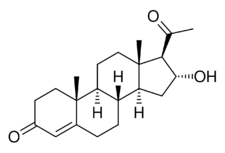
Chemistry:16α-Hydroxyprogesterone
| |
| Names | |
|---|---|
| IUPAC name
16α-Hydroxypregn-4-ene-3,20-dione
| |
| Systematic IUPAC name
(1R,2R,3aS,3bS,9aR,9bS,11aS)-1-Acetyl-2-hydroxy-9a,11a-dimethyl-1,2,3,3a,3b,4,5,8,9,9a,9b,10,11,11a-tetradecahydro-7H-cyclopenta[a]phenanthren-7-one | |
| Other names
16α-OHP; 16α-OH-PROG
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C21H30O3 | |
| Molar mass | 330.468 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
16α-Hydroxyprogesterone (16α-OHP), also known as 16α-hydroxypregn-4-ene-3,20-dione, is a minor endogenous progestogen steroid hormone and a metabolite of progesterone that is formed in lower amounts than 17α-hydroxyprogesterone (17α-OHP).[1] It occurs in micromolar concentrations and its physiological relevance hence is questionable.[1] However, it may accumulate in target tissues and could have a physiological role in the reproductive system and mammary gland development as well as the cardiovascular and central nervous systems.[1]
16α-OHP is formed from progesterone via 16α-hydroxylation primarily by CYP17A1 and primarily in steroidogenic tissues including the adrenal glands, testes, and ovaries.[1] It is also synthesized from progesterone during pregnancy by hepatic cytochrome P450 enzymes like CYP3A4 and CYP1A1 in the fetal liver as well as placenta.[1] It appears to be an end metabolite of progesterone and does not seem to be further metabolized.[1]
16α-OHP has approximately 67% and 43% of the affinity of progesterone for the PR-A and PR-B, respectively, and acts as an agonist of these receptors similarly to progesterone.[1] It was found to produce natriuresis similar to that produced by spironolactone when administered to humans, suggesting that it also has antimineralocorticoid activity similarly to progesterone.[1] However, surprisingly, 16α-OHP showed low affinity for the mineralocorticoid receptor (MR) of greater than 1 μM (compared to 1 nM for progesterone) and showed no antagonism of the MR at up to a concentration of 1 μM (whereas progesterone shows potent such activity).[1] However, the findings of another study suggested that 16α-OHP antagonizes the effects of aldosterone via the MR, and it may still be possible that 16α-OHP has significant antimineralocorticoid activity in some cells in spite of its weak MR affinity.[1]
See also
- 5α-Dihydroprogesterone
- 11-Deoxycorticosterone (21-hydroxyprogesterone)
- 20-Dihydroprogesterone
- Algestone (16α,17α-dihydroxyprogesterone)
References
 |
