Biology:Conjugated estrogens
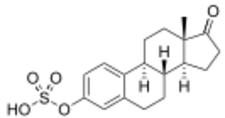 Estrone sulfate, the primary active component in conjugated estrogens (constitutes about 50 to 70% of total content) | |
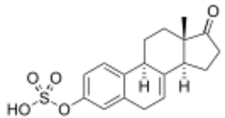 Equilin sulfate, the second most major active component in conjugated estrogens (constitutes about 20 to 30% of total content) | |
| Combination of | |
|---|---|
| Estrone sulfate | Estrogen |
| Equilin sulfate | Estrogen |
| 17α-Dihydro-equilin sulfate | Estrogen |
| Clinical data | |
| Trade names | Cenestin, Enjuvia, Congest, C.E.S., Premarin, Prempro (with MPA), Premphase (with MPA), others |
| Other names | CEs; Conjugated equine estrogens; CEEs; Pregnant mares' urine; Premarin; Estrogens, conjugated |
| AHFS/Drugs.com | Consumer Drug Information |
| Pregnancy category |
|
| Routes of administration | By mouth, topical, vaginal, intravenous injection, intramuscular injection[1][2] |
| Drug class | Estrogen |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | Variable[3] |
| Protein binding | High (to albumin and SHBG)[3][1] |
| Metabolism | Liver[3][1] |
| Elimination half-life | Estrone: 26.7 hours Estrone (BA): 14.8 hours Equilin: 11.4 hours[4] |
| Excretion | Urine[3] |
| Identifiers | |
| CAS Number | |
| PubChem CID | |
| PubChem SID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| (verify) | |
Conjugated estrogens (CEs), or conjugated equine estrogens (CEEs), sold under the brand name Premarin among others, is an estrogen medication which is used in menopausal hormone therapy and for various other indications.[5][3][1][6] It is a mixture of the sodium salts of estrogen conjugates found in horses, such as estrone sulfate and equilin sulfate.[1][6][5] CEEs are available in the form of both natural preparations manufactured from the urine of pregnant mares and fully synthetic replications of the natural preparations.[7][8] They are formulated both alone and in combination with progestins such as medroxyprogesterone acetate.[5] CEEs are usually taken by mouth, but can also be given by application to the skin or vagina as a cream or by injection into a blood vessel or muscle.[1][2]
Side effects of CEEs include breast tenderness and enlargement, headache, fluid retention, and nausea among others.[3][1] It may increase the risk of endometrial hyperplasia and endometrial cancer in women with an intact uterus if it is not taken together with a progestogen like progesterone.[3][1] The medication may also increase the risk of blood clots, cardiovascular disease, and, when combined with most progestogens, breast cancer.[9] CEEs are estrogens, or agonists of the estrogen receptor, the biological target of estrogens like estradiol.[1][3] Compared to estradiol, certain estrogens in CEEs are more resistant to metabolism, and the medication shows relatively increased effects in certain parts of the body like the liver.[1] This results in an increased risk of blood clots and cardiovascular problems with CEEs relative to estradiol.[1][10]
Premarin, the major brand of CEEs in use, is manufactured by Pfizer and was first marketed in 1941 in Canada and in 1942 in the United States .[6] It is the most commonly used form of estrogen in menopausal hormone therapy in the United States.[11][12] However, it has begun to fall out of favor relative to bioidentical estradiol, which is the most widely used form of estrogen in Europe for menopausal hormone therapy.[12][13][14][15] CEEs are available widely throughout the world.[5] An estrogen preparation very similar to CEEs but differing in source and composition is esterified estrogens.[1] In 2020, it was the 283rd most commonly prescribed medication in the United States, with more than 1 million prescriptions.[16][17]
Medical uses
CEEs are a form of hormone therapy used in women.[18] It is used most commonly in postmenopausal women who have had a hysterectomy to treat hot flashes, and burning, itching, and dryness of the vagina and surrounding areas.[19] It must be used in combination with a progestogen in women who have not had a hysterectomy.[1] For women already taking the medication, it can be used to treat osteoporosis, although it is not recommended solely for this use.[20] Some lesser known uses are as a means of high-dose estrogen therapy in the treatment of breast cancer in both women and men and in the treatment of prostate cancer in men.[21][22] It has been used at a dosage of 2.5 mg three times per day (7.5 mg/day total) for prostate cancer.[23][24]
CEEs are specifically approved in countries such as the United States and Canada for the treatment of moderate to severe vasomotor symptoms (hot flashes) and vulvovaginal atrophy (atrophic vaginitis, atrophic urethritis) associated with menopause, hypoestrogenism due to hypogonadism, ovariectomy, or primary ovarian failure, abnormal uterine bleeding, the palliative treatment of metastatic breast cancer in women, the palliative treatment of advanced androgen-dependent prostate cancer in men, and the prevention of postmenopausal osteoporosis.[4][25][5] The intravenous formulation of CEEs is specifically used to rapidly limit bleeding in women with hemorrhage due to dysfunctional uterine bleeding.[2][26]: 318 [27]: 60
Available forms
Natural CEEs, as Premarin, are available in the form of oral tablets (0.3 mg, 0.625 mg, 0.9 mg, 1.25 mg, or 2.5 mg), creams for topical or vaginal administration (0.625 mg/g), and vials for intravenous or intramuscular injection (25 mg/vial).[2][28] Synthetic CEEs, such as Cenestin (Synthetic A), Enjuvia (Synthetic B), and generic formulations, are available in the form of oral tablets (0.3 mg, 0.45 mg, 0.625 mg, 0.9 mg, or 1.25 mg) and creams for topical or vaginal administration (0.625 mg/g).[2][29]
Contraindications
Contraindications of CEEs include breast cancer and a history of venous thromboembolism, among others.[citation needed]
Side effects
The most common side effects associated with CEEs are vaginal yeast infections, vaginal spotting or bleeding, painful menses, and cramping of the legs. While there are some contradictory data, estrogen alone does not appear to increase the risk of coronary heart disease or breast cancer, unlike the case of estrogen in combination with certain progestins such as levonorgestrel or medroxyprogesterone acetate.[30] Only a few clinical studies have assessed differences between oral CEEs and oral estradiol in terms of health parameters.[31] Oral CEEs have been found to possess a significantly greater risk of thromboembolic and cardiovascular complications than oral estradiol (OR = 2.08) and oral esterified estrogens (OR = 1.78).[31][32][33] However, in another study, the increase in venous thromboembolism risk with oral CEEs plus medroxyprogesterone acetate and oral estradiol plus norethisterone acetate was found to be equivalent (RR = 4.0 and 3.9, respectively).[34][35] As of present, there are no randomized controlled trials that would allow for unambiguous conclusions.[31]
| Clinical outcome | Hypothesized effect on risk |
Estrogen and progestogen (CEs 0.625 mg/day p.o. + MPA 2.5 mg/day p.o.) (n = 16,608, with uterus, 5.2–5.6 years follow up) |
Estrogen alone (CEs 0.625 mg/day p.o.) (n = 10,739, no uterus, 6.8–7.1 years follow up) | ||||
|---|---|---|---|---|---|---|---|
| HR | 95% CI | AR | HR | 95% CI | AR | ||
| Coronary heart disease | Decreased | 1.24 | 1.00–1.54 | +6 / 10,000 PYs | 0.95 | 0.79–1.15 | −3 / 10,000 PYs |
| Stroke | Decreased | 1.31 | 1.02–1.68 | +8 / 10,000 PYs | 1.37 | 1.09–1.73 | +12 / 10,000 PYs |
| Pulmonary embolism | Increased | 2.13 | 1.45–3.11 | +10 / 10,000 PYs | 1.37 | 0.90–2.07 | +4 / 10,000 PYs |
| Venous thromboembolism | Increased | 2.06 | 1.57–2.70 | +18 / 10,000 PYs | 1.32 | 0.99–1.75 | +8 / 10,000 PYs |
| Breast cancer | Increased | 1.24 | 1.02–1.50 | +8 / 10,000 PYs | 0.80 | 0.62–1.04 | −6 / 10,000 PYs |
| Colorectal cancer | Decreased | 0.56 | 0.38–0.81 | −7 / 10,000 PYs | 1.08 | 0.75–1.55 | +1 / 10,000 PYs |
| Endometrial cancer | – | 0.81 | 0.48–1.36 | −1 / 10,000 PYs | – | – | – |
| Hip fractures | Decreased | 0.67 | 0.47–0.96 | −5 / 10,000 PYs | 0.65 | 0.45–0.94 | −7 / 10,000 PYs |
| Total fractures | Decreased | 0.76 | 0.69–0.83 | −47 / 10,000 PYs | 0.71 | 0.64–0.80 | −53 / 10,000 PYs |
| Total mortality | Decreased | 0.98 | 0.82–1.18 | −1 / 10,000 PYs | 1.04 | 0.91–1.12 | +3 / 10,000 PYs |
| Global index | – | 1.15 | 1.03–1.28 | +19 / 10,000 PYs | 1.01 | 1.09–1.12 | +2 / 10,000 PYs |
| Diabetes | – | 0.79 | 0.67–0.93 | 0.88 | 0.77–1.01 | ||
| Gallbladder disease | Increased | 1.59 | 1.28–1.97 | 1.67 | 1.35–2.06 | ||
| Stress incontinence | – | 1.87 | 1.61–2.18 | 2.15 | 1.77–2.82 | ||
| Urge incontinence | – | 1.15 | 0.99–1.34 | 1.32 | 1.10–1.58 | ||
| Peripheral artery disease | – | 0.89 | 0.63–1.25 | 1.32 | 0.99–1.77 | ||
| Probable dementia | Decreased | 2.05 | 1.21–3.48 | 1.49 | 0.83–2.66 | ||
| Abbreviations: CEs = conjugated estrogens. MPA = medroxyprogesterone acetate. p.o. = per oral. HR = hazard ratio. AR = attributable risk. PYs = person–years. CI = confidence interval. Notes: Sample sizes (n) include placebo recipients, which were about half of patients. "Global index" is defined for each woman as the time to earliest diagnosis for coronary heart disease, stroke, pulmonary embolism, breast cancer, colorectal cancer, endometrial cancer (estrogen plus progestogen group only), hip fractures, and death from other causes. Sources: See template. | |||||||
| v]] · [[Template talk:Risk of venous thromboembolism with hormone therapy and birth control pills (QResearch/CPRD) | d]] · e
Risk of venous thromboembolism (VTE) with hormone therapy and birth control (QResearch/CPRD) | ||
| Type | Route | Medications | Odds ratio (95% CI) |
|---|---|---|---|
| Menopausal hormone therapy | Oral | Estradiol alone ≤1 mg/day >1 mg/day |
1.27 (1.16–1.39)* 1.22 (1.09–1.37)* 1.35 (1.18–1.55)* |
| Conjugated estrogens alone ≤0.625 mg/day >0.625 mg/day |
1.49 (1.39–1.60)* 1.40 (1.28–1.53)* 1.71 (1.51–1.93)* | ||
| Estradiol/medroxyprogesterone acetate | 1.44 (1.09–1.89)* | ||
| Estradiol/dydrogesterone ≤1 mg/day E2 >1 mg/day E2 |
1.18 (0.98–1.42) 1.12 (0.90–1.40) 1.34 (0.94–1.90) | ||
| Estradiol/norethisterone ≤1 mg/day E2 >1 mg/day E2 |
1.68 (1.57–1.80)* 1.38 (1.23–1.56)* 1.84 (1.69–2.00)* | ||
| Estradiol/norgestrel or estradiol/drospirenone | 1.42 (1.00–2.03) | ||
| Conjugated estrogens/medroxyprogesterone acetate | 2.10 (1.92–2.31)* | ||
| Conjugated estrogens/norgestrel ≤0.625 mg/day CEEs >0.625 mg/day CEEs |
1.73 (1.57–1.91)* 1.53 (1.36–1.72)* 2.38 (1.99–2.85)* | ||
| Tibolone alone | 1.02 (0.90–1.15) | ||
| Raloxifene alone | 1.49 (1.24–1.79)* | ||
| Transdermal | Estradiol alone ≤50 μg/day >50 μg/day |
0.96 (0.88–1.04) 0.94 (0.85–1.03) 1.05 (0.88–1.24) | |
| Conjugated estrogens alone | 1.04 (0.76–1.43) | ||
| Estradiol/progestogen | 0.88 (0.73–1.01) | ||
| Vaginal | Estradiol alone | 0.84 (0.73–0.97) | |
| Combined birth control | Oral | Ethinylestradiol/norethisterone | 2.56 (2.15–3.06)* |
| Ethinylestradiol/levonorgestrel | 2.38 (2.18–2.59)* | ||
| Ethinylestradiol/norgestimate | 2.53 (2.17–2.96)* | ||
| Ethinylestradiol/desogestrel | 4.28 (3.66–5.01)* | ||
| Ethinylestradiol/gestodene | 3.64 (3.00–4.43)* | ||
| Ethinylestradiol/drospirenone | 4.12 (3.43–4.96)* | ||
| Ethinylestradiol/cyproterone acetate | 4.27 (3.57–5.11)* | ||
| Notes: (1) Nested case–control studies (2015, 2019) based on data from the QResearch and Clinical Practice Research Datalink (CPRD) databases. (2) Bioidentical progesterone was not included, but is known to be associated with no additional risk. Footnotes: * = Statistically significant (p < 0.01). Sources: See template. | |||
Overdose
Estrogens, including CEEs, are relatively safe in acute overdose.[citation needed]
Interactions
Inhibitors and inducers of cytochrome P450 enzymes may interact with CEEs.[citation needed]
Pharmacology
Pharmacodynamics
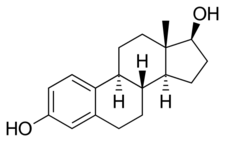
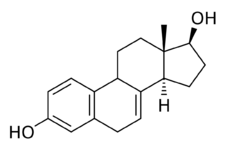
CEEs are a combination of estrogens, or agonists of the estrogen receptors.[1] The major estrogen in CEEs, sodium estrone sulfate, itself is inactive, and rather serves as a prodrug of estrone and then of estradiol.[1][36][37] The transformation of estrone sulfate to estrone is catalyzed by steroid sulfatase, and of estrone into estradiol by 17β-hydroxysteroid dehydrogenase.[1][38] CEEs (as Premarin) and estrone have been found to be equivalent in potency in an animal model of estrogenic activity.[6] On the other hand, the active forms of the equine estrogens in CEEs, such as equilin and 17β-dihydroequilin, have greater potency in the liver relative to bioidentical estradiol, similarly to synthetic estrogens like ethinylestradiol and diethylstilbestrol.[1] This results in disproportionate effects on liver protein production compared to estradiol, although to a lesser extent than ethinylestradiol and diethylstilbestrol.[1] In addition, 17β-dihydroequilenin has shown a selective estrogen receptor modulator (SERM)-like profile of estrogenic activity in studies with monkeys, in which beneficial effects on bone and the cardiovascular system were observed but proliferative responses in breast or endometrium were not seen, although the clinical significance of this is unknown.[39]
CEEs consists of the sodium salts of the sulfate esters of equine estrogens in a specific and consistent composition (see the table).[1][6] The major estrogens in CEEs are sodium estrone sulfate and sodium equilin sulfate, which together account for approximately 71.5–92.0% of the total content of CEEs.[5][1][6] CEEs are prodrugs of the active forms of the estrogens.[1][6][5] Sodium estrone sulfate is a prodrug of estrone, which in turn is a prodrug of estradiol, while sodium equilin sulfate is a prodrug of equilin and then of 17β-dihydroequilin.[1] As such, the major active estrogens with CEEs are estradiol and 17β-dihydroequilin, which have potent estrogenic activity and account for most of the effects of CEEs.[1] The 17α-estrogens in CEEs such as 17α-estradiol and 17α-dihydroequilin have low estrogenicity and are thought to contribute minimally to its effects.[1] There are many different steroids in natural CEE products like Premarin, as many as 230 compounds and including even androgens and progestogens, but only the estrogens are present in sufficient amounts to produce clinically-relevant effects.[6][40][11]
A dosage of 0.625 mg/day oral CEEs has been found to increase SHBG levels by 100%.[40][41] For comparison, 1 mg/day oral estradiol increased SHBG levels by 45%, while 50 µg/day transdermal estradiol increased SHBG levels by 12%.[40][41] Ethinylestradiol is more potent in its effects on liver protein synthesis than either CEEs or estradiol, with 10 µg/day oral ethinylestradiol having been found to be approximately equivalent to 1.25 mg/day CEEs.[40]
| Compound | Synonym | Proportion (%) | Relative potency in the vagina (%) |
Relative potency in the uterus (%) |
RBA for ERα (%) |
RBA for ERβ (%) |
ERα / ERβ RBA ratio |
|---|---|---|---|---|---|---|---|
| Conjugated estrogens | – | 100 | 38 | 100 | – | – | – |
| Estrone | – | 49.1–61.5 | 30 | 32 | 26 | 52 | 0.50 |
| Equilin | Δ7-Estrone | 22.4–30.5 | 42 | 80 | 13 | 49 | 0.26 |
| 17α-Dihydroequilin | Δ7-17α-Estradiol | 13.5–19.5 | 0.06 | 2.6 | 41 | 32 | 1.30 |
| 17α-Estradiol | – | 2.5–9.5 | 0.11 | 3.5 | 19 | 42 | 0.45 |
| Δ8-Estrone | – | 3.5–3.9 | ? | ? | 19 | 32 | 0.60 |
| Equilenin | Δ6,8-Estrone | 2.2–2.8 | 1.3 | 11.4 | 15 | 20–29 | 0.50–0.75 |
| 17β-Dihydroequilin | Δ7-17β-Estradiol | 0.5–4.0 | 83 | 200 | 113 | 108 | 1.05 |
| 17α-Dihydroequilenin | Δ6,8-17α-Estradiol | 1.2–1.6 | 0.018 | 1.3 | 20 | 49 | 0.40 |
| 17β-Estradiol | – | 0.56–0.9 | 100 | ? | 100 | 100 | 1.00 |
| 17β-Dihydroequilenin | Δ6,8-17β-Estradiol | 0.5–0.7 | 0.21 | 9.4 | 68 | 90 | 0.75 |
| Δ8-17β-Estradiol | – | Small amounts | ? | ? | 68 | 72 | 0.94 |
| Notes: All listed compounds are present in conjugated estrogen products specifically in the form of the sodium salts of the Sulfate|Sulfate esters (i.e., as sodium estrone sulfate, sodium equilin sulfate, etc.). Sources: See template. | |||||||
Antigonadotropic effects

A preliminary study of ovulation inhibition in women found that oral CEEs was 33% effective at 1.25 mg/day and 94% at 3.75 mg/day.[43][44] A dosage of oral CEEs of 2.5 mg three times daily (7.5 mg/day total) has been found to suppress total testosterone levels in men to an equivalent extent as 3 mg/day oral diethylstilbestrol, which is the minimum dosage of diethylstilbestrol required to consistently suppress total testosterone levels into the castrate range (<50 ng/dL).[45]
Pharmacokinetics
CEEs are hydrolyzed in the intestines during first-pass metabolism upon oral administration.[46][5] Following their absorption, they are resulfated mainly in the liver also during the first pass.[46] Following this, they serve as a circulating reservoir and are slowly rehydrolyzed into their unconjugated active forms.[46]
Oral CEEs, at a daily dosage of 0.625 mg, achieve estrone and estradiol levels of 150 pg/mL and 30–50 pg/mL, respectively, while a daily oral dosage of 1.25 mg achieves levels of 120–200 pg/mL and 40–60 pg/mL of estrone and estradiol, respectively.[47] The oral ingestion of 10 mg CEEs, which contains about 4.5 mg sodium estrone sulfate and 2.5 mg sodium equilin sulfate, produces maximal plasma concentrations of estrone and equilin of 1,400 pg/mL and 560 pg/mL within three and five hours, respectively.[47] By 24 hours post-dose of 10 mg, the levels of estrone and equilin fall to 280 pg/mL and 125 pg/mL, respectively.[47] Oral CEEs 1.25 mg/daily and oral micronized estradiol 1 mg/daily result in similar plasma concentrations of estrone and estradiol (150–300 pg/mL and 30–50 pg/mL for micronized estradiol, respectively) (oral estradiol is extensively metabolized into estrone during hepatic first-pass metabolism),[47] although this does not account for equilin and other equine estrogens involved in the effects of CEEs, which may be significantly more potent in comparison to estrone.[48][49] The pharmacokinetics of vaginal CEEs[50] and of intravenous CEEs have been studied as well.[51]
Eoncentrations of equilin that are very high relative to those of other estrogens are produced by typical clinical doses of CEEs.[52] With a dosage of 1.25 mg oral CEEs, equilin levels of 1,082 to 2,465 pg/mL have been observed.[52] The clinical significance of these levels of equilin is unknown.[52]
The active forms are metabolized primarily in the liver.[5] There is some enterohepatic recirculation of CEEs.[5] Following a single oral dose of 0.625 CEEs, the biological half-life of estrone was 26.7 hours, of baseline-adjusted estrone was 14.8 hours, and of equilin was 11.4 hours.[4]
| Compound | RBA to SHBG (%) |
Bound to SHBG (%) |
Bound to albumin (%) |
Total bound (%) |
MCR (L/day/m2) |
|---|---|---|---|---|---|
| 17β-Estradiol | 50 | 37 | 61 | 98 | 580 |
| Estrone | 12 | 16 | 80 | 96 | 1050 |
| Estriol | 0.3 | 1 | 91 | 92 | 1110 |
| Estrone sulfate | 0 | 0 | 99 | 99 | 80 |
| 17β-Dihydroequilin | 30 | ? | ? | ? | 1250 |
| Equilin | 8 | 26 | 13 | ? | 2640 |
| 17β-Dihydroequilin sulfate | 0 | ? | ? | ? | 375 |
| Equilin sulfate | 0 | ? | ? | ? | 175 |
| Δ8-Estrone | ? | ? | ? | ? | 1710 |
| Notes: RBA for SHBG (%) is compared to 100% for testosterone. Sources: See template. | |||||
Chemistry
CEEs are naturally occurring estrane steroids.[1][6] They are in conjugate form, as the sodium salts of the C17β sulfate esters.[1][6] The estrogens in CEEs, in their unconjugated active forms, include bioidentical human estrogens like estradiol and estrone as well as equine-specific estrogens such as equilin and 17β-dihydroequilin.[1][6] The equine estrogens differ from human estrogens in that they have additional double bonds in the B ring of the steroid nucleus.[1][6] CEEs contain both 17β-estrogens like estradiol and 17β-dihydroequilin and the C17α epimers like 17α-estradiol and 17α-dihydroequilin.[1][6]
History
Conjugated estriol, an extract of the urine of pregnant women and sold under the brand names Progynon and Emmenin in the 1930s, was the predecessor of Premarin.[53] Both of these products contained conjugated estrogens similarly to Premarin, but the estrogens were human estrogens as opposed to equine estrogens and the composition differed. The major active ingredient in Progynon and Emmenin was estriol glucuronide.
Estrone sulfate was first isolated from the urine of pregnant mares in the late 1930s by researchers in the Department of Biochemistry at University of Toronto.[54] Premarin was first introduced in 1941 by Wyeth Ayerst as a treatment for hot flashes and other symptoms of menopause; at that time, Wyeth Ayerst only had to prove its safety, and not its efficacy.[55] In response to the 1962 Kefauver Harris Amendment the FDA had its efficacy reviewed, and in 1972 found it effective for menopausal symptoms and probably effective for osteoporosis.[56] The review also determined that two estrogens – estrone sulfate and equilin sulfate – were primarily responsible for the activity of Premarin, and it laid the groundwork for Abbreviated New Drug Application (ANDA) submissions of generic versions.[55] In 1984 an NIH consensus panel found that estrogens were effective for preventing osteoporosis[57] and 1986 the FDA announced in the Federal Register that Premarin was effective for preventing osteoporosis.[58] This announcement led to a rapid growth in sales, and interest from generic manufacturers to introduce generic versions.[55]
Conjugated estrogens was introduced for medical use under the brand name Premarin in Canada in 1941, in the United States in 1942, and in the United Kingdom in 1956.[59]
The manufacturer of Premarin secretly paid gynecologist Robert A. Wilson to promote its use by menopausal women in his 1966 book, Feminine Forever, leading to increased sales.[60]
Society and culture
Names
Estrogens, conjugated is the generic name of the drug and its USP and JAN.[61] It is also known as conjugated estrogens or as conjugated equine estrogens.[4] The brand name Premarin is a contraction of "pregnant mares' urine".[62][63][64]
CEEs are marketed under a large number of brand names throughout the world.[5] The major brand name of the natural form of CEEs manufactured from the urine of pregnant mares is Premarin.[5] Major brand names of fully synthetic versions of CEEs include Cenestin and Enjuvia in the United States and C.E.S. and Congest in Canada .[5][7][8] CEEs are also formulated in combination with progestins.[5] Major brand names of CEEs in combination with medroxyprogesterone acetate include Prempro and Premphase in the United States, Premplus in Canada, Premique in the United Kingdom and Ireland, Premia in Australia and New Zealand, and Premelle in South Africa .[5][65] Prempak-C is a combination of CEEs and norgestrel which is used in the United Kingdom and Ireland, and Prempak N is a combination of CEEs and medrogestone which is used in South Africa.[5] Many of the aforementioned brand names are also used in other, non-English-speaking countries.[5]
Availability
CEEs are marketed and available widely throughout the world.[5][25] This includes in all English-speaking countries, throughout the European Union, Latin America, Asia, and elsewhere in the world.[5][25]
Health effects
Research starting in 1975 showed substantially increased risk of endometrial cancer.[66][67] Since 1976 the drug has carried a label warning about the risk.[68] As part of the Women's Health Initiative sponsored by the National Institutes of Health, a large-scale clinical trial of menopausal HRT showed that long-term use of estrogen and a progestin may increase the risk of strokes, heart attacks, blood clots, and breast cancer.[69] Following these results, Wyeth experienced a significant decline in its sales of Premarin, Prempro (CEEs and medroxyprogesterone acetate), and related products, from over $2 billion in 2002 to just over $1 billion in 2006.[70]
Litigation
This drug has been the subject of litigation; more than 13,000 people have sued Wyeth between 2002 and 2009. Wyeth and Pharmacia & Upjohn prevailed in the vast majority of hormone therapy cases previously set for trial through a combination of rulings by judges, verdicts by juries, and dismissals by plaintiffs themselves.[71] Of the company's losses, two of the jury verdicts were reversed post-trial and others are being challenged on appeal. Wyeth also won five summary judgments on Prempro cases and had 15 cases voluntarily dismissed by plaintiffs. The company won dismissals in another 3,000 cases.[72] In 2006, Mary Daniel, in a trial in Philadelphia, was awarded $1.5 million in compensatory damages as well as undisclosed punitive damages. As of 2010, Wyeth had won the last four of five cases, most recently in Virginia, finding that they were not responsible for the breast cancer of plaintiff Georgia Torkie-Tork.[73] Wyeth has been quoted as saying "many risk factors associated with breast cancer have been identified, but science cannot establish what role any particular risk factor or combination play in any individual woman's breast cancer."[74] Wyeth's counsel in the case also noted that in the WHI trial, 99.62% of women took the drug and "did not get breast cancer."[72]
Animal welfare
Animal welfare groups claim that animal husbandry and urine collection methods used in the production of CEEs cause undue stress and suffering to the mares involved. Animal activists have made claims of abuses ranging from inadequate stall size, long periods of confinement, cumbersome urine collection, and continuous breeding cycles. After reaching advanced age, many of the mares are adopted for recreation use, while some are sent to feed lots for slaughter. Despite the controversy, the USDA called the CEEs HRT industry a model of self-regulation.[75]
Notes
References
- ↑ 1.00 1.01 1.02 1.03 1.04 1.05 1.06 1.07 1.08 1.09 1.10 1.11 1.12 1.13 1.14 1.15 1.16 1.17 1.18 1.19 1.20 1.21 1.22 1.23 1.24 1.25 1.26 1.27 1.28 1.29 1.30 "Pharmacology of estrogens and progestogens: influence of different routes of administration". Climacteric 8 (Suppl 1): 3–63. August 2005. doi:10.1080/13697130500148875. PMID 16112947.
- ↑ 2.0 2.1 2.2 2.3 2.4 "Drugs@FDA: FDA Approved Drug Products". United States Food and Drug Administration. http://www.accessdata.fda.gov/scripts/cder/daf/.
- ↑ 3.0 3.1 3.2 3.3 3.4 3.5 3.6 3.7 "PREMARIN- estrogens, conjugated tablet, film coated Wyeth Pharmaceuticals LLC, a subsidiary of Pfizer Inc.". http://labeling.pfizer.com/showlabeling.aspx?id=131.
- ↑ 4.0 4.1 4.2 4.3 "Conjugated estrogens". https://www.drugbank.ca/drugs/DB00286.
- ↑ 5.00 5.01 5.02 5.03 5.04 5.05 5.06 5.07 5.08 5.09 5.10 5.11 5.12 5.13 5.14 5.15 5.16 5.17 5.18 "Sex hormones and their modulators". Martindale: The Complete Drug Reference (36th ed.). London: Pharmaceutical Press. 2009. p. 2087. ISBN 978-0-85369-840-1. https://www.medicinescomplete.com/mc/rem/current/mono-E61.htm?q=C&t=advanced&ss=mn&p=57.
- ↑ 6.00 6.01 6.02 6.03 6.04 6.05 6.06 6.07 6.08 6.09 6.10 6.11 6.12 Clinical Gynecologic Endocrinology and Infertility. Lippincott Williams & Wilkins. 28 March 2012. pp. 751–3. ISBN 978-1-4511-4847-3. https://books.google.com/books?id=KZLubBxJEwEC&pg=PA751.
- ↑ 7.0 7.1 Pharmacology for Pharmacy Technicians Pageburst E-Book on VitalSource2: Pharmacology for Pharmacy Technicians Pageburst E-Book on VitalSource. Elsevier Health Sciences. 1 December 2012. pp. 573–. ISBN 978-0-323-08578-6. https://books.google.com/books?id=1nvESqVnxc0C&pg=PA573.
- ↑ 8.0 8.1 IARC Working Group on the Evaluation of Carcinogenic Risks to Humans; World Health Organization; International Agency for Research on Cancer (2007). Combined Estrogen-progestogen Contraceptives and Combined Estrogen-progestogen Menopausal Therapy. World Health Organization. pp. 378–. ISBN 978-92-832-1291-1. https://books.google.com/books?id=aGDU5xibtNgC&pg=PA378.
- ↑ "Safety and benefit considerations for menopausal hormone therapy". Expert Opinion on Drug Safety 16 (8): 941–954. August 2017. doi:10.1080/14740338.2017.1343298. PMID 28664754.
- ↑ "Hormones and venous thromboembolism among postmenopausal women". Climacteric 17 (Suppl 2): 34–37. December 2014. doi:10.3109/13697137.2014.956717. PMID 25223916.
- ↑ 11.0 11.1 Cite error: Invalid
<ref>tag; no text was provided for refs namedpmid24176763 - ↑ 12.0 12.1 "Hormone Therapy (I): Estrogens, Progestogens, and Androgens". Menopause. Springer. 2017. pp. 181–196. doi:10.1007/978-3-319-59318-0_11. ISBN 978-3-319-59317-3.
- ↑ "Bioidentical menopausal hormone therapy: registered hormones (non-oral estradiol ± progesterone) are optimal". Climacteric 20 (4): 331–338. August 2017. doi:10.1080/13697137.2017.1291607. PMID 28301216.
- ↑ "What if the Women's Health Initiative had used transdermal estradiol and oral progesterone instead?". Menopause 21 (7): 769–783. July 2014. doi:10.1097/GME.0000000000000169. PMID 24398406.
- ↑ "The bioidentical hormone debate: are bioidentical hormones (estradiol, estriol, and progesterone) safer or more efficacious than commonly used synthetic versions in hormone replacement therapy?". Postgraduate Medicine 121 (1): 73–85. January 2009. doi:10.3810/pgm.2009.01.1949. PMID 19179815.
- ↑ "The Top 300 of 2020". https://clincalc.com/DrugStats/Top300Drugs.aspx.
- ↑ "Estrogens, Conjugated - Drug Usage Statistics". https://clincalc.com/DrugStats/Drugs/EstrogensConjugated.
- ↑ (in en) Women's vascular health. CRC Press. 29 December 2006. ISBN 9780340809976. https://books.google.com/books?id=GsPie79hwsIC&pg=PA222.
- ↑ (in en) Nezhat's Operative gynecologic laparoscopy and hysteroscopy. Cambridge University Press. 7 July 2008. ISBN 9781139472005. https://books.google.com/books?id=Z0gYy2hdn3QC&pg=PA266. Retrieved 7 May 2015.
- ↑ "An overview on the treatment of postmenopausal osteoporosis". Arquivos Brasileiros de Endocrinologia e Metabologia 58 (2): 162–171. March 2014. doi:10.1590/0004-2730000003039. PMID 24830593.
- ↑ "Breast cancer: major risk factors and recent developments in treatment". Asian Pacific Journal of Cancer Prevention 15 (8): 3353–3358. 2014. doi:10.7314/apjcp.2014.15.8.3353. PMID 24870721.
- ↑ (in en) 2015 Nurse's Drug Handbook. Jones & Bartlett Publishers. 2015-01-14. ISBN 9781284091373. https://books.google.com/books?id=o_vPBgAAQBAJ&q=premarin+prostate+cancer&pg=PA448. Retrieved 2015-05-07.
- ↑ Estrogens and Antiestrogens II: Pharmacology and Clinical Application of Estrogens and Antiestrogen. Springer Science & Business Media. 6 December 2012. pp. 540–. ISBN 978-3-642-60107-1. https://books.google.com/books?id=wBvyCAAAQBAJ&pg=PA540.
- ↑ Textbook of Prostate Cancer: Pathology, Diagnosis and Treatment: Pathology, Diagnosis and Treatment. CRC Press. 1 March 1999. pp. 297–. ISBN 978-1-85317-422-3. https://books.google.com/books?id=GreZlojD-tYC&pg=PA297.
- ↑ 25.0 25.1 25.2 "Premarin". https://www.drugs.com/international/premarin.html.
- ↑ "Hormonal Treatment of Disorders of the Menstrual Cycle". Ovarian Function and its Disorders. Developments in Obstetrics and Gynecology. Springer Science & Business Media. 1981. pp. 309–332. doi:10.1007/978-94-009-8195-9_11. ISBN 978-94-009-8195-9. https://books.google.com/books?id=7IrpCAAAQBAJ&pg=PA310.
- ↑ The Cyclopedia of Medicine, Surgery, Specialties. F. A. Davis Company. 1975. https://books.google.com/books?id=RiE9K5bgKcsC.
- ↑ Endocrinology of Aging. Springer Science & Business Media. 5 November 1999. pp. 172–. ISBN 978-1-59259-715-4. https://books.google.com/books?id=hGD0BwAAQBAJ&pg=PA172.
- ↑ Drugs for the Geriatric Patient E-Book: Text with BONUS Handheld Software. Elsevier Health Sciences. 11 April 2007. pp. 462–. ISBN 978-1-4377-1035-9. https://books.google.com/books?id=-gmjBQAAQBAJ&pg=PA462.
- ↑ "Premarin (Conjugated estrogens) Vaginal Cream". https://www.fda.gov/Safety/MedWatch/SafetyInformation/Safety-RelatedDrugLabelingChanges/ucm121062.htm.
- ↑ 31.0 31.1 31.2 "Menopausal hormone therapy and venous thromboembolism". Przeglad Menopauzalny = Menopause Review 13 (5): 267–272. October 2014. doi:10.5114/pm.2014.46468. PMID 26327865.
- ↑ "Lower risk of cardiovascular events in postmenopausal women taking oral estradiol compared with oral conjugated equine estrogens". JAMA Internal Medicine 174 (1): 25–31. January 2014. doi:10.1001/jamainternmed.2013.11074. PMID 24081194.
- ↑ "Esterified estrogens and conjugated equine estrogens and the risk of venous thrombosis". JAMA 292 (13): 1581–1587. October 2004. doi:10.1001/jama.292.13.1581. PMID 15467060.
- ↑ "How do you decide on hormone replacement therapy in women with risk of venous thromboembolism?". Blood Reviews 31 (3): 151–157. May 2017. doi:10.1016/j.blre.2016.12.001. PMID 27998619.
- ↑ "The risk of venous thrombosis in women over 50 years old using oral contraception or postmenopausal hormone therapy". Journal of Thrombosis and Haemostasis 11 (1): 124–131. January 2013. doi:10.1111/jth.12060. PMID 23136837.
- ↑ The Menopause. Springer Science & Business Media. 6 December 2012. pp. 64–. ISBN 978-1-4612-5525-3. https://books.google.com/books?id=z0LuBwAAQBAJ&pg=PA64.
- ↑ Clinical Reproductive Medicine and Surgery: A Practical Guide. Springer Science & Business Media. 22 May 2013. pp. 5–6. ISBN 978-1-4614-6837-0. https://books.google.com/books?id=TAYnR1b8jRkC&pg=PA5.
- ↑ Primary Care in Obstetrics and Gynecology: A Handbook for Clinicians. Springer Science & Business Media. January 1998. pp. 220, 227. ISBN 978-0-387-94739-6. https://books.google.com/books?id=jfmB3aNSGfoC&pg=PA227. "Conjugated estrogens are absorbed with peak levels at 4 hours and a half-life of approximately 12 hours."
- ↑ "Assessing the mammary gland of nonhuman primates: effects of endogenous hormones and exogenous hormonal agents and growth factors". Birth Defects Research. Part B, Developmental and Reproductive Toxicology 80 (2): 126–146. April 2007. doi:10.1002/bdrb.20112. PMID 17443713.
- ↑ 40.0 40.1 40.2 40.3 "Clinical opinion: the biologic and pharmacologic principles of estrogen therapy for symptomatic menopause". MedGenMed 8 (1): 85. March 2006. PMID 16915215.
- ↑ 41.0 41.1 "Serum estradiol-binding profiles in postmenopausal women undergoing three common estrogen replacement therapies: associations with sex hormone-binding globulin, estradiol, and estrone levels". Menopause 7 (4): 243–250. 2000. doi:10.1097/00042192-200007040-00006. PMID 10914617.
- ↑ 42.0 42.1 "Plasma testosterone: an accurate monitor of hormone treatment in prostatic cancer". British Journal of Urology 45 (6): 668–677. December 1973. doi:10.1111/j.1464-410x.1973.tb12238.x. PMID 4359746.
- ↑ "Antiovulatory Activity of Several Synthetic and Natural Estrogens". Ovulation: Stimulation, Suppression, and Detection. Lippincott. 1966. pp. 243–253. ISBN 9780397590100. https://books.google.com/books?id=le1qAAAAMAAJ.
- ↑ "Biological Properties of Estrogen Sulfates". Chemical and Biological Aspects of Steroid Conjugation. Springer. 1970. pp. 368–408. doi:10.1007/978-3-642-49793-3_8. ISBN 978-3-642-49506-9.
- ↑ "Hormonal Therapy of Prostatic Cancer". Cancer 45 (Suppl 7): 1929–1936. April 1980. doi:10.1002/cncr.1980.45.s7.1929. PMID 29603164.
- ↑ 46.0 46.1 46.2 "Bioavailability of orally administered sex steroids used in oral contraception and hormone replacement therapy". Contraception 54 (2): 59–69. August 1996. doi:10.1016/0010-7824(96)00136-9. PMID 8842581.
- ↑ 47.0 47.1 47.2 47.3 Treatment of the Postmenopausal Woman: Basic and Clinical Aspects. Academic Press. 5 June 2007. pp. 771–. ISBN 978-0-08-055309-2. https://books.google.com/books?id=HB2XO5MhKakC&pg=PA771.
- ↑ The Climacteric in Perspective: Proceedings of the Fourth International Congress on the Menopause, held at Lake Buena Vista, Florida, October 28–November 2, 1984. Springer Science & Business Media. 6 December 2012. pp. 395–. ISBN 978-94-009-4145-8. https://books.google.com/books?id=VM0hBQAAQBAJ&pg=PA395.
- ↑ Gonadotrophins: Current Research. Ardent Media. 1974. pp. 157–. ISBN 978-0-8422-7205-6. https://books.google.com/books?id=s21mXv6g5NkC&pg=PA157.
- ↑ "The vaginal absorption of oestrogens in post-menopausal women". Maturitas 2 (4): 321–326. December 1980. doi:10.1016/0378-5122(80)90034-1. PMID 7231202.
- ↑ "Serum and urinary estrone sulfate during the menstrual cycle, measured by a direct radioimmunoassay, and fate of exogenously injected estrone sulfate". Hormone Research 27 (2): 61–68. 1987. doi:10.1159/000180788. PMID 3653846.
- ↑ 52.0 52.1 52.2 "Current status of estrogen therapy for the menopause". Fertility and Sterility 37 (1): 5–25. January 1982. doi:10.1016/S0015-0282(16)45970-4. PMID 6277697.
- ↑ Women, Health and Nation: Canada and the United States Since 1945. McGill-Queen's Press - MQUP. 2003. pp. 103–. ISBN 978-0-7735-2501-6. https://books.google.com/books?id=CRjtHlq1INcC&pg=PA103.
- ↑ "The isolation of estrone sulfate from the urine of pregnant mares". Journal of Biological Chemistry 126 (2): 663–669. 1938. doi:10.1016/S0021-9258(18)73874-X.
- ↑ 55.0 55.1 55.2 "The Strange Case of Premarin". Modern Drug Discovery 3 (8): 46–52. October 2000. http://pubs.acs.org/subscribe/archive/mdd/v03/i08/html/kling.html.
- ↑ "Federal register [microform"]. Washington : [Office of the Federal Register, National Archives and Records Service, General Services Administration : Distributed by the Supt. of Docs., U.S. G.P.O.]. 3 June 1972. https://archive.org/details/federalregister37gunit.
- ↑ National Institutes of Health Consensus Development Conference Statement. April 2–4, 1984 Osteoporosis
- ↑ Food and Drug Administration. May 5, 1997 Conjugated Estrogens - Letter from Dr. Janet Woodcock: Approvability of a Synthetic Generic Version of Premarin
- ↑ Managing the Menopause. Cambridge University Press. 20 August 2015. pp. 118–. ISBN 978-1-107-45182-7. https://books.google.com/books?id=l0pLCgAAQBAJ&pg=PA118. "Premarin (Pregnant Mares Urine) was introduced in Canada in 1941, in the USA in 1942 and in the UK in 1956."
- ↑ "Women Have Been Misled About Menopause" (in en-US). The New York Times. 2023-02-01. ISSN 0362-4331. https://www.nytimes.com/2023/02/01/magazine/menopause-hot-flashes-hormone-therapy.html. "Every woman has the right — indeed the duty — to counteract the chemical castration that befalls her during her middle years,” the gynecologist Robert Wilson wrote in 1966. The U.S. Food and Drug Administration approved the first hormone-therapy drug in 1942, but Wilson’s blockbuster book, “Feminine Forever,” can be considered a kind of historical landmark...Within a decade of the book’s publication, Premarin — a mix of estrogens derived from the urine of pregnant horses — was the fifth-most-prescribed drug in the United States. (Decades later, it was revealed that Wilson received funding from the pharmaceutical company that sold Premarin.)"
- ↑ "ChemIDplus - 12126-59-9 - QTTMOCOWZLSYSV-QWAPEVOJSA-M - Estrogens, conjugated [USP:JAN] - Similar structures search, synonyms, formulas, resource links, and other chemical information". https://chem.nlm.nih.gov/chemidplus/rn/12126-59-9.
- ↑ "The history of hormone therapy use and recent controversy related to heart disease and breast cancer arising from prevention trial outcomes". Journal of Midwifery & Women's Health 57 (6): 547–557. 2012. doi:10.1111/j.1542-2011.2012.00247.x. PMID 23217066.
- ↑ "Postmenopausal hormone therapy: from monkey glands to transdermal patches". The Journal of Endocrinology 185 (2): 207–222. May 2005. doi:10.1677/joe.1.05847. PMID 15845914.
- ↑ "Estrogen replacement therapy for the treatment of postmenopausal genitourinary tract dysfunction". Discovery Medicine 10 (55): 500–510. December 2010. PMID 21189221.
- ↑ The AARP Guide to Pills: Essential Information on More Than 1,200 Prescription and Nonprescription Medications, Including Generics. Sterling Publishing Company Incorporated. 1 January 2007. pp. 235–. ISBN 978-1-4027-4446-4. https://books.google.com/books?id=_OvwGMswS6gC&pg=PA235.
- ↑ "Increased risk of endometrial carcinoma among users of conjugated estrogens". The New England Journal of Medicine 293 (23): 1167–1170. December 1975. doi:10.1056/NEJM197512042932303. PMID 171569.
- ↑ "Exogenous estrogen and endometrial carcinoma: case-control and incidence study". American Journal of Obstetrics and Gynecology 127 (6): 572–580. March 1977. doi:10.1016/0002-9378(77)90351-9. PMID 190887.
- ↑ "Menopause, as Brought to You by Big Pharma". New York Times. 12 December 2009. https://www.nytimes.com/2009/12/13/business/13drug.html.
- ↑ "Effects of conjugated equine estrogen on health-related quality of life in postmenopausal women with hysterectomy: results from the Women's Health Initiative Randomized Clinical Trial". Archives of Internal Medicine 165 (17): 1976–1986. September 2005. doi:10.1001/archinte.165.17.1976. PMID 16186467. https://escholarship.org/content/qt1279619t/qt1279619t.pdf?t=ptra6f.
- ↑ "Earnings Results for the 2006 Fourth Quarter and Full Year" (PDF) (Press release). Wyeth. Archived from the original (PDF) on 2007-11-27. Retrieved 2018-02-20.
- ↑ "Pfizer Statement on Prempro". Indy News Channel. 2009-11-24. http://www.theindychannel.com/health/21716786/detail.html.
- ↑ 72.0 72.1 "Pfizer wins trial over claim Prempro caused cancer". Bloomberg. February 24, 2010.
- ↑ "Pfizer properly warned about Prempro risks, jury finds". 3 December 2010. https://www.bloomberg.com/news/2010-12-03/pfizer-properly-warned-about-prempro-health-risks-jury-finds.html.
- ↑ "Legal Intelligencer: Philadelphia jury returns defense verdict in HRT case, Amaris Elliott Engel". http://www.law.com/jsp/article.jsp?id=1202444500915.
- ↑ "The HRT horses". 19 January 2004. http://www.nbcnews.com/id/3995076/ns/dateline_nbc/t/hrt-horses/.
Further reading
- "The saga of the ring B unsaturated equine estrogens". Endocrine Reviews 9 (4): 396–416. November 1988. doi:10.1210/edrv-9-4-396. PMID 3065072.
- "Bioequivalence of conjugated estrogen products". Clinical Pharmacokinetics 24 (4): 271–274. April 1993. doi:10.2165/00003088-199324040-00001. PMID 8387902.
- "Pharmacokinetic and pharmacologic variation between different estrogen products". Journal of Clinical Pharmacology 35 (9S): 18S–24S. September 1995. doi:10.1002/j.1552-4604.1995.tb04143.x. PMID 8530713.
- "The effects of estradiol valerate plus medroxyprogesterone acetate and conjugated estrogens plus medrogestone on climacteric symptoms and metabolic variables in perimenopausal women". Acta Obstetricia et Gynecologica Scandinavica 75 (4): 386–393. April 1996. doi:10.3109/00016349609033337. PMID 8638462.
- "Pharmacokinetics and pharmacodynamics of conjugated equine estrogens: chemistry and metabolism". Proceedings of the Society for Experimental Biology and Medicine 217 (1): 6–16. January 1998. doi:10.3181/00379727-217-44199. PMID 9421201.
- "Conjugated estrogens--the natural SERMs". Gynecological Endocrinology 13 (Suppl 6): 9–12. December 1999. PMID 10862263.
- "Conjugated estrogens and breast cancer risk". Gynecological Endocrinology 13 (Suppl 6): 13–19. December 1999. PMID 10862264.
- "Estrogens and menopause: pharmacology of conjugated equine estrogens and their potential role in the prevention of neurodegenerative diseases such as Alzheimer's". The Journal of Steroid Biochemistry and Molecular Biology 85 (2–5): 473–482. June 2003. doi:10.1016/S0960-0760(03)00220-6. PMID 12943738.
- "[Postmenopausal hormone replacement therapy and cardiovascular risk: role of conjugated equine estrogens and medroxyprogesterone acetate]" (in de). Praxis 93 (21): 904–914. May 2004. doi:10.1024/0369-8394.93.21.904. PMID 15216975.
- "Pharmacology of estrogens and progestogens: influence of different routes of administration". Climacteric 8 (Suppl 1): 3–63. August 2005. doi:10.1080/13697130500148875. PMID 16112947.
- "[New evidence of conjugated estrogen and 17beta-estradiol for treatment and prevention of osteoporosis]" (in ja). Nihon Rinsho. Japanese Journal of Clinical Medicine 65 (Suppl 9): 369–373. November 2007. PMID 18161134.
- "Use of conjugated estrogens in life-threatening gastrointestinal bleeding in hemodialysis patients--a review". Clinical and Applied Thrombosis/Hemostasis 19 (3): 334–337. June 2013. doi:10.1177/1076029612437575. PMID 22411999.
- "Conjugated estrogens for the treatment of menopausal symptoms: a review of safety data". Expert Opinion on Drug Safety 13 (1): 45–56. January 2014. doi:10.1517/14740338.2013.824965. PMID 23919270.
- "Pharmacology of conjugated equine estrogens: efficacy, safety and mechanism of action". The Journal of Steroid Biochemistry and Molecular Biology 142: 16–29. July 2014. doi:10.1016/j.jsbmb.2013.10.011. PMID 24176763.
- "Pharmaco- and toxicokinetics of selected exogenous and endogenous estrogens: a review of the data and identification of knowledge gaps". Critical Reviews in Toxicology 44 (8): 696–724. September 2014. doi:10.3109/10408444.2014.930813. PMID 25099693.
External links
- "Estrogens, conjugated". Drug Information Portal. U.S. National Library of Medicine. https://druginfo.nlm.nih.gov/drugportal/name/estrogens%2C%20conjugated.
- "Steroidal estrogens". Drug Information Portal. U.S. National Library of Medicine. https://druginfo.nlm.nih.gov/drugportal/name/steroidal%20estrogens.
- WHI Follow-up Study Confirms Health Risks of Long-Term Combination Hormone Therapy Outweigh Benefits for Postmenopausal Women NIH press release, March 4, 2008
 |
