Chemistry:Hydroxyphenamate
 | |
| Clinical data | |
|---|---|
| Trade names | Listica |
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| Chemical and physical data | |
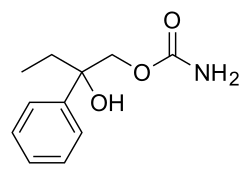
| Formula | C11H15NO3 |
| Molar mass | 209.245 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Hydroxyphenamate or oxyfenamate (trade name Listica) is a sedative and anxiolytic drug of the carbamate class which is no longer marketed in the US. Like other carbamate sedatives, it is chemically related to meprobamate (Miltown). It was introduced to the US market in 1961. The dosage for adults is 200 mg 3 to 4 times daily.[1]
Synthesis

The reaction of Propiophenone [93-55-0] (1) with cyanide gives 2-hydroxy-2-phenylbutanenitrile CID:13089766 (2). Acid hydrolysis of the nitrile to a carboxylic acid gives 2-hydroxy-2-phenylbutanoic acid [35468-69-0] (3). The reduction by hydride of the acid to the alcohol gives 2-phenyl-1,2-butanediol, CID:13260818 (4).
For the final step in the synthesis, treatment with Ethyl chloroformate followed by addition of aqueous ammonia gave the carbamate, and hence hydroxyphenamate (5).
References
- ↑ American Medical Association Dept of Drugs (1977). AMA Drug Evaluations (3rd ed.). Littleton, Mass.: Pub. Sciences Group. p. N-61. ISBN 978-0-88416-175-2. OCLC 1024170745.
- ↑ Lednicer & Mitscher, book 1, page 220.
- ↑ Robert H Sifferd & Leo D Braitberg, U.S. Patent 3,066,164 (1962 to Armour Pharmaceutical Co).
 |
