate | C3, C17β | Octadecanoic acid (×2) | Straight-chain fatty acid | 18 (×2) | 2.96 | 0.34 | 20.2 | ||
| Estradiol sulfate | C3 | Sulfuric acid | Water-soluble conjugate | – | 1.29 | 0.77 | 0.3–3.8 | ||
| Estradiol glucuronide | C17β | Glucuronic acid | Water-soluble conjugate | – | 1.65 | 0.61 | 2.1–2.7 | ||
| Estramustine phosphated | C3, C17β | Normustine, phosphoric acid | Water-soluble conjugate | – | 1.91 | 0.52 | 2.9–5.0 | ||
| Polyestradiol phosphatee | C3–C17β | Phosphoric acid | Water-soluble conjugate | – | 1.23f | 0.81f | 2.9g | ||
| Footnotes: a = Length of ester in carbon atoms for straight-chain fatty acids or approximate length of ester in carbon atoms for aromatic fatty acids. b = Relative estradiol content by weight (i.e., relative estrogenic potency). c = Experimental or predicted octanol/water partition coefficient (i.e., lipophilicity/[[hydrophobicity]]). Retrieved from PubChem, ChemSpider, and DrugBank. d = Also known as estradiol normustine phosphate. e = Polymer of estradiol phosphate (~13 repeat units). f = Relative molecular weight or estradiol content per repeat unit. g = logP of repeat unit (i.e., estradiol phosphate). Sources: See individual articles. | |||||||||
See also
- Pharmacokinetics of estradiol
- List of estrogen esters
- List of estrogens
- Steroid ester
- Progestogen ester
- Androgen ester
References
- ↑ 1.00 1.01 1.02 1.03 1.04 1.05 1.06 1.07 1.08 1.09 "Pharmacology of estrogens and progestogens: influence of different routes of administration". Climacteric 8 (Suppl 1): 3–63. 2005. doi:10.1080/13697130500148875. PMID 16112947. http://hormonebalance.org/images/documents/Kuhl%2005%20%20Pharm%20Estro%20Progest%20Climacteric_1313155660.pdf.
- ↑ 2.0 2.1 2.2 2.3 2.4 2.5 2.6 2.7 2.8 Michael Oettel; Ekkehard Schillinger (6 December 2012). Estrogens and Antiestrogens II: Pharmacology and Clinical Application of Estrogens and Antiestrogen. Springer Science & Business Media. pp. 235–237,261,271. ISBN 978-3-642-60107-1. https://books.google.com/books?id=wBvyCAAAQBAJ&pg=PA261. "Natural estrogens considered here include: [...] Esters of 17β-estradiol, such as estradiol valerate, estradiol benzoate and estradiol cypionate. Esterification aims at either better absorption after oral administration or a sustained release from the depot after intramuscular administration. During absorption, the esters are cleaved by endogenous esterases and the pharmacologically active 17β-estradiol is released; therefore, the esters are considered as natural estrogens."
- ↑ 3.0 3.1 R. S. Satoskar; S. D. Bhandarkar &nirmala N. Rege (1969). Pharmacology And Pharmacotherapeutics (New Revised 21 St Ed.). Popular Prakashan. p. 24. ISBN 978-81-7991-527-1. https://books.google.com/books?id=7d493VOD4P8C&pg=PA24. Retrieved 29 May 2012.
- ↑ Gordon L. Amidon; Ping I. Lee; Elizabeth M. Topp (2000). Transport Processes in Pharmaceutical Systems. CRC Press. pp. 188–189. ISBN 978-0-8247-6610-8. https://books.google.com/books?id=Rz3BM4C-tXoC&pg=PA188. Retrieved 29 May 2012.
- ↑ "Effective Absorption of Hormones". Br Med J 1 (4024): 371–3. February 1938. doi:10.1136/bmj.1.4024.371. PMID 20781252.
- ↑ "Pharmacokinetic and pharmacological features of oestradiol valerate". Maturitas 4 (4): 315–24. December 1982. doi:10.1016/0378-5122(82)90064-0. PMID 7169965.
- ↑ J. Elks (14 November 2014). The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. Springer. pp. 897–. ISBN 978-1-4757-2085-3. https://books.google.com/books?id=0vXTBwAAQBAJ&pg=PA897.
- ↑ Index Nominum 2000: International Drug Directory. Taylor & Francis US. 2000. pp. 404–406. ISBN 978-3-88763-075-1. https://books.google.com/books?id=5GpcTQD_L2oC&pg=PA406. Retrieved 13 September 2012.
- ↑ William Andrew Publishing (22 October 2013). Pharmaceutical Manufacturing Encyclopedia, 3rd Edition. Elsevier. pp. 1477–. ISBN 978-0-8155-1856-3. https://books.google.com/books?id=_J2ti4EkYpkC&pg=PA1477.
- ↑ 10.0 10.1 10.2 "A comparison of the pharmacokinetic properties of three estradiol esters". Contraception 21 (4): 415–24. April 1980. doi:10.1016/s0010-7824(80)80018-7. PMID 7389356.
- ↑ 11.0 11.1 C. W. Emmens (22 October 2013). Hormone Assay. Elsevier Science. pp. 394–395. ISBN 978-1-4832-7286-3. https://books.google.com/books?id=cCzgBAAAQBAJ&pg=PA394.
- ↑ 12.0 12.1 Gudermann, T. (2005). "Endokrinpharmakologie". Klinische Endokrinologie für Frauenärzte. pp. 187–220. doi:10.1007/3-540-26406-X_10. ISBN 3-540-44162-X.
- ↑ "Comparison of the ligand binding specificity and transcript tissue distribution of estrogen receptors alpha and beta". Endocrinology 138 (3): 863–70. March 1997. doi:10.1210/endo.138.3.4979. PMID 9048584.
- ↑ "Biological esterification of steroids". Endocr. Rev. 19 (3): 331–48. June 1998. doi:10.1210/edrv.19.3.0330. PMID 9626557.
- ↑ "The interaction of C-17 esters of estradiol with the estrogen receptor". Endocrinology 114 (4): 1180–6. April 1984. doi:10.1210/endo-114-4-1180. PMID 6705734.
- ↑ "Estrone sulfate and dehydroepiandrosterone sulfate: Transactivation of the estrogen and androgen receptor". Steroids 105: 50–8. January 2016. doi:10.1016/j.steroids.2015.11.009. PMID 26666359.
- ↑ Clark, Barbara J.; Prough, Russell A.; Klinge, Carolyn M. (2018). "Mechanisms of Action of Dehydroepiandrosterone". Dehydroepiandrosterone. Vitamins and Hormones. 108. pp. 29–73. doi:10.1016/bs.vh.2018.02.003. ISBN 9780128143612.
- ↑ H.J. Buchsbaum (6 December 2012). The Menopause. Springer Science & Business Media. pp. 62–. ISBN 978-1-4612-5525-3. https://books.google.com/books?id=z0LuBwAAQBAJ&pg=PA62.
- ↑ "Pharmacodynamic assessment of dihydroxyprogesterone acetophenide plus estradiol enanthate as a monthly injectable contraceptive". Contraception 33 (6): 579–89. June 1986. doi:10.1016/0010-7824(86)90046-6. PMID 3769482.
- ↑ "Pharmacokinetic studies of estradiol enantate in menopausic women". Arzneimittelforschung 36 (11): 1674–7. 1986. PMID 3814225.
- ↑ "Longacting steroid preparations". Acta Clin Belg 30 (1): 48–55. 1975. doi:10.1080/17843286.1975.11716973. PMID 1231448.
- ↑ R. S. Satoskar; S. D. Bhandarkar &nirmala N. Rege (1973). Pharmacology and Pharmacotherapeutics. Popular Prakashan. pp. 934–. ISBN 978-81-7991-527-1. https://books.google.com/books?id=7d493VOD4P8C&pg=PA934.
- ↑ 23.0 23.1 23.2 23.3 23.4 23.5 "Clinical pharmacology of polyestradiol phosphate". Prostate 13 (4): 299–304. 1988. doi:10.1002/pros.2990130405. PMID 3217277.
- ↑ 24.0 24.1 "Pharmacokinetics and testosterone suppression of a single dose of polyestradiol phosphate (Estradurin) in prostatic cancer patients". Prostate 28 (5): 307–10. 1996. doi:10.1002/(SICI)1097-0045(199605)28:5<307::AID-PROS6>3.0.CO;2-8. PMID 8610057.
- ↑ 25.0 25.1 Dinnendahl, V; Fricke, U, eds (2010) (in de). Arzneistoff-Profile. 4 (23 ed.). Eschborn, Germany: Govi Pharmazeutischer Verlag. ISBN 978-3-7741-98-46-3.
- ↑ "Estradiol-fatty acid esters. Endogenous long-lived estrogens". Ann. N. Y. Acad. Sci. 595 (1): 74–92. 1990. doi:10.1111/j.1749-6632.1990.tb34284.x. PMID 2197972. Bibcode: 1990NYASA.595...74H.
- ↑ Shellenberger, T. E. (1986). "Pharmacology of estrogens". The Climacteric in Perspective. pp. 393–410. doi:10.1007/978-94-009-4145-8_36. ISBN 978-94-010-8339-3.
Further reading
- "Longacting steroid preparations". Acta Clin Belg 30 (1): 48–55. 1975. doi:10.1080/17843286.1975.11716973. PMID 1231448.
- "A comparison of the pharmacokinetic properties of three estradiol esters". Contraception 21 (4): 415–24. 1980. doi:10.1016/s0010-7824(80)80018-7. PMID 7389356.
- "Pharmacokinetic and pharmacological features of oestradiol valerate". Maturitas 4 (4): 315–24. 1982. doi:10.1016/0378-5122(82)90064-0. PMID 7169965.
- "Pharmacodynamic effects of once-a-month combined injectable contraceptives". Contraception 49 (4): 361–85. 1994. doi:10.1016/0010-7824(94)90033-7. PMID 8013220.
 |