Chemistry:Fluoroestradiol F-18
From HandWiki
Short description: Chemical compound
 | |
| Clinical data | |
|---|---|
| Trade names | Cerianna |
| Other names | [18F]16α-Fluoroestradiol; [18F]16α-Fluoroestra-1,3,5(10)-triene-3,17β-diol |
| License data | |
| Pregnancy category |
|
| Routes of administration | Intravenous injection |
| Drug class | Estrogen; Diagnostic radiopharmaceutical |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Metabolism | Liver[1] |
| Excretion | Gallbladder and kidney[1] |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| Chemical and physical data | |
| Formula | C18H23[18F]O2 |
| Molar mass | 289.37 [1] |
| 3D model (JSmol) | |
| |
| |
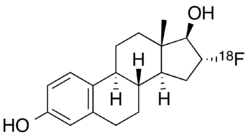
Fluoroestradiol F-18, also known as [18F]16α-fluoroestradiol and sold under the brand name Cerianna, is a radioactive diagnostic agent indicated for use with positron emission tomography (PET) imaging.[1][2][3][4][5] It is an analog of estrogen and is used to detect estrogen receptor-positive breast cancer lesions.[1]
Chemistry
Chemically, fluoroestradiol F-18 is [18F]16α-fluoro-3,17β-diol-estratriene-1,3,5(10).[1]
History
Fluoroestradiol F-18 was approved for medical use in the United States in May 2020.[6]
See also
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 1.6 "Cerianna- fluoroestradiol f 18 injection". 20 May 2020. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=b2c2a2df-cf3e-4a29-a033-231ed5a94b58.
- ↑ "The quest for improving the management of breast cancer by functional imaging: The discovery and development of 16α-[18Ffluoroestradiol (FES), a PET radiotracer for the estrogen receptor, a historical review"]. Nucl. Med. Biol. 92: 24–37. February 2020. doi:10.1016/j.nucmedbio.2020.02.007. ISSN 0969-8051. PMID 32229068.
- ↑ "18F-Fluoroestradiol Positron Emission Tomography in Breast Cancer Patients: Systematic Review of the Literature & Meta-Analysis". Curr Radiopharm 9 (3): 244–257. 2016. doi:10.2174/1874471009666161019144950. PMID 27774910.
- ↑ "18F-Fluoroestradiol PET: Current Status and Potential Future Clinical Applications". J. Nucl. Med. 57 (8): 1269–75. August 2016. doi:10.2967/jnumed.116.175596. PMID 27307345.
- ↑ "18F-Fluoroestradiol". Semin Nucl Med 37 (6): 470–6. November 2007. doi:10.1053/j.semnuclmed.2007.08.003. PMID 17920354.
- ↑ "Cerianna: FDA-Approved Drugs". https://www.accessdata.fda.gov/scripts/cder/daf/index.cfm?event=overview.process&ApplNo=212155.
External links
- "Fluoroestradiol F-18". Drug Information Portal. U.S. National Library of Medicine. https://druginfo.nlm.nih.gov/drugportal/name/fluoroestradiol%20f-18.
- "Fluoroestradiol F-18". 4 May 2020. https://ncit.nci.nih.gov/ncitbrowser/ConceptReport.jsp?dictionary=NCI_Thesaurus&ns=ncit&code=C62541.
 |

