Chemistry:Estriol (medication)
 | |
 | |
| Clinical data | |
|---|---|
| Pronunciation | /ˈɛstriɒl, -traɪɒl/[1] ESS-TREE-ohl[1] |
| Trade names | Ovestin, others[2][3] |
| Other names | Oestriol; E3; 16α-Hydroxyestradiol; Estra-1,3,5(10)-triene-3,16α,17β-triol |
| Routes of administration | By mouth, vaginal, intramuscular injection[4][5][6] |
| Drug class | Estrogen |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | Oral: ~1–2%[4][6] Vaginal: ~10–20%[5][4][6] |
| Protein binding | 92%:[4] • Albumin: 91%[4] • SHBG: 1%[4] • Free: 8%[4] |
| Metabolism | Liver, intestines (conjugation (glucuronidation, sulfation), oxidation, hydroxylation)[4] |
| Metabolites | • Estriol 16α-glucuronide[7][5] • Estriol 3-glucuronide[7][5] • Estriol 3-sulfate[7][5] • Estriol 3-sulfate 16α-gluc.[7][5] • 16α-Hydroxyestrone[4][8] • Others (minor)[4] |
| Excretion | Urine: >95% (as conjugates)[4][5] |
| Identifiers | |
| |
| CAS Number |
|
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| Chemical and physical data | |
| Formula | C18H24O3 |
| Molar mass | 288.387 g·mol−1 |
| 3D model (JSmol) | |
| Melting point | 82 to 86 °C (180 to 187 °F) (experimental) |
| Solubility in water | 0.119 mg/mL (20 °C) |
| |
| |
| (verify) | |
Estriol (E3), sold under the brand name Ovestin among others, is an estrogen medication and naturally occurring steroid hormone which is used in menopausal hormone therapy.[9][4][6][10] It is also used in veterinary medicine as Incurin to treat urinary incontinence due to estrogen deficiency in dogs.[11][12][13][14] The medication is taken by mouth in the form of tablets, as a cream that is applied to the skin, as a cream or pessary that is applied in the vagina, and by injection into muscle.[4][5][6]
Estriol is well-tolerated and produces relatively few adverse effects.[9][15] Side effects may include breast tenderness, vaginal discomfort and discharge, and endometrial hyperplasia.[9][15] Estriol is a naturally occurring and bioidentical estrogen, or an agonist of the estrogen receptor, the biological target of estrogens like endogenous estradiol.[4] It is an atypical and relatively weak estrogen, with much lower potency than estradiol.[4][6][16] When present continuously at adequate concentrations however, estriol produces full estrogenic effects similarly to estradiol.[17][18]
Estriol was first discovered in 1930,[19][20] and was introduced for medical use shortly thereafter.[21][22] Estriol esters such as estriol succinate are also used.[4][15][3] Although it is less commonly employed than other estrogens like estradiol and conjugated estrogens, estriol is widely available for medical use in Europe and elsewhere throughout the world.[4][2][3][6]
Medical uses
Estriol is used in menopausal hormone therapy to treat menopausal symptoms, such as hot flashes, vulvovaginal atrophy, and dyspareunia (difficult or painful sexual intercourse).[9][4][10][23][15] The benefits of estriol on bone mineral density and osteoporosis prevention have been inconsistent and are less clear.[15][9] Estriol has been found to reduce the risk of urinary tract infections and other urogenital symptoms.[4][9] A combination of estriol and lactobacilli as a dual estrogen and probiotic has been marketed for the treatment of vaginal atrophy and urinary tract infections.[24]
Available forms (except USA)
Estriol is available in oral tablet, vaginal cream, and vaginal suppository forms.[10] It is also available over-the-counter or from compounding pharmacies in the form of topical creams.[25] The medication is available both as estriol and in the form of estriol ester prodrugs such as estriol succinate, estriol acetate benzoate, and estriol tripropionate, as well as the polymeric ester prodrug polyestriol phosphate.[4][15][26][2][3]
Estriol was originally marketed in the 1930s in the form of oral capsules containing 0.06, 0.12, or 0.24 mg estriol under the brand names Theelol (Parke-Davis) and Estriol (Lilly, Abbott).[27][28][29][30][31] Subsequently, many decades later, oral tablets containing 0.35, 1, or 2 mg estriol were introduced under brand names such as Gynäsan, Hormomed, Ovestin, and Ovo-Vinces.[32]
Contraindications
General contraindications of estrogens include breast cancer, endometrial cancer, and others.[16] In animals, estriol is contraindicated during pregnancy and in ferrets.[14]
Side effects
Estriol is well-tolerated and produces relatively few adverse effects.[9][15] Breast tenderness may sometimes occur as a side effect of estriol.[9] Local reactions with vaginal estriol such as discomfort (irritation, burning, itching) and discharge may occur.[9] Estriol may produce endometrial hyperplasia similarly to estradiol and other estrogens, and hence should be combined with a progestogen in women with intact uteruses to prevent this risk.[33][4] However, it appears that typical clinical dosages of vaginal estriol are not associated with an important risk of endometrial proliferation or hyperplasia.[9][23] As such, combination with a progestogen may not be needed in the case of vaginal estriol.[9][23] Some studies suggest that this may also be true for oral estriol.[15] However, dosage and frequency of administration, as well as meal consumption, may be determining factors as to whether or not estriol produces endometrial proliferation.[4]
Overdose
Estrogens and other steroids are relatively safe in acute overdose.[citation needed] Estriol has been assessed in single oral doses of up to 75 mg.[34][35] General symptoms of estrogen overdose in humans may include nausea, vomiting, vaginal bleeding, and reversible feminization.[36][13] While there are no known studies describing the acute toxicity of estrogen overdose in dogs, this species is known to be more sensitive to the toxic effects of estrogens than humans and other animals.[13] The most serious short-term adverse effect of estrogens in dogs is bone marrow suppression and consequent pancytopenia, which can be life-threatening.[13]
Interactions
Interactions with estriol might be expected to be similar to those of estradiol.[37] No interactions with estriol have been reported in animals.[14] However, it should not be used in combination with other drugs that suppress bone marrow production in dogs.[14]
Pharmacology
Pharmacodynamics
Estriol is an estrogen, or an agonist of the estrogen receptors (ERs), ERα and ERβ.[4][38][39] In terms of relative binding affinities (RBA) for the ERs compared to estradiol, it was found in one study to possess 11 to 14% of the RBA for the human ERα and 18 to 21% of the RBA for the human ERβ.[39] Its relative transactivational capacities at the ERs compared to estradiol were 11% at ERα and 17% at ERβ.[39] In addition to being a ligand of the classical nuclear ERs, estriol is an antagonist of the G protein-coupled estrogen receptor (GPER), a membrane estrogen receptor (mER), at high concentrations (~1,000–10,000 μM).[40][41][38][42] This is in contrast to estradiol, which is an agonist of this receptor.[41][38][42] Like other estrogens, estriol does not importantly interact with other steroid hormone receptors.[43][44][45][46][47]
Estriol is a much less potent estrogen than is estradiol, and is somewhat weak and atypical in its properties.[4][39][41][16] Given by subcutaneous injection in mice, estradiol is about 10-fold more potent than estrone and about 100-fold more potent than estriol.[48] With clinical use, estriol is said to be weakly estrogenic in certain tissues, such as the liver and endometrium, but produces pronounced and full estrogenic responses in the vaginal epithelium.[4] The medication has been found to reduce hot flashes, improve vaginal atrophy, reverse the postmenopausal decline in skin thickness and collagen content, suppress gonadotropin secretion, and produce proliferation of breast epithelium.[4] Conversely, estriol does not consistently affect bone resorption or fracture risk, does not seem to increase breast density, and, at oral doses of 2 to 4 mg/day, does not affect liver proteins, lipid metabolism, or hemostatic parameters.[4][15] Additionally, vaginal estriol does not appear to produce endometrial proliferation or increase the risk of endometrial hyperplasia, and some studies have found this to be the case for oral estriol as well.[4][15][49] On the other hand, it appears that estriol may be able to stimulate the growth of active breast cancer.[15][9] In rodents, estriol induces mammary gland development similar to that with estrone.[50] By the oral route in women, estriol has approximately 30% of the potency of estradiol in terms of hot flashes relief and suppression of follicle-stimulating hormone secretion, and about 20% of the potency of estradiol on stimulation of liver production of high-density lipoprotein (HDL) cholesterol.[4] A study of ovulation inhibition by estrogens in women found that prevention of ovulation occurred with 5 mg/day oral estriol in only 1 of 7 cycles.[51][52] Due to its differing effects from those of estradiol, estriol may be considered a safer estrogen in certain regards.[9]

The weak and atypical estrogenicity of estriol is thought to be related to its short duration in the body and hence the fact that it stays bound to the ER for a relatively short amount of time.[4][18] Whereas estradiol remains bound to the ER for 6 to 24 hours with a single short-acting injection, estriol dissociates from the receptor much more rapidly and stays bound for only 1 to 6 hours.[4][18][55][56] As a result, estriol can only induce estrogenic effects which require short-term interaction with the ERs.[4][18] Induction of endometrial mitoses requires the ligand to remain bound for at least 9 to 12 hours, and this is thought to be responsible for the lack of endometrial proliferation with estriol in many studies.[4][18] If estriol is delivered more continuously than a single administration per day however, for instance if it is given as a subcutaneous pellet, as a depot injection, or in multiple doses two or three times per day, this results in more sustained exposure to estriol and full estrogenic responses equivalent to those of estradiol occur.[4][18][9] For these reasons, estriol has been described as a "short-acting" estrogen and it has been said that descriptors like "weak" and "impeded" are inaccurate.[18] Consumption of food after oral administration of estriol also results in more prolonged exposure to estriol, due to enterohepatic recycling and resurgences in estriol levels.[4] As such, if avoidance of endometrial hyperplasia or other full estrogenic effects is intended, it may be preferable to take estriol in a single dose, as low as possible, once per day at night before bedtime.[4][49]
Although estriol is an estrogen, it has also been reported to have mixed agonist–antagonist or partial agonist activity at the ERs.[4][18][16] On its own, it is said to be weakly estrogenic, but in the presence of estradiol, it has been found to be antiestrogenic.[4][41] However, this is again due to the fact that estriol is a "short-acting" estrogen.[18] If estriol is present continuously with estradiol, it shows no antagonism of estradiol.[18] The co-administration of estriol with estradiol has been found not to influence the effects of the latter in women, including neither enhancing nor antagonizing the effects of estradiol.[49][57]
| Estrogen | Form | Major brand names | EPD | CIC-D | Duration | |
|---|---|---|---|---|---|---|
| Estradiol | Oil solution | – | 40–60 mg | – | 1–2 mg ≈ 1–2 days | |
| Aqueous suspension | Aquadiol, Diogyn, Progynon, Mego-E | ? | 3.5 mg | 0.5–2 mg ≈ 2–7 days; 3.5 mg ≈ >5 days | ||
| Microspheres | Juvenum-E, Juvenum | ? | – | 1 mg ≈ 30 days | ||
| Estradiol benzoate | Oil solution | Progynon-B | 25–35 mg | – | 1.66 mg ≈ 2–3 days; 5 mg ≈ 3–6 days | |
| Aqueous suspension | Agofollin-Depot, Ovocyclin M | 20 mg | – | 10 mg ≈ 16–21 days | ||
| Emulsion | Menformon-Emulsion, Di-Pro-Emulsion | ? | – | 10 mg ≈ 14–21 days | ||
| Estradiol dipropionate | Oil solution | Agofollin, Di-Ovocylin, Progynon DP | 25–30 mg | – | 5 mg ≈ 5–8 days | |
| Estradiol valerate | Oil solution | Delestrogen, Progynon Depot, Mesigyna | 20–30 mg | 5 mg | 5 mg ≈ 7–8 days; 10 mg ≈ 10–14 days; 40 mg ≈ 14–21 days; 100 mg ≈ 21–28 days | |
| Estradiol benzoate butyrate | Oil solution | Redimen, Soluna, Unijab | ? | 10 mg | 10 mg ≈ 21 days | |
| Estradiol cypionate | Oil solution | Depo-Estradiol, Depofemin | 20–30 mg | – | 5 mg ≈ 11–14 days | |
| Aqueous suspension | Cyclofem, Lunelle | ? | 5 mg | 5 mg ≈ 14–24 days | ||
| Estradiol enanthate | Oil solution | Perlutal, Topasel, Yectames | ? | 5–10 mg | 10 mg ≈ 20–30 days | |
| Estradiol dienanthate | Oil solution | Climacteron, Lactimex, Lactostat | ? | – | 7.5 mg ≈ >40 days | |
| Estradiol undecylate | Oil solution | Delestrec, Progynon Depot 100 | ? | – | 10–20 mg ≈ 40–60 days; 25–50 mg ≈ 60–120 days | |
| Polyestradiol phosphate | Aqueous solution | Estradurin | 40–60 mg | – | 40 mg ≈ 30 days; 80 mg ≈ 60 days; 160 mg ≈ 120 days | |
| Estrone | Oil solution | Estrone, Kestrin, Theelin | ? | – | 1–2 mg ≈ 2–3 days | |
| Aqueous suspension | Estrone Aq. Susp., Kestrone, Theelin Aq. | ? | – | 0.1–2 mg ≈ 2–7 days | ||
| Estriol | Oil solution | – | ? | – | 1–2 mg ≈ 1–4 days | |
| Polyestriol phosphate | Aqueous solution | Gynäsan, Klimadurin, Triodurin | ? | – | 50 mg ≈ 30 days; 80 mg ≈ 60 days | |
| Notes: All aqueous suspensions are of microcrystalline particle size. Estradiol production during the menstrual cycle is 30–640 µg/day (6.4–8.6 mg total per month or cycle). The vaginal epithelium maturation dosage of estradiol benzoate or estradiol valerate has been reported as 5 to 7 mg/week. An effective ovulation-inhibiting dose of estradiol undecylate is 20–30 mg/month. Sources: See template. | ||||||
Pharmacokinetics

Absorption
Estriol has significant bioavailability, but its potency is limited by rapid metabolism and excretion and its relatively weak estrogenic activity.[6][15] With oral administration, during first-pass metabolism, a considerable portion of estriol is conjugated via sulfation into estriol sulfate and rapidly excreted.[6][4][49] Only about 10 to 20% of a dose of estriol remains in the circulation, and of this, only about 1 to 2% is present in its active, unconjugated form.[4][6][49] Peak levels of estriol occur about 1 to 3 hours after an oral dose.[4][5] Similarly to the case of progesterone, taking oral estriol with food greatly enhances its absorption.[6] In addition, due to enterohepatic recycling, consuming a meal 4 hours after taking oral estriol can produce a second peak in estriol levels.[4][5] Dosages of oral estriol of 4 to 10 mg have been found to result in a fairly large range of maximal estriol levels of 80 to 340 pg/mL.[5] After a single oral dose of 8 mg estriol in postmenopausal women, maximal levels of 65 pg/mL estriol and 60 ng/mL estriol conjugates were produced within an hour.[4] With continued daily administration, this increased to peak levels of 130 pg/mL estriol, whereas maximal levels of estriol conjugates remained at 60 ng/mL.[4] Levels of estriol rapidly decreased to low levels following occurrence of peak levels.[4] Consumption of a meal 4 hours after taking an oral dose of 8 mg estriol during continuous daily administration resulted in a second estriol peak 2 hours later of 120 pg/mL, with estriol levels declining slowly thereafter to about 25 pg/mL after 24 hours.[4]
The bioavailability of estriol is markedly increased with vaginal administration compared to oral administration.[6] The relative bioavailability of oral estriol was found to be about 10% of that of vaginal estriol.[5] In accordance, a single dose of 8 mg oral estriol and of 0.5 mg vaginal estriol have been found to produce similar circulating concentrations of estriol.[4] It has been said that 0.5 to 1 mg vaginal estriol may be equivalent in clinical effect to 8 to 12 mg oral estriol.[15] The high bioavailability of vaginal estriol is due to rapid absorption and low metabolism in atrophic vaginal mucosa.[4] Vaginal estriol at typical clinical dosages results both in high local concentrations of estriol in the vagina and in systemic action.[4] Vaginal administration of low doses of 30 µg estriol and of higher doses of 0.5 and 1 mg estriol have been found to produce equivalent local effects in the vagina and improvement of vaginal symptoms, suggesting that a saturation of estrogenic effect of vaginal estriol has been reached in the vagina by a dose of only 30 µg estriol.[4] In contrast to higher doses of vaginal estriol however, 30 µg/day estriol is not associated with systemic effects.[4] Similarly, the use of 0.5 mg vaginal estriol twice a week instead of daily has been said to largely attenuate the systemic effects of estriol.[4] Whereas oral estriol results in high levels of estriol conjugates which greatly exceed those of unconjugated estriol, vaginal estriol has been found to produce levels of unconjugated estriol and estriol conjugates that are similar.[4]
The absorption of estrogens by the skin is described as low for estriol, moderate for estradiol, and high for estrone.[4] This is related to the number of hydroxyl groups in the molecules; the more hydroxyl groups, the lower the skin permeability.[4] This may account for the relative lack of use of transdermal or topical estriol.[6]
Rectal administration of estriol has been assessed in one study.[58] Administration of a rectal suppository containing 100 mg estriol resulted in estriol levels in pregnant women at term increasing by about 53%.[58] Estriol levels at term are normally between 5,000 and 20,000 pg/mL, which suggests that estriol levels may have increased following the suppository by about 5,000 to 10,000 pg/mL (precise levels were not provided).[59][60][61]
Estriol succinate is an ester prodrug of estriol which is used medically via oral and vaginal routes similarly.[4] In estriol succinate, two of the hydroxyl groups of estriol, those at the C16α and C17β positions, are esterified with succinic acid.[4] As such, when adjusted for differences in molecular weight, a dose of 2 mg estriol succinate is equivalent to 1.18 mg unconjugated estriol.[4] Unlike other estrogen esters, such as estradiol valerate, estriol succinate is hydrolyzed almost not at all in the intestinal mucosa when taken orally, and in relation to this, is absorbed more slowly than is estriol.[4] Consequently, oral estriol succinate is a longer-acting form of estriol than oral estriol.[17] Instead of in the gastrointestinal tract, oral estriol succinate is cleaved into estriol mainly in the liver.[4] After a single 8 mg oral dose of estriol succinate, maximum levels of circulating estriol of 40 pg/mL are attained within 12 hours, and this increases up to 80 pg/mL with continued daily administration.[4]
Distribution
Similarly to estradiol, but unlike estrone, estriol is accumulated in target tissues.[4][62] The plasma protein binding of estriol is approximately 92%, with about 91% bound to albumin, 1% bound to sex hormone-binding globulin (SHBG), and 8% free or unbound.[4] Estriol has very low affinity for SHBG, with only about 0.3% of the affinity of testosterone for this protein (or about 0.6% of that of estradiol).[4][63][64] Relative to estradiol, which is about 98% plasma protein-bound, a significantly greater fraction of estriol is unbound in the circulation and hence available for biological activity (2% relative to 8%, respectively).[64][4][15] This appears to account for the greater than expected biological activity of estriol relative to estradiol when considering its affinities for the estrogen receptors.[65]
Metabolism
Estriol is metabolized extensively via conjugation, including glucuronidation and sulfation.[4][6][5][7] Glucuronidation of estriol takes place mainly in the intestinal mucosa, while sulfation occurs in the liver.[4] More minor amounts of estriol can be oxidized and hydroxylated at various positions.[4] One such reaction is transformation into 16α-hydroxyestrone.[4] Estriol is an end-product of phase I estrogen metabolism and cannot be converted into estradiol or estrone.[4][49] The main metabolites of estriol are estrogen conjugates, including estriol sulfates, estriol glucuronides, and mixed estriol sulfate/glucuronide conjugates.[4] 16α-Hydroxyestrone is known to occur as a metabolite of estriol as well.[66][67][62]
The biological half-life of oral estriol has been reported to be in the range of 5 to 10 hours.[68][8][49] The elimination half-life of estriol following an intramuscular injection of 1 mg estriol has been found to be 1.5 to 5.3 hours.[5] The blood half-life of unconjugated estriol has been reported to be 20 minutes.[69][70] The metabolic clearance rate of estriol is approximately 1,110 L/day/m2, which is about twice that of estradiol.[4] Hence, estriol is eliminated from the body more rapidly than is estradiol.[4] Enterohepatic recycling may extend the duration of oral estriol.[15]
A single 1 to 2 mg dose of estriol in oil solution by intramuscular injection has a duration of about 3 or 4 days.[71] Estriol esters such as estriol dipropionate and estriol dihexanoate, when administered via intramuscular injection in an oil solution, have been found to maintain elevated levels of estriol for much longer amounts of time than oral or vaginal estriol, in the range of days to months.[5] These two estriol esters have not been marketed, but estriol acetate benzoate and estriol tripropionate are medically used estriol esters which are given via depot intramuscular injection and are long-acting similarly.[26] Polyestriol phosphate is an ester of estriol in the form of a polymer, and has a very long duration of action.[72][48]
Excretion
Estriol is excreted more than 95% in urine.[4] This is due to the fact that estriol conjugates in the colon are completely hydrolyzed via bacterial enzymes and in turn estriol in this part of the body is reabsorbed into the body.[4] The main urinary metabolites of exogenous estriol administered via intravenous injection in baboons have been found to be estriol 16α-glucuronide (65.8%), estriol 3-glucuronide (14.2%), estriol 3-sulfate (13.4%), and estriol 3-sulfate 16α-glucuronide (5.1%).[5][7] The metabolism and excretion of estriol in these animals closely resembled that which has been observed in humans.[7]
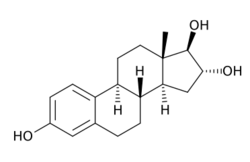
Chemistry
Estriol, also known as 16α-hydroxyestradiol or as estra-1,3,5(10)-triene-3,16α,17β-triol, is a naturally occurring estrane steroid with double bonds between the C1 and C2, C3 and C4, and C5 and C10 positions and hydroxyl groups at the C3, C16α, and C17β positions.[26][2] The name estriol and the abbreviation E3 were derived from the chemical terms estrin (estra-1,3,5(10)-triene) and triol (three hydroxyl groups).[73]
Analogues
A variety of analogues of estriol are known, including both naturally occurring isomers and synthetic substituted derivatives and esters.[26][2] 16β-Epiestriol (epiestriol), 17α-epiestriol, and 16β,17α-epiestriol are isomers of estriol that are endogenous weak estrogens.[26] Mytatrienediol (16α-methyl-16β-epiestriol 3-methyl ether) is a synthetic derivative of 16β-epiestriol that was never marketed.[26] Estriol acetate benzoate, estriol succinate, and estriol tripropionate are synthetic estriol esters that have been marketed for medical use, whereas estriol dihexanoate, estriol dipropionate, and estriol triacetate have not been introduced.[26][2] Quinestradol is the 3-cyclopentyl ether of estriol and has also been marketed.[26][2] Polyestriol phosphate, an ester of estriol in the form of a polymer, has been marketed previously as well.[72][74][48][75] These esters, ethers, and polymers are prodrugs of estriol.[4] Ethinylestriol and nilestriol are synthetic C17α ethynylated derivatives of estriol.[26][2] Ethinylestriol has not been marketed, but nilestriol, which is the 3-cyclopentyl ether of ethinylestriol and a prodrug of it, has been.[26][2]
Estetrol (E4), also known as 15α-hydroxyestriol, is a naturally occurring analogue of estriol with an additional hydroxyl group, at the C15α position.[76][77] It is closely related to estriol and has similar but non-identical pharmacological properties.[76][77] Like estriol, estetrol is a relatively weak and atypical estrogen.[76][77] Estetrol is under development for potential clinical use for a variety of indications, such as menopausal hormone therapy and hormonal birth control.[78][79]
History
Estriol was discovered in 1930.[19][20] Subsequently, it was introduced for medical use in oral and transdermal formulations under brand names such as Estriol, Oestrosalve, Theelol, and Tridestrin.[80][81][82][22][83][84][21] In addition, conjugated estriol, containing mainly estriol glucuronide, was marketed in the 1930s, under the brand names Emmenin and Progynon.[80][82][22][83][85][86] They were the first orally active estrogen preparations to be introduced in medicine.[85][86] In contrast to estrone, free estriol was never introduced for use by intramuscular injection.[87] Estriol continues to be used medically today, widely throughout the world and in a variety of different formulations and brand names.[2][3][6]
Society and culture
Generic names
Estriol is the generic name of estriol in American English and its INN, USP, BAN, DCF, and JAN.[26][2][88][3] It is pronounced /ˌɛstraɪoʊl/ ESS-TREE-ohl.[1] Estriolo is the name of estriol in Italian[3] and estriolum is its name in Latin, whereas its name remains unchanged as estriol in Spanish, Portuguese, French, and German.[3][2] Oestriol, in which the "O" is silent, was the former BAN of estriol and its name in British English,[26][88][2] but the spelling was eventually changed to estriol.[3]
Brand names
Estriol is or has been marketed under a variety of brand names throughout the world, including Aacifemine, Colpogyn, Elinol, Estriel, Estriol, Estriosalbe, Estrokad, Evalon, Gydrelle, Gynäsan, Gynest, Gynoflor (in combination with lactobacilli), Incurin (veterinary), Klimax-E, OeKolp, Oestro-Gynaedron, Orgestriol, Ortho-Gynest, Ovesterin, Ovestin, Ovestinon, Ovestrion, Ovo-Vinces, Pausanol, Physiogine, Sinapause, Synapause, Synapause-E, Trophicrème, Vago-Med, Vacidox, and Xapro.[2][3]
Estriol succinate has been marketed under the brand names Blissel, Evalon, Gelistrol, Hemostyptanon, Orgastyptin, Ovestin, Pausan, Sinapause, Styptanon, Synapsa, Synapasa, Synapausa, and Synapause.[26][2][3] Estriol diacetate benzoate has been marketed under the brand name Holin-Depot and estriol tripropionate has been marketed under the brand name Estriel.[26] Polyestriol phosphate has been marketed under the brand names Gynäsan, Klimadurin, and Triodurin.[74][89][90] Emmenin and Progynon were estriol products marketed in the 1930s which were manufactured from the urine of pregnant women and contained estriol conjugates, primarily estriol glucuronide.[85][86]
Estriol for multiple sclerosis had the tentative brand name Trimesta but did not complete development and was never marketed.[91]
Availability
Estriol is marketed widely throughout the world, including in Europe, South Africa , Australia , New Zealand, Asia, Latin America, and elsewhere.[2][3] The medication is also available in some countries in the form of estriol succinate, an ester prodrug of estriol.[2][26][92] Estriol and its esters are not approved for use in the United States or Canada , although estriol has been produced and sold by compounding pharmacies in North America for use as a component of bioidentical hormone therapy.[33][93] In addition, topical creams containing estriol are not regulated in the United States and are available over-the-counter in this country.[25]
Research
Estriol may have immunomodulatory effects and has been of investigational interest in the treatment of multiple sclerosis and a number of other conditions.[15] Estriol succinate was under development for the treatment of multiple sclerosis in the United States and worldwide, and reached phase II clinical trials for this indication, but development was discontinued due to insufficient effectiveness.[91] It had the tentative brand name Trimesta.[91]
Veterinary use
Estriol is used in veterinary medicine, under the brand name Incurin, in the treatment of urinary incontinence due to estrogen deficiency in dogs.[11][12][13][14] Certain estrogens, like estradiol, can cause bone marrow suppression in dogs, which can be fatal, but estriol appears to pose less or possibly no risk.[14][94]
References
- ↑ 1.0 1.1 1.2 Estriol. Dictionary.com.
- ↑ 2.00 2.01 2.02 2.03 2.04 2.05 2.06 2.07 2.08 2.09 2.10 2.11 2.12 2.13 2.14 2.15 2.16 Index Nominum 2000: International Drug Directory. Taylor & Francis. January 2000. pp. 407–. ISBN 978-3-88763-075-1. https://books.google.com/books?id=5GpcTQD_L2oC&pg=PA407.
- ↑ 3.00 3.01 3.02 3.03 3.04 3.05 3.06 3.07 3.08 3.09 3.10 3.11 "Estriol". https://www.drugs.com/international/estriol.html.
- ↑ 4.00 4.01 4.02 4.03 4.04 4.05 4.06 4.07 4.08 4.09 4.10 4.11 4.12 4.13 4.14 4.15 4.16 4.17 4.18 4.19 4.20 4.21 4.22 4.23 4.24 4.25 4.26 4.27 4.28 4.29 4.30 4.31 4.32 4.33 4.34 4.35 4.36 4.37 4.38 4.39 4.40 4.41 4.42 4.43 4.44 4.45 4.46 4.47 4.48 4.49 4.50 4.51 4.52 4.53 4.54 4.55 4.56 4.57 4.58 4.59 4.60 4.61 4.62 4.63 4.64 4.65 4.66 4.67 4.68 4.69 4.70 4.71 4.72 4.73 4.74 4.75 4.76 4.77 "Pharmacology of estrogens and progestogens: influence of different routes of administration". Climacteric 8 (Suppl 1): 3–63. 2005. doi:10.1080/13697130500148875. PMID 16112947. http://hormonebalance.org/images/documents/Kuhl%2005%20%20Pharm%20Estro%20Progest%20Climacteric_1313155660.pdf.
- ↑ 5.00 5.01 5.02 5.03 5.04 5.05 5.06 5.07 5.08 5.09 5.10 5.11 5.12 5.13 5.14 5.15 Estrogens and Antiestrogens II: Pharmacology and Clinical Application of Estrogens and Antiestrogen. Springer Science & Business Media. 6 December 2012. pp. 265, 273. ISBN 978-3-642-60107-1. https://books.google.com/books?id=wBvyCAAAQBAJ&pg=PA265.
- ↑ 6.00 6.01 6.02 6.03 6.04 6.05 6.06 6.07 6.08 6.09 6.10 6.11 6.12 6.13 6.14 "Unconventional estrogens: estriol, biest, and triest". Clin Obstet Gynecol 44 (4): 864–79. December 2001. doi:10.1097/00003081-200112000-00024. PMID 11600867.
- ↑ 7.0 7.1 7.2 7.3 7.4 7.5 7.6 "Estriol metabolism in the baboon: analysis of urinary and biliary metabolites". Steroids 22 (6): 795–817. December 1973. doi:10.1016/0039-128X(73)90054-8. PMID 4203562.
- ↑ 8.0 8.1 Cite error: Invalid
<ref>tag; no text was provided for refs namedDörwald2013 - ↑ 9.00 9.01 9.02 9.03 9.04 9.05 9.06 9.07 9.08 9.09 9.10 9.11 9.12 9.13 "The efficacy and safety of estriol to treat vulvovaginal atrophy in postmenopausal women: a systematic literature review". Climacteric 20 (4): 321–330. August 2017. doi:10.1080/13697137.2017.1329291. PMID 28622049.
- ↑ 10.0 10.1 10.2 Hormones in Obstetrics and Gynaecology. Jaypee Brothers Publishers. 1 January 2005. pp. 101–. ISBN 978-81-8061-427-9. https://books.google.com/books?id=IBxBbaDjXw0C&pg=PA101.
- ↑ 11.0 11.1 Textbook of Veterinary Internal Medicine - eBook. Elsevier Health Sciences. 11 January 2017. pp. 6017, 6380. ISBN 978-0-323-31239-4. https://books.google.com/books?id=57XBDQAAQBAJ&pg=PT6017.
- ↑ 12.0 12.1 Small Animal Clinical Pharmacology and Therapeutics - E-Book. Elsevier Health Sciences. 25 July 2011. pp. 2350–2351. ISBN 978-1-4377-2357-1. https://books.google.com/books?id=yDjDr_MLGSsC&pg=PT2350.
- ↑ 13.0 13.1 13.2 13.3 13.4 Kirk's Current Veterinary Therapy XIV - E-Book. Elsevier Health Sciences. 10 July 2008. pp. 772, 4442. ISBN 978-1-4377-1152-3. https://books.google.com/books?id=NaamlrzbTwoC&pg=PT4442.
- ↑ 14.0 14.1 14.2 14.3 14.4 14.5 Saunders Handbook of Veterinary Drugs: Small and Large Animal. Elsevier Health Sciences. 1 October 2015. pp. 304–. ISBN 978-0-323-24485-5. https://books.google.com/books?id=ip8_CwAAQBAJ&pg=PA304.
- ↑ 15.00 15.01 15.02 15.03 15.04 15.05 15.06 15.07 15.08 15.09 15.10 15.11 15.12 15.13 15.14 15.15 "Estriol: emerging clinical benefits". Menopause 24 (9): 1081–1085. September 2017. doi:10.1097/GME.0000000000000855. PMID 28375935.
- ↑ 16.0 16.1 16.2 16.3 "The maternal-fetal-placental unit.". Principles and Practice of Endocrinology and Metabolism. Lippincott Williams & Wilkins. 2001. pp. 932, 989, 1061. ISBN 978-0-7817-1750-2. https://books.google.com/books?id=FVfzRvaucq8C&pg=PA1061.
- ↑ 17.0 17.1 "The agonistic and antagonistic effects of short acting estrogens: a review". Pharmacol. Ther. 21 (3): 429–53. 1983. doi:10.1016/0163-7258(83)90063-3. PMID 6356176.
- ↑ 18.0 18.1 18.2 18.3 18.4 18.5 18.6 18.7 18.8 18.9 "The agonistic and antagonistic actions of estriol". J. Steroid Biochem. 20 (4B): 1005–13. April 1984. doi:10.1016/0022-4731(84)90011-6. PMID 6202959.
- ↑ 19.0 19.1 Gynecologic Endocrinology. Springer Science & Business Media. 11 November 2013. pp. 31–. ISBN 978-1-4613-2157-6. https://books.google.com/books?id=9vv2BwAAQBAJ&pg=PA31.
- ↑ 20.0 20.1 Antineoplastic and Immunosuppressive Agents. Springer Science & Business Media. 27 November 2013. pp. 104–. ISBN 978-3-642-65806-8. https://books.google.com/books?id=aU_oCAAAQBAJ&pg=PA104.
- ↑ 21.0 21.1 "Estriol: a review". Physiological Reviews 38 (3): 463–480. July 1958. doi:10.1152/physrev.1958.38.3.463. PMID 13567043.
- ↑ 22.0 22.1 22.2 "Estrogenic Hormones: Their Clinical Usage". California and Western Medicine 49 (5): 362–366. November 1938. PMID 18744783.
- ↑ 23.0 23.1 23.2 "Review of the endometrial safety during intravaginal treatment with estriol". Eur. J. Obstet. Gynecol. Reprod. Biol. 62 (1): 101–6. September 1995. doi:10.1016/0301-2115(95)02170-c. PMID 7493689. https://www.ejog.org/article/0301-2115(95)02170-C/pdf.
- ↑ "Estriol/Lactobacillus - Acerus Pharmaceuticals/Medinova -". AdisInsight. Springer Nature Switzerland AG. https://adisinsight.springer.com/drugs/800050964.
- ↑ 25.0 25.1 "Bioidentical hormone therapy: a review of the evidence". J Womens Health (Larchmt) 16 (5): 600–31. June 2007. doi:10.1089/jwh.2006.0311. PMID 17627398.
- ↑ 26.00 26.01 26.02 26.03 26.04 26.05 26.06 26.07 26.08 26.09 26.10 26.11 26.12 26.13 26.14 The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. Springer. 14 November 2014. pp. 899–. ISBN 978-1-4757-2085-3. https://books.google.com/books?id=0vXTBwAAQBAJ&pg=PA899.
- ↑ Diagnosis and treatment of menstrual disorders and sterility. Hoeber. 1946. p. 525. https://books.google.com/books?id=nNVLAAAAMAAJ. "8. PREPARATIONS OF ESTRIOL. Estriol is the least active of all commercially available natural estrogenic substances. A milligram of estriol yields approximately 350 Allen-Doisy rat units. Estriol (Abbott). Capsules containing 0.06, 0.12, and 0.24 mg. Estriol (Lilly). Puvules containing 0.06, 0.12, and 0.24 mg. Theelol (Parke-Davis). Capsules containing 0.06, 0.12, and 0.24 mg."
- ↑ Modern Medical Therapy in General Practice. William & Wilkins Company. 1940. p. 194. ISBN 9780598668332. https://books.google.com/books?id=V5HJymGfFP8C. "Estriol. Estriol (theelol) is trihydroxyestrin. It is a crystalline estrogenic steroid obtained from the urine of pregnant women. It is less actively estrogenic than estrone. Several pharmaceutical houses supply capsules containing 0.06 or 0.12 mg. These may be obtained as Theelol (Parke-Davis), Estriol (Abbott), and Estriol (Lilly)."
- ↑ A manual of pharmacology and its applications to therapeutics and toxicology. W. B. Saunders. 1948. https://books.google.com/books?id=LdtsAAAAMAAJ. "Estriol (Theelol), N.N.R.; characters and solubility as for estrone; considerably less potent. Marketed as capsules of 0.06, 0.12 and 0.24 mg. Dose, 0.06 to 0.12 mg. once to four times daily."
- ↑ Council on Drugs (American Medical Association) (1950). New and Nonofficial Drugs. Lippincott. p. 322. https://books.google.com/books?id=b3YYAQAAIAAJ. "Abbott Laboratories Capsules Estriol: 0.12 mg. and 0.24 mg. Eli Lilly and Company Pulvules Estriol: 0.06 mg., 0.12 mg. and 0.24 mg. Parke, Davis & Company Kapseals Theelol: 0.24 mg."
- ↑ New York State Journal of Medicine. Medical Society of the State of New York. 1939. p. 1760. https://books.google.com/books?id=9K5MAQAAIAAJ.
- ↑ "Natürliche und Synthetische Sexualhormone – Biologische Grundlagen und Behandlungsprinzipien" (in de). Grundlagen und Klinik der Menschlichen Fortpflanzung. Walter de Gruyter. 1988. pp. 229–306. ISBN 978-3110109689. OCLC 35483492. https://books.google.com/books?id=v4HvAQAACAAJ.
- ↑ 33.0 33.1 "Bioidentical hormones for menopausal hormone therapy: variation on a theme". J Gen Intern Med 22 (7): 1030–4. July 2007. doi:10.1007/s11606-007-0141-4. PMID 17549577.
- ↑ "The fate of a large bolus of exogenous estrogen administered to postmenopausal women". Maturitas 2 (1): 29–35. January 1980. doi:10.1016/0378-5122(80)90057-2. PMID 6250009.
- ↑ "Mass spectrometric and mass fragmentographic determination of natural and synthetic steroids in biological fluids". J. Steroid Biochem. 6 (3–4): 247–59. 1975. doi:10.1016/0022-4731(75)90140-5. PMID 1186230.
- ↑ Pocket Books (2005). The PDR Pocket Guide to Prescription Drugs. Simon and Schuster. pp. 1540–. ISBN 978-1-4165-1085-7. https://books.google.com/books?id=Cm9K0j4GqTYC&pg=PA1540.
- ↑ Mosby's Handbook of Herbs and Supplements and Their Therapeutic Uses. Mosby/Healthgate. 2003. ISBN 978-0-323-02015-2. https://archive.org/details/isbn_9780323020152.
- ↑ 38.0 38.1 38.2 "The Biological Target Potential of Organometallic Steroids". Bioorganometallic Chemistry: Applications in Drug Discovery, Biocatalysis, and Imaging. John Wiley & Sons. 20 April 2015. pp. 45–. ISBN 978-3-527-33527-5. https://books.google.com/books?id=n7ynBQAAQBAJ&pg=PA45.
- ↑ 39.0 39.1 39.2 39.3 "Evaluation of ligand selectivity using reporter cell lines stably expressing estrogen receptor alpha or beta". Biochemical Pharmacology 71 (10): 1459–1469. May 2006. doi:10.1016/j.bcp.2006.02.002. PMID 16554039.
- ↑ "International Union of Basic and Clinical Pharmacology. XCVII. G Protein-Coupled Estrogen Receptor and Its Pharmacologic Modulators". Pharmacological Reviews 67 (3): 505–540. July 2015. doi:10.1124/pr.114.009712. PMID 26023144.
- ↑ 41.0 41.1 41.2 41.3 "Estriol acts as a GPR30 antagonist in estrogen receptor-negative breast cancer cells". Molecular and Cellular Endocrinology 320 (1–2): 162–170. May 2010. doi:10.1016/j.mce.2010.02.006. PMID 20138962.
- ↑ 42.0 42.1 "Inhibition of GPR30 by estriol prevents growth stimulation of triple-negative breast cancer cells by 17β-estradiol". BMC Cancer 14 (1): 935. December 2014. doi:10.1186/1471-2407-14-935. PMID 25496649.
- ↑ "Receptor Binding as a Tool in the Development of New Bioactive Steroids". Drug Design. Medicinal Chemistry: A Series of Monographs 11: 169–214. 1979. doi:10.1016/B978-0-12-060308-4.50010-X. ISBN 9780120603084. https://books.google.com/books?id=bhAlBQAAQBAJ&pg=PA169.
- ↑ "Unique steroid congeners for receptor studies". Cancer Research 38 (11 Pt 2): 4186–4198. November 1978. PMID 359134. http://cancerres.aacrjournals.org/content/38/11_Part_2/4186.short.
- ↑ "Towards the mapping of the progesterone and androgen receptors". Journal of Steroid Biochemistry 27 (1–3): 255–269. 1987. doi:10.1016/0022-4731(87)90317-7. PMID 3695484.
- ↑ "Steroid hormone receptors and pharmacology". Journal of Steroid Biochemistry 12: 143–157. January 1980. doi:10.1016/0022-4731(80)90264-2. PMID 7421203.
- ↑ "Transport of steroid hormones: binding of 21 endogenous steroids to both testosterone-binding globulin and corticosteroid-binding globulin in human plasma". The Journal of Clinical Endocrinology and Metabolism 53 (1): 58–68. July 1981. doi:10.1210/jcem-53-1-58. PMID 7195404.
- ↑ 48.0 48.1 48.2 Clinical Endocrinology: Theory and Practice. Springer Science & Business Media. 6 December 2012. pp. 548,551. ISBN 978-3-642-96158-8. https://books.google.com/books?id=DAgJCAAAQBAJ&pg=PA551. "The polymer of estradiol or estriol and phosphoric acid has an excellent depot action when given intramuscularly (polyestriol phosphate or polyestradiol phosphate) (Table 16). Phosphoric acid combines with the estrogen molecule at C3 and C17 to form a macromolecule. The compound is stored in the liver and spleen where the estrogen is steadily released by splitting off of the phosphate portion due to the action of alkaline phosphatase. [...] Conjugated estrogens and polyestriol and estradiol phosphate can also be given intravenously in an aqueous solution. Intravenous administration of ovarian hormones offers no advantages, however, and therefore has no practical significance. [...] The following duarations of action have been obtained with a single administration (WlED, 1954; LAURITZEN, 1968): [...] 50 mg polyestradiol phosphate ~ 1 month; 50 mg polyestriol phosphate ~ 1 month; 80 mg polyestriol phosphate ~ 2 months."
- ↑ 49.0 49.1 49.2 49.3 49.4 49.5 49.6 "Pharmacokinetics of oestrogens and progestogens". Maturitas 12 (3): 171–97. September 1990. doi:10.1016/0378-5122(90)90003-O. PMID 2170822.
- ↑ "Endocrine Control of the Mammary Gland". Physiological Reviews 16 (3): 488–526. 1936. doi:10.1152/physrev.1936.16.3.488. ISSN 0031-9333.
- ↑ "Antiovulatory Activity of Several Synthetic and Natural Estrogens". Ovulation: Stimulation, Suppression, and Detection. Lippincott. 1966. pp. 243–253. ISBN 9780397590100. https://books.google.com/books?id=le1qAAAAMAAJ.
- ↑ "Biological Properties of Estrogen Sulfates". Chemical and Biological Aspects of Steroid Conjugation. Springer. 1970. pp. 368–408. doi:10.1007/978-3-642-49793-3_8. ISBN 978-3-642-49506-9. https://link.springer.com/chapter/10.1007/978-3-642-95177-0_8.
- ↑ 53.0 53.1 53.2 "Control of Steroid Receptor Levels and Steroid Antagonism". Female Sex Steroids: Receptors and Function. Monographs on Endocrinology. 14. Springer. 1979. pp. 99–134. doi:10.1007/978-3-642-81339-9_6. ISBN 978-3-642-81341-2.
- ↑ 54.0 54.1 54.2 "Nuclear binding and retention of the receptor estrogen complex: relation to the agonistic and antagonistic properties of estriol". Endocrinology 100 (1): 91–6. January 1977. doi:10.1210/endo-100-1-91. PMID 830547.
- ↑ "Hormontherapie". Gynäkologische Endokrinologie und Fortpflanzungsmedizin: Band 1: Gynäkologische Endokrinologie. Springer-Verlag. 17 April 2013. pp. 88–. ISBN 978-3-662-07635-4. https://books.google.com/books?id=mBF9BwAAQBAJ&pg=PA88.
- ↑ "Hormone der Gonaden". Hormonelle Therapie für die Frau. Kliniktaschenbücher. Springer. 1991. pp. 59–124. doi:10.1007/978-3-642-95670-6_6. ISBN 978-3-540-54554-5.
- ↑ "Comparison of a triphasic oestradiol/norethisterone acetate preparation with and without an oestriol component in the treatment of climacteric complaints". Maturitas 5 (4): 233–43. April 1984. doi:10.1016/0378-5122(84)90016-1. PMID 6429481.
- ↑ 58.0 58.1 "Maternal plasma progesterone levels fall after rectal administration of estriol". J. Clin. Endocrinol. Metab. 78 (1): 70–2. January 1994. doi:10.1210/jcem.78.1.8288717. PMID 8288717.
- ↑ Hormones. Academic Press. 23 October 1997. pp. 398–. ISBN 978-0-08-053413-8. https://archive.org/details/hormones00norm.
- ↑ Textbook of Perinatal Medicine, Second Edition. CRC Press. 25 September 2006. pp. 699–. ISBN 978-1-4398-1469-7. https://books.google.com/books?id=qQpz7Oj5zZMC&pg=PA699.
- ↑ Creasy and Resnik's Maternal-Fetal Medicine: Principles and Practice E-Book. Elsevier Health Sciences. 25 November 2008. pp. 115–. ISBN 978-1-4377-2135-5. https://books.google.com/books?id=AvwqSi_MbNMC&pg=PA115.
- ↑ 62.0 62.1 "In vivo uptake and subcellular distribution of tritium-labeled estrogens in human endometrium, myometrium, and vagina". J. Clin. Endocrinol. Metab. 56 (1): 76–86. January 1983. doi:10.1210/jcem-56-1-76. PMID 6847874.
- ↑ The Menopause. Springer Science & Business Media. 6 December 2012. pp. 62–. ISBN 978-1-4612-5525-3. https://books.google.com/books?id=z0LuBwAAQBAJ&pg=PA62.
- ↑ 64.0 64.1 Osteoimmunology: Interactions of the Immune and Skeletal Systems. Elsevier Science. 23 September 2015. pp. 216–. ISBN 978-0-12-800627-6. https://books.google.com/books?id=eOGcBAAAQBAJ&pg=PA216.
- ↑ "Nuclear receptor-estrogen complex: in vivo and in vitro binding of estradiol and estriol as influenced by serum albumin". J. Steroid Biochem. 5 (2): 103–7. April 1974. doi:10.1016/0022-4731(74)90114-9. PMID 4366454.
- ↑ "Identification of 16 alpha-hydroxy-estrone as a metabolite of estriol". Gynecol. Endocrinol. 2 (3): 215–21. September 1988. doi:10.3109/09513599809029346. PMID 3227988.
- ↑ "Uptake and metabolism of estradiol by normal and abnormal breast tissues". Ann. N. Y. Acad. Sci. 586 (1): 252–8. 1990. doi:10.1111/j.1749-6632.1990.tb17813.x. PMID 2357005. Bibcode: 1990NYASA.586..252T.
- ↑ Cite error: Invalid
<ref>tag; no text was provided for refs namedWentz1988 - ↑ Cite error: Invalid
<ref>tag; no text was provided for refs namedpmid18464021 - ↑ Cite error: Invalid
<ref>tag; no text was provided for refs namedUniversityofCalifornia1986 - ↑ "The relationship between urinary oestrogens and oestrogens produced in the body". The Journal of Endocrinology 16 (2): 202–212. December 1957. doi:10.1677/joe.0.0160202. PMID 13491750.
- ↑ 72.0 72.1 "Clinical investigations of a long-acting oestriol (polyoestriol phosphate)". Acta Endocrinologica 38 (1): 73–87. September 1961. doi:10.1530/acta.0.0380073. PMID 13759555.
- ↑ Human Physiology, Biochemistry and Basic Medicine. Elsevier Science. 13 October 2015. pp. 122–. ISBN 978-0-12-803717-1. https://books.google.com/books?id=BzYdCAAAQBAJ&pg=PA122.
- ↑ 74.0 74.1 "Behandlung klimakterisher Beschwerden mit Polyöstriolphosphat" (in de). Munch Med Wochenschr 113 (5): 166–9. January 1971. PMID 5107471.
- ↑ The Management of the Menopause & Post-Menopausal Years: The Proceedings of the International Symposium held in London 24–26 November 1975 Arranged by the Institute of Obstetrics and Gynaecology, The University of London. Springer Science & Business Media. 6 December 2012. pp. 395–. ISBN 978-94-011-6165-7. https://books.google.com/books?id=WT3sCAAAQBAJ&pg=PA395. "In the Federal Republic of Germany between 10 and 20% of all climacteric women are on estrogen treatment. We have the following oral estrogens for a treatment. (t) Conjugated estrogens, (2) estradiol valerate, (3) ethinyl-estradiol and its cyclopentyl-enol ether, (4) stilbestrol, (5) ethinyl-estradiol-methyltestosterone, (6) estriol and estriol succinate, most of them as coated tablets. Several long acting injectable preparations are available: several esters of combined estradiol-testosterone, one of estradiol-dehydroepiandrosterone enanthate and a prolonged polyestriol phosphate are also available. Lastly, depot injections of estradiol- and stilbestrol-esters are on the market."
- ↑ 76.0 76.1 76.2 "Estetrol review: profile and potential clinical applications". Climacteric 11 (Suppl 1): 47–58. 2008. doi:10.1080/13697130802073425. PMID 18464023.
- ↑ 77.0 77.1 77.2 "Clinical applications for estetrol". J. Steroid Biochem. Mol. Biol. 114 (1–2): 85–9. March 2009. doi:10.1016/j.jsbmb.2008.12.013. PMID 19167495. http://www.kup.at/kup/pdf/9090.pdf.
- ↑ "Estetrol - Mithra Pharmaceuticals/Pantarhei Bioscience". AdisInsight. Springer Nature Switzerland AG. http://adisinsight.springer.com/drugs/800044874.
- ↑ "Drospirenone/Estetrol - Mithra Pharmaceuticals". AdisInsight. Springer Nature Switzerland AG. https://adisinsight.springer.com/drugs/800034634.
- ↑ 80.0 80.1 "Endocrine Therapy for Gynecologic Disorders". Medical Clinics of North America 25 (1): 155–168. 1941. doi:10.1016/S0025-7125(16)36624-X. ISSN 0025-7125.
- ↑ "Sex Hormone Therapy in Gynæcology". Edinburgh Medical Journal 43 (11): 680–695. November 1936. PMID 29648134.
- ↑ 82.0 82.1 "Estrogens: Their Use in Pediatrics". California and Western Medicine 55 (5): 237–239. November 1941. PMID 18746057.
- ↑ 83.0 83.1 "Clinical Use of Extracts from the Ovaries". Journal of the American Medical Association 125 (1): 1. 1944. doi:10.1001/jama.1944.02850190003001. ISSN 0002-9955.
- ↑ "The Use of Œstrogens in Obstetrics and Gynæcology". Edinburgh Medical Journal 47 (6): 406–424. June 1940. PMID 29646930.
- ↑ 85.0 85.1 85.2 The Quest for Cortisone. MSU Press. 1 January 2012. pp. 54–. ISBN 978-1-60917-326-5. https://books.google.com/books?id=70vvFrtpePoC&pg=PT54.
- ↑ 86.0 86.1 86.2 "Studies on estrogen conjugates". Recent Progress in Hormone Research: The Proceedings of the Laurentian Hormone Conference. Elsevier Science. 22 October 2013. pp. 307–. ISBN 978-1-4832-1945-5. https://books.google.com/books?id=jgTgBAAAQBAJ&pg=PA307.
- ↑ "A survey of estrogens". Postgraduate Medicine 20 (3): 224–231. September 1956. doi:10.1080/00325481.1956.11691266. PMID 13359169.
- ↑ 88.0 88.1 Concise Dictionary of Pharmacological Agents: Properties and Synonyms. Springer Science & Business Media. 6 December 2012. pp. 206, 905. ISBN 978-94-011-4439-1. https://books.google.com/books?id=tsjrCAAAQBAJ&pg=PA206.
- ↑ Organic-chemical drugs and their synonyms: (an international survey). Wiley-VCH. 4 October 2001. ISBN 978-3-527-30247-5. https://books.google.com/books?id=zWpqAAAAMAAJ. "8075-01 (6628-01) 37452-43-0 R Polymeric ester with phosphoric acid S Klimadurin, Polyestriol phosphate, Polyostriolphosphat, Triodurin U Depot-estrogen"
- ↑ The Extra Pharmacopoeia. Pharmaceutical Press. 1993. p. 2258. ISBN 978-0-85369-300-0. https://books.google.com/books?id=EGZWAAAAYAAJ. "Polyoestriol Phosphate. [...] ingredient of Klimadurin. [...] Triodurin [...]."
- ↑ 91.0 91.1 91.2 "Estriol succinate - Synthetic Biologics". AdisInsight. Springer Nature Switzerland AG. https://adisinsight.springer.com/drugs/800026520.
- ↑ Geriatrics 3: Gynecology · Orthopaedics · Anesthesiology · Surgery · Otorhinolaryngology · Ophthalmology · Dermatology. Springer Science & Business Media. 6 December 2012. pp. 6–. ISBN 978-3-642-68976-5. https://books.google.com/books?id=vwzpCAAAQBAJ&pg=PA6.
- ↑ "Bioidentical hormone therapy". Mayo Clin. Proc. 86 (7): 673–80, quiz 680. July 2011. doi:10.4065/mcp.2010.0714. PMID 21531972.
- ↑ Canine and Feline Endocrinology and Reproduction. Elsevier Health Sciences. 1 January 2004. pp. 839–. ISBN 0-7216-9315-6. https://books.google.com/books?id=elPuvsucuY8C&pg=PA839.
Further reading
- "Estriol: a review". Physiol. Rev. 38 (3): 463–80. 1958. doi:10.1152/physrev.1958.38.3.463. PMID 13567043.
- "Mechanism of action of estrogen agonists and antagonists". J. Anim. Sci. 49 (Suppl 2): 46–65. 1979. doi:10.1093/ansci/49.supplement_ii.46. PMID 400777.
- "The agonistic and antagonistic effects of short acting estrogens: a review". Pharmacol. Ther. 21 (3): 429–53. 1983. doi:10.1016/0163-7258(83)90063-3. PMID 6356176.
- "The agonistic and antagonistic actions of estriol". J. Steroid Biochem. 20 (4B): 1005–13. April 1984. doi:10.1016/0022-4731(84)90011-6. PMID 6202959.
- "Estriol in the postmenopause". Acta Obstet Gynecol Scand Suppl 139: 2–23. 1987. doi:10.3109/00016348709156470. PMID 3475930.
- "Estriol: a weak estrogen or a different hormone?". Gynecol. Endocrinol. 5 (2): 131–53. June 1991. doi:10.3109/09513599109028436. PMID 1927578.
- "Review of the endometrial safety during intravaginal treatment with estriol". Eur. J. Obstet. Gynecol. Reprod. Biol. 62 (1): 101–6. September 1995. doi:10.1016/0301-2115(95)02170-c. PMID 7493689. https://www.ejog.org/article/0301-2115(95)02170-C/pdf.
- "Estriol: safety and efficacy". Altern Med Rev 3 (2): 101–13. 1998. PMID 9577246. http://archive.foundationalmedicinereview.com/publications/3/2/101.pdf.
- "Unconventional estrogens: estriol, biest, and triest". Clin Obstet Gynecol 44 (4): 864–79. December 2001. doi:10.1097/00003081-200112000-00024. PMID 11600867.
- "Znaczenie endogennego i egzogennego estriolu w praktyce klinicznej" (in pl). Ginekol. Pol. 77 (7): 559–65. July 2006. PMID 17076208.
- "[The role of estriol in therapy of urogenital disorders in postmenopausal women]" (in ru). Urologiia (3): 102–104, 107. 2007. PMID 17722629.
- "The bioidentical hormone debate: are bioidentical hormones (estradiol, estriol, and progesterone) safer or more efficacious than commonly used synthetic versions in hormone replacement therapy?". Postgrad Med 121 (1): 73–85. January 2009. doi:10.3810/pgm.2009.01.1949. PMID 19179815.
- "Use of lactobacilli and estriol combination in the treatment of disturbed vaginal ecosystem: a review". J Turk Ger Gynecol Assoc 12 (4): 239–46. 2011. doi:10.5152/jtgga.2011.57. PMID 24592002.
- "Estriol: emerging clinical benefits". Menopause 24 (9): 1081–1085. September 2017. doi:10.1097/GME.0000000000000855. PMID 28375935.
- "The efficacy and safety of estriol to treat vulvovaginal atrophy in postmenopausal women: a systematic literature review". Climacteric 20 (4): 321–330. August 2017. doi:10.1080/13697137.2017.1329291. PMID 28622049.
- "Treatment of vaginal atrophy with estriol and lactobacilli combination: a clinical review". Climacteric 21 (2): 140–147. April 2018. doi:10.1080/13697137.2017.1421923. PMID 29381086.
- "Treatment of genitourinary syndrome of menopause: the potential effects of intravaginal ultralow-concentration oestriol and intravaginal dehydroepiandrosterone on quality of life and sexual function". Prz Menopauzalny 18 (2): 116–122. June 2019. doi:10.5114/pm.2019.86836. PMID 31488961.
 |

