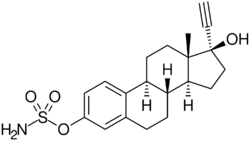
Chemistry:Ethinylestradiol sulfamate
 | |
| Clinical data | |
|---|---|
| Other names | 17α-Ethynylestradiol 3-O-sulfamate; J1028; EEMATE; EE2MATE; 17α-Ethynylestra-1,3,5(10)-triene-3,17β-diol 3-sulfamate |
| Routes of administration | By mouth[1] |
| Drug class | Estrogen; Estrogen ester |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| UNII | |
| Chemical and physical data | |
| Formula | C20H25NO4S |
| Molar mass | 375.48 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Ethinylestradiol sulfamate (developmental code name J1028), or 17α-ethynylestradiol 3-O-sulfamate, is a synthetic estrogen and estrogen ester which was never marketed.[1][2][3] It is the C3 sulfamate ester of ethinylestradiol.[1] The drug shows considerably improved oral estrogenic potency (uterotrophic) relative to ethinylestradiol in rats but without an increase in hepatic estrogenic potency.[1][3] Related compounds like ethinylestradiol N,N-diethylsulfamate (J271) and ethinylestradiol pyrrolidinosulfonate (J272) have also been developed, and have similar properties in animals.[4] However, the closely related compound estradiol sulfamate (E2MATE) failed to show estrogenic activity in humans, which is due to the fact that it is additionally a highly potent inhibitor of steroid sulfatase and prevents its own bioactivation into estradiol.[5]
See also
References
- ↑ 1.0 1.1 1.2 1.3 "Sulfamates of various estrogens are prodrugs with increased systemic and reduced hepatic estrogenicity at oral application". The Journal of Steroid Biochemistry and Molecular Biology 55 (3–4): 395–403. December 1995. doi:10.1016/0960-0760(95)00214-6. PMID 8541236.
- ↑ Schwarz S, Elger W, Siemann HJ, Reddersen G, Schneider B, "Estra-1,3,5(10)-trien derivatives, processes for their preparation and pharmaceutical compositions containing these compounds", US patent 6080735, issued 27 June 2000, assigned to Sterix Ltd
- ↑ 3.0 3.1 "Prodrug Approaches to Enhancing the Oral Delivery of Poorly Permeable Drugs". Prodrugs: Challenges and Rewards. Springer Science & Business Media. 12 March 2007. p. 58. ISBN 978-0-387-49782-2. https://books.google.com/books?id=qkjHxX5TgHEC&pg=RA1-PA32.
- ↑ "Novel oestrogen sulfamates: a new approach to oral hormone therapy". Expert Opinion on Investigational Drugs 7 (4): 575–589. April 1998. doi:10.1517/13543784.7.4.575. PMID 15991994.
- ↑ "Estradiol prodrugs (EP) for efficient oral estrogen treatment and abolished effects on estrogen modulated liver functions". The Journal of Steroid Biochemistry and Molecular Biology 165 (Pt B): 305–311. January 2017. doi:10.1016/j.jsbmb.2016.07.008. PMID 27449818.
 |

