(diff) ← Older revision | Latest revision (diff) | Newer revision → (diff)
Short description
Vinbarbital Clinical data Routes of Oral ATC code Legal status Legal status
Identifiers
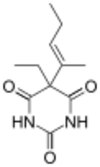
5-ethyl-5-[(1E )-1-methylbut-1-en-1-yl]pyrimidine-2,4,6(1H ,3H ,5H )-trione
CAS Number PubChem CID ChemSpider UNII KEGG ChEMBL Chemical and physical data Formula C 11 H 16 N 2 O 3 Molar mass −1 3D model (JSmol )
O=C1NC(=O)NC(=O)C1(/C(=C/CC)C)CC
InChI=1S/C11H16N2O3/c1-4-6-7(3)11(5-2)8(14)12-10(16)13-9(11)15/h6H,4-5H2,1-3H3,(H2,12,13,14,15,16)/b7-6+
Y Key:RAFOHKSPUDGZPR-VOTSOKGWSA-N
Y N Y (what is this?) (verify)
Vinbarbital is a hypnotic drug which is a barbiturate derivative.[1] [2]
References Alcohols Barbiturates Benzodiazepines Carbamates Flavonoids Imidazoles Kava constituentsMonoureides Neuroactive steroids Nonbenzodiazepines Phenols Piperidinediones Pyrazolopyridines Quinazolinones Volatiles /gases Others/unsorted
3-Hydroxybutanal α-EMTBL AA-29504 Avermectins (e.g., ivermectin )Bromide compounds (e.g., lithium bromide , potassium bromide , sodium bromide) Carbamazepine Chloralose Chlormezanone Clomethiazole DEABL Dihydroergolines (e.g., dihydroergocryptine , dihydroergosine , dihydroergotamine , ergoloid (dihydroergotoxine) )DS2 Efavirenz Etazepine Etifoxine Fenamates (e.g., flufenamic acid , mefenamic acid , niflumic acid , tolfenamic acid )Fluoxetine Flupirtine Hopantenic acid Lanthanum Lavender oil Lignans (e.g., 4-O-methylhonokiol , honokiol , magnolol , obovatol )Loreclezole Menthyl isovalerate (validolum) Monastrol Niacin Nicotinamide (niacinamide) Org 25,435 Phenytoin Propanidid Retigabine (ezogabine) Safranal Seproxetine Stiripentol Sulfonylalkanes (e.g., sulfonmethane (sulfonal) , tetronal , trional )Terpenoids (e.g., borneol )Topiramate Valerian constituents (e.g., isovaleric acid , isovaleramide , valerenic acid , valerenol )
Original source: https://en.wikipedia.org/wiki/Vinbarbital. Read more





 (0 votes)
(0 votes)