Chemistry:Delmadinone acetate
 | |
| Clinical data | |
|---|---|
| Trade names | Tardak, others |
| Other names | DMA; RS-1310; 1-Dehydrochlormadinone acetate; 1,6-Didehydro-6-chloro-17α-acetoxyprogesterone; 6-Chloro-17α-hydroxypregna-1,4,6-triene-3,20-dione |
| Drug class | Progestogen; Progestin; Progestogen ester; Steroidal antiandrogen |
| ATCvet code | |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| ChEMBL | |
| Chemical and physical data | |
| Formula | C23H27ClO4 |
| Molar mass | 402.92 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Delmadinone acetate (DMA), sold under the brand name Tardak among others, is a progestin and antiandrogen which is used in veterinary medicine to treat androgen-dependent conditions such as benign prostatic hyperplasia.[1][2][3][4] It must be used with care as it has the potential to cause adrenal insufficiency via inhibition of adrenocorticotropic hormone (ACTH) secretion from the pituitary gland.[5] DMA is the C17α acetate ester of delmadinone, which, in contrast to DMA, was never marketed for medical use.[1][2]
Uses
Veterinary
DMA is used to treat androgen-dependent conditions in animals.[6] It is most commonly used to treat benign prostatic hyperplasia.[6] However, it can also be used to treat hypersexuality in male dogs and cats, perianal gland tumors in dogs, and hormone-driven aggression in dogs.[6]
Pharmacology
Pharmacodynamics
DMA is a progestogen with antigonadotropic and hence antiandrogenic and antiestrogenic effects. In addition, DMA binds to the androgen receptor, and likely acts as an antagonist of this receptor similarly to related drugs like chlormadinone acetate and osaterone acetate.[7]
Chemistry
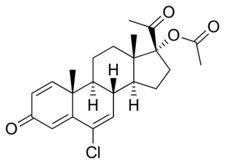
DMA, also known as 1-dehydrochlormadinone acetate, as well as 1,6-didehydro-6-chloro-17α-acetoxyprogesterone or '6-chloro-17α-hydroxypregna-1,4,6-triene-3,20-dione, is a synthetic pregnane steroid and a derivative of progesterone.[1][2][4] It is specifically a derivative of 17α-hydroxyprogesterone with a chlorine atom at the C6 position, a double bond between the C1 and C2 positions, another double bond between the C6 and C7 positions, and an acetate ester at the C17α position.[1][2] Analogues of DMA include other 17α-hydroxyprogesterone derivatives such as chlormadinone acetate, cyproterone acetate, hydroxyprogesterone caproate, medroxyprogesterone acetate, megestrol acetate, and osaterone acetate.[1][2]
History
DMA was first described in the literature in 1959 and has been marketed since at least 1972.[1][8][9] It was marketed by this year in Europe and the United Kingdom under the brand names Tardak and Zenadrex.[9] It was under development for use in the United States as well but does not seem to have ultimately been marketed in this country.[9]
Society and culture
Generic names
Delmadinone acetate is the generic name of the drug and its USAN and BANM.[1][2][3][4] Delmadinone is the INN and BAN of the unesterified free alcohol form.[1][2][3][4]
Brand names
DMA is most commonly sold as Tardak, but has also been marketed under a variety of other brand names including Delmate, Estrex, Tardastren, Tardastrex, Vetadinon, and Zenadrex.[1][2][4]
Availability
DMA is available in Europe and Oceania.[2][4] It is specifically marketed in the United Kingdom , France , Belgium, Germany , Austria, Switzerland , the Netherlands, Finland , Australia , and New Zealand.[2][4]
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 1.6 1.7 1.8 The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. Springer. 14 November 2014. pp. 354–. ISBN 978-1-4757-2085-3. https://books.google.com/books?id=0vXTBwAAQBAJ&pg=PA354.
- ↑ 2.0 2.1 2.2 2.3 2.4 2.5 2.6 2.7 2.8 2.9 Index Nominum 2000: International Drug Directory. Taylor & Francis US. 2000. p. 298. ISBN 978-3-88763-075-1. https://books.google.com/books?id=5GpcTQD_L2oC&pg=PA298. Retrieved 30 May 2012.
- ↑ 3.0 3.1 3.2 Concise Dictionary of Pharmacological Agents: Properties and Synonyms. Springer. 1999. p. 92. ISBN 978-0-7514-0499-9. https://books.google.com/books?id=mqaOMOtk61IC&pg=PA92. Retrieved 30 May 2012.
- ↑ 4.0 4.1 4.2 4.3 4.4 4.5 4.6 "List of Progestins". Drugs.com. https://www.drugs.com/international/delmadinone.html.
- ↑ "Effects of delmadinone acetate on pituitary-adrenal function, glucose tolerance and growth hormone in male dogs". Australian Veterinary Journal 76 (8): 555–560. August 1998. doi:10.1111/j.1751-0813.1998.tb10216.x. PMID 9741725. https://rvc-repository.worktribe.com/preview/1659487/kura-et-al-2023-can-mass-drug-administration-of-moxidectin-accelerate-onchocerciasis-elimination-in-africa.pdf.
- ↑ 6.0 6.1 6.2 "Update on medical management of benign prostatic hyperplasia". Companion Animal 13 (7): 39–41. 2008. doi:10.1111/j.2044-3862.2008.tb00313.x. ISSN 1464-4630.
- ↑ "Structure-activity relationships of synthetic progestins in a yeast-based in vitro androgen bioassay". The Journal of Steroid Biochemistry and Molecular Biology 110 (1–2): 39–47. May 2008. doi:10.1016/j.jsbmb.2007.10.008. PMID 18395441.
- ↑ "Steroids. Cxxvii.16-Halo Progestational Agents". Journal of the American Chemical Society 81 (13): 3485–3486. 1959. doi:10.1021/ja01522a090. ISSN 0002-7863.
- ↑ 9.0 9.1 9.2 Modern Veterinary Practice. 53. 1972. p. 46. https://books.google.com/books?id=F-8OAQAAMAAJ. "John T. Bryans, University of Kentucky, Lexington. Thoroughbred Record 194(22): 1634-1636, 1971. In the clinical review entitled "Antiandrogen Treatment of Prostate Disorders" (MVP Oct, p 46} the product should have been identified as delta-chlor- madinone acetate (delta-CAP, rather than as CAP). This compound, also known as delmadinone acetate, has unique properties and is marketed in Europe and the UK as Tardak® and Zenadrex®; It is presently subject to clinical investigation in the US for FDA approval."
 |

