Chemistry:Fulvestrant
 | |
| Clinical data | |
|---|---|
| Pronunciation | /fʊlˈvɛstrənt/ fuul-VES-trənt |
| Trade names | Faslodex, others |
| Other names | ICI-182780; ZD-182780; ZD-9238; 7α-[9-[(4,4,5,5,5-Pentafluoropentyl)-sulfinyl]nonyl]estra-1,3,5(10)-triene-3,17β-diol |
| AHFS/Drugs.com | Monograph |
| License data | |
| Pregnancy category |
|
| Routes of administration | Intramuscular injection |
| Drug class | Antiestrogen |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | Low[1] |
| Protein binding | 99%[1] |
| Metabolism | Hydroxylation, conjugation (glucuronidation, sulfation)[1] |
| Elimination half-life | IM: 40–50 days[1] |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| Chemical and physical data | |
| Formula | C32H47F5O3S |
| Molar mass | 606.78 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Fulvestrant, sold under the brand name Faslodex among others, is an antiestrogenic medication used to treat hormone receptor (HR)-positive metastatic breast cancer in postmenopausal women with disease progression as well as HR-positive, HER2-negative advanced breast cancer in combination with abemaciclib or palbociclib in women with disease progression after endocrine therapy.[2] It is given by injection into a muscle.[3]
Fulvestrant is a selective estrogen receptor degrader (SERD) and was first-in-class to be approved.[4] It works by binding to the estrogen receptor and destabilizing it, causing the cell's normal protein degradation processes to destroy it.[4]
Fulvestrant was approved for medical use in the United States in 2002.[5]
Medical uses
Breast cancer
Fulvestrant is used for the treatment of hormone receptor positive metastatic breast cancer or locally advanced unresectable disease in postmenopausal women; it is given by injection.[3] A 2017 Cochrane review found it is as safe and effective as first line or second line endocrine therapy.[3]
It is also used to treat ER-positive, HER2-negative advanced or metastatic breast cancer in combination with abemaciclib or palbociclib in women with disease progression after first-line endocrine therapy.[2]
Due to the medication's having a chemical structure similar to that of estrogen, it can interact with immunoassays for blood estradiol concentrations and show falsely elevated results.[6][7][8] This can improperly lead to discontinuing the treatment.[6]
Early puberty
Fulvestrant has been used in the treatment of peripheral precocious puberty in girls with McCune–Albright syndrome.[9][10][11]
Available forms
Fulvestrant is provided in a castor oil solution also containing alcohol, benzyl alcohol, and benzyl benzoate.[2] It is supplied at a concentration of 250 mg/5 mL.[2]
Contraindications
Fulvestrant should not be used in women with kidney failure or who are pregnant.[2][12]
Side effects
Very common (occurring in more than 10% of people) adverse effects include nausea, injection site reactions, weakness, and elevated transaminases. Common (between 1% and 10%) adverse effects include urinary tract infections, hypersensitivity reactions, loss of appetite, headache, blood clots in veins, hot flushes, vomiting, diarrhea, elevated bilirubin, rashes, and back pain.[12] In a large clinical trial, the incidence of venous thromboembolism (VTE) with fulvestrant was 0.9%.[2]
Pharmacology
Pharmacodynamics
Fulvestrant is an antiestrogen which acts as an antagonist of the estrogen receptor (ER) and additionally as a selective estrogen receptor degrader (SERD).[4] It works by binding to the estrogen receptor and making it more hydrophobic, which makes the receptor unstable and misfold, which in turn leads normal processes inside the cell to degrade it.[4]
In addition to its antiestrogenic activity, fulvestrant is an agonist of the G protein-coupled estrogen receptor (GPER), albeit with relatively low affinity (10–100 nM, relative to 3–6 nM for estradiol).[13][14][15][16][17]
Pharmacokinetics
Fulvestrant after an intramuscular injection is slowly absorbed and maximal levels (Cmax) are reached after 5 days on average with a range of 2 to 19 days.[18] The elimination half-life of fulvestrant with intramuscular injection is 40 to 50 days.[19][2] This is 40 times longer than the half-life of fulvestrant by intravenous injection, indicating that its long half-life with intramuscular injection is due to slow absorption from the injection site.[18] Levels of fulvestrant with 500 mg/month by intramuscular injection (and a single additional 500 mg loading dose on day 15 of therapy) in postmenopausal women with advanced breast cancer were 25.1 ng/mL (25,100 pg/mL) at peak and 28.0 ng/mL (28,000 pg/mL) at trough with a single dose and 28.0 ng/mL (28,000 pg/mL) at peak and 12.2 ng/mL (12,200 pg/mL) at trough after multiple doses at steady state.[2]
Fulvestrant does not cross the blood–brain barrier in animals and may not in humans as well.[20][21][22] Accordingly, no effects of fulvestrant on brain function have been observed in preclinical or clinical research.[21][22] Fulvestrant is highly (99%) bound to plasma proteins.[19][2] It is bound to very low density lipoprotein, low density lipoprotein, and high density lipoprotein, but not to sex hormone-binding globulin.[19]
Fulvestrant appears to be metabolized along similar pathways as endogenous steroids; CYP3A4 may be involved, but non-cytochrome P450 routes appear to be more important. It does not inhibit any cytochrome P450 enzymes. Elimination is almost all via feces.[12]
Fulvestrant can form colloidal aggregates at certain concentration ranges and this can limit its activity as well as produce bell-shaped concentration–response curves.[23][24][25]
Chemistry
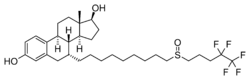
Fulvestrant, also known as 7α-[9-[(4,4,5,5,5-pentafluoropentyl)sulfinyl]nonyl]estradiol, is a synthetic estrane steroid and a derivative of estradiol. An alkyl-sulfinyl moiety was added to the endogenous estrogen receptor ligand.[4]
It was discovered through rational drug design, but was selected for further development via phenotypic screening.[26]
History
Fulvestrant was the first selective estrogen receptor degrader to be approved.[4] It was approved in the United States in 2002[2] and in Europe in 2004.[12]
Society and culture
NICE evaluation
The U.K. National Institute for Health and Clinical Excellence (NICE) said in 2011 that it found no evidence Faslodex was significantly better than existing treatments, so its widespread use would not be a good use of resources for the country's National Health Service. The first month's treatment of Faslodex, which starts with a loading dose, costs £1,044.82 ($1,666), and subsequent treatments cost £522.41 a month.[citation needed] In the 12 months ending June 2015, the UK price (excluding VAT) of a month's supply of anastrozole (Arimidex), which is off patent, cost 89 pence/day, and letrozole (Femara) cost £1.40/day.[27][28][29]
Patent extension
The original patent for Faslodex expired in October 2004. Drugs subject to pre-marketing regulatory review are eligible for patent extension, and for this reason AstraZeneca got an extension of the patent to December 2011.[30][31] AstraZeneca has filed later patents. A generic version of Faslodex has been approved by the FDA. However, this does not mean that the product will necessarily be commercially available - possibly because of drug patents and/or drug exclusivity.[32] A later patent for Faslodex expires in January 2021.[33] Atossa Genetics has a patent for the administration of fulvestrant into the breast via a microcatheter invented by Susan Love.[34]
Research
Fulvestrant was studied in endometrial cancer but results were not promising and as of 2016 development for this use was abandoned.[35]
Because fulvestrant cannot be given orally, efforts have been made to develop SERD drugs that can be taken by mouth, including brilanestrant and elacestrant.[4] The clinical success of fulvestrant also led to efforts to discover and develop a parallel drug class of selective androgen receptor degraders (SARDs).[4]
ZB716, or fulvestrant-3-boronic acid, is an oral prodrug of fulvestrant which is under development.[36][37][38]
References
- ↑ 1.0 1.1 1.2 1.3 Dörwald, Florencio Zaragoza (4 February 2013). Lead Optimization for Medicinal Chemists: Pharmacokinetic Properties of Functional Groups and Organic Compounds. John Wiley & Sons. pp. 486–. ISBN 978-3-527-64565-7. https://books.google.com/books?id=YTeY9ZEfNccC&pg=PA486.
- ↑ 2.0 2.1 2.2 2.3 2.4 2.5 2.6 2.7 2.8 2.9 "US Label: Fulvestrant". FDA. July 2016. https://www.accessdata.fda.gov/drugsatfda_docs/label/2019/021344Orig1s039lbl.pdf.
- ↑ 3.0 3.1 3.2 "Fulvestrant for hormone-sensitive metastatic breast cancer". The Cochrane Database of Systematic Reviews 1 (1): CD011093. January 2017. doi:10.1002/14651858.CD011093.pub2. PMID 28043088.
- ↑ 4.0 4.1 4.2 4.3 4.4 4.5 4.6 4.7 "Induced protein degradation: an emerging drug discovery paradigm". Nature Reviews. Drug Discovery 16 (2): 101–114. February 2017. doi:10.1038/nrd.2016.211. PMID 27885283.
- ↑ "Fulvestrant". The American Society of Health-System Pharmacists. https://www.drugs.com/monograph/fulvestrant.html.
- ↑ 6.0 6.1 "Estradiol immunoassays – interference from the drug fulvestrant (Faslodex®) may cause falsely elevated estradiol results Medical safety alert - GOV.UK" (in en). UK Medicines and Healthcare products Regulatory Agency. 24 March 2016. https://www.gov.uk/drug-device-alerts/-estradiol-immunoassays-interference-from-the-drug-fulvestrant-faslodex-may-cause-falsely-elevated-estradiol-results.
- ↑ "Oestradiol measurement during fulvestrant treatment for breast cancer". Br J Cancer 120 (4): 404–406. February 2019. doi:10.1038/s41416-019-0378-9. PMID 30679781.
- ↑ "Fulvestrant falsely elevates oestradiol levels in immunoassays in postmenopausal women with breast cancer". Eur J Cancer 126: 104–105. February 2020. doi:10.1016/j.ejca.2019.10.015. PMID 31927211.
- ↑ "Treatment and outcomes of precocious puberty: an update". The Journal of Clinical Endocrinology and Metabolism 98 (6): 2198–207. June 2013. doi:10.1210/jc.2013-1024. PMID 23515450.
- ↑ "Disorders of Puberty: Pharmacotherapeutic Strategies for Management". Pediatric Pharmacotherapy. Handbook of Experimental Pharmacology. 261. Springer. May 2019. pp. 507–538. doi:10.1007/164_2019_208. ISBN 978-3-030-50493-9.
- ↑ "Fulvestrant treatment of precocious puberty in girls with McCune-Albright syndrome". International Journal of Pediatric Endocrinology 2012 (1): 26. September 2012. doi:10.1186/1687-9856-2012-26. PMID 22999294.
- ↑ 12.0 12.1 12.2 12.3 "Faslodex 250 mg solution for injection - Summary of Product Characteristics". UK Electronic Medicines Compendium. 21 July 2016. https://www.medicines.org.uk/emc/medicine/14381.
- ↑ "International Union of Basic and Clinical Pharmacology. XCVII. G Protein-Coupled Estrogen Receptor and Its Pharmacologic Modulators". Pharmacol. Rev. 67 (3): 505–40. July 2015. doi:10.1124/pr.114.009712. PMID 26023144.
- ↑ "Identity of an estrogen membrane receptor coupled to a G protein in human breast cancer cells". Endocrinology 146 (2): 624–32. February 2005. doi:10.1210/en.2004-1064. PMID 15539556.
- ↑ "The G-protein-coupled estrogen receptor GPER in health and disease". Nat Rev Endocrinol 7 (12): 715–26. August 2011. doi:10.1038/nrendo.2011.122. PMID 21844907.
- ↑ "Estrogen biology: new insights into GPER function and clinical opportunities". Mol. Cell. Endocrinol. 389 (1–2): 71–83. May 2014. doi:10.1016/j.mce.2014.02.002. PMID 24530924.
- ↑ "Position paper: The membrane estrogen receptor GPER--Clues and questions". Steroids 77 (10): 935–42. August 2012. doi:10.1016/j.steroids.2012.04.001. PMID 22521564.
- ↑ 18.0 18.1 "Fulvestrant: pharmacokinetics and pharmacology". Br J Cancer 90 (Suppl 1): S7–10. March 2004. doi:10.1038/sj.bjc.6601630. PMID 15094758.
- ↑ 19.0 19.1 19.2 "Fulvestrant: a review of its use in the management of hormone receptor-positive metastatic breast cancer in postmenopausal women". Drugs 71 (3): 363–80. February 2011. doi:10.2165/11204810-000000000-00000. PMID 21319872.
- ↑ "ICI 182,780 (Fulvestrant)--the first oestrogen receptor down-regulator--current clinical data". Br. J. Cancer 85 (Suppl 2): 11–4. November 2001. doi:10.1054/bjoc.2001.1982. PMID 11900210.
- ↑ 21.0 21.1 "Clinical development of fulvestrant ("Faslodex")". Cancer Treat. Rev. 31 (Suppl 2): S3–9. 2005. doi:10.1016/j.ctrv.2005.08.010. PMID 16198055.
- ↑ 22.0 22.1 "Fulvestrant (Faslodex): current status in the therapy of breast cancer". Expert Rev Anticancer Ther 2 (2): 151–60. April 2002. doi:10.1586/14737140.2.2.151. PMID 12113237.
- ↑ "Colloidal aggregation: from screening nuisance to formulation nuance". Nano Today 19: 188–200. April 2018. doi:10.1016/j.nantod.2018.02.011. PMID 30250495.
- ↑ "Colloidal Drug Aggregate Stability in High Serum Conditions and Pharmacokinetic Consequence". ACS Chem Biol 14 (4): 751–757. April 2019. doi:10.1021/acschembio.9b00032. PMID 30840432.
- ↑ "Colloidal drug formulations can explain "bell-shaped" concentration-response curves". ACS Chem Biol 9 (3): 777–84. March 2014. doi:10.1021/cb4007584. PMID 24397822.
- ↑ "Phenotypic screening in cancer drug discovery - past, present and future". Nature Reviews. Drug Discovery 13 (8): 588–602. August 2014. doi:10.1038/nrd4366. PMID 25033736.
- ↑ UK Department of Health Commercial Medicines Unit Electronic Medicines Information Tool, London, 2015
- ↑ UK’s NICE says no to AstraZeneca breast cancer drug Faslodex, The Pharma Letter, 10 November 2011
- ↑ National Institute for Health and Clinical Excellence Guidance Breast cancer (metastatic) - fulvestrant
- ↑ Patent Term Extensions The United States Patent and Trademark Office.
- ↑ Determination of Regulatory Review Period for Purposes of Patent Extension; FASLODEX A Notice by the Food and Drug Administration on 04/17/2003
- ↑ Generic Faslodex Availability, Drugs.COM
- ↑ Pink Ribbon Blues: How Breast Cancer Culture Undermines Women's Health By Gayle A. Sulik, Oxford University Press (Oct. 2010)
- ↑ Hung DT, Love S, "Methods for identifying treating or monitoring asymptomatic patients for risk reduction or therapeutic treatment of breast cancer", US patent granted 6638727, issued 28 October 2003, assigned to Cytyc Health Corp
- ↑ "Fulvestrant for the treatment of endometrial cancer". Expert Opinion on Investigational Drugs 25 (4): 475–83. 2016. doi:10.1517/13543784.2016.1154532. PMID 26882357.
- ↑ Ahmad, I., Mathew, S., & Rahman, S. (2020). Recent progress in selective estrogen receptor downregulators (SERDs) for the treatment of breast cancer. RSC Medicinal Chemistry, 11(4), 438–454. https://doi.org/10.1039/C9MD00570F
- ↑ "Fulvestrant-3 Boronic Acid (ZB716): An Orally Bioavailable Selective Estrogen Receptor Downregulator (SERD)". J. Med. Chem. 59 (17): 8134–40. 2016. doi:10.1021/acs.jmedchem.6b00753. PMID 27529700.
- ↑ ClinicalTrials.gov: NCT04669587
External links
- "Fulvestrant". Drug Information Portal. U.S. National Library of Medicine. https://druginfo.nlm.nih.gov/drugportal/name/fulvestrant.
 |

