Chemistry:Etonogestrel
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Circlet, Implanon, Nexplanon, others |
| AHFS/Drugs.com | Professional Drug Facts |
| MedlinePlus | a604032 |
| Pregnancy category |
|
| Routes of administration | Subcutaneous implant, vaginal ring |
| Drug class | Progestogen; Progestin |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | Implant: 100%[3] Vaginal ring: 100%[4] |
| Protein binding | ≥98% (66% to albumin, 32% to SHBG)[3] |
| Metabolism | Liver (CYP3A4)[3][4] |
| Elimination half-life | 21–38 hours[5][6][3][4] |
| Excretion | Urine (major), feces (minor)[3][4] |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| Chemical and physical data | |
| Formula | C22H28O2 |
| Molar mass | 324.464 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
| Etonogestrel birth control implant | |
|---|---|
 Implanon | |
| Background | |
| Type | Hormonal Progestin-only implant |
| First use | 1998 |
| Synonyms | Etonogestrel contraceptive implant |
| Trade names | Implanon, Nexplanon, others |
| AHFS/Drugs.com | FDA Professional Drug Information |
| Failure rates (first year) | |
| Perfect use | 0.05%[7] |
| Typical use | 0.05%[7] |
| Usage | |
| Duration effect | 3 to 5 years[8][9] |
| Reversibility | Yes |
| User reminders | Requires removal after the 3–5 years[10] |
| Advantages and disadvantages | |
| STI protection | No |
| Weight | May cause weight gain |
| Period disadvantages | May cause irregular or prolonged bleeding |
| Period advantages | Minimizes pain. In 33% no periods. |
| Benefits | Long-term contraception. |
Etonogestrel is a medication which is used as a means of birth control for women.[3][4][11][12] It is available as an implant placed under the skin of the upper arm under the brand names Nexplanon and Implanon. It is a progestin that is also used in combination with ethinylestradiol, an estrogen, as a vaginal ring under the brand names NuvaRing and Circlet.[13] Etonogestrel is effective as a means of birth control and lasts at least three or four years with some data showing effectiveness for five years.[8][10] Following removal, fertility quickly returns.[14]
Side effects of etonogestrel include menstrual irregularities, breast tenderness, mood changes, acne, headaches, vaginitis, and others.[3] Etonogestrel is a progestin, or a synthetic progestogen, and hence is an agonist of the progesterone receptor, the biological target of progestogens like progesterone.[15] It works by stopping ovulation, thickening the mucus around the opening of the cervix, and altering the lining of the uterus.[16] It has very weak androgenic and glucocorticoid activity and no other important hormonal activity.[15]
Etonogestrel was patented in 1972 and introduced for medical use in 1998.[17][18][19] It became available in the United States in 2006.[17][18] Etonogestrel implants are approved in more than 90 countries and used by about three million women globally as of 2010.[16][20]
A closely related and more widely known and used progestin, desogestrel, is a prodrug of etonogestrel in the body.[15]
Medical uses
Etonogestrel is used in hormonal contraception in form of the etonogestrel contraceptive implant[3] and the contraceptive vaginal ring (brand names NuvaRing, Circlet), the latter in combination with ethinylestradiol.[4]
Etonogestrel birth control implants are a type of long-acting reversible contraception, which has been shown to be one of the most effective form of birth control.[21] The failure rate of the implants is 0.05% for both perfect use and typical use because the method requires no user action after placement.[22] Studies of one type, which include over 2,467 women-years of exposure, found no pregnancies.[23][24][25]
Other studies have found some failures with this method, some attributed to failures of the method itself and others to improper placement, drug interactions, or conception prior to method insertion.[26]
In comparison, tubal sterilization has a failure rate of 0.5% and IUDs have a failure rate of 0.2–0.8%.[22] A single implant is approved for three years with data showing effectiveness for five years.[27][10]
Contraindications
Women should not use implants if they:[28]
- are, or think they are, pregnant
- are allergic to etonogestrel
- have vaginal bleeding that has not been explained
- have some forms of severe liver disease.
Women should not use combined hormone contraceptives (CHC) if they have migraines with auras.[29]
A full list of contraindications can be found in the WHO Medical Eligibility Criteria for Contraceptive Use 2015 and the CDC United States Medical Eligibility Criteria for Contraceptive Use 2016.
Side effects
Irregular bleeding and spotting: Many women will experience some type of irregular, unpredictable, prolonged, frequent, or infrequent bleeding.[30] Some women also experience amenorrhea. For some women, prolonged bleeding will decline after the first three months of use. However, other women may experience this bleeding pattern through all five years of use. While these patterns are not dangerous, they are the most common reason that women give for discontinuing the use of the implant. After removal, bleeding patterns return to previous patterns in most women.[23][24][25]
Insertion complications: Some minor side effects such as bruising, skin irritation, or pain around the insertion site are common.[23] However, there are some rare complications that can occur, such as infection or expulsion.[23][31] In some cases, a serious complication occurs when the provider fails to insert, and the rod is left in the inserter. An Australian study reported 84 pregnancies as a result of such failure.[26]
Migration: Although very rare, the rod can sometimes move slightly within the arm. This can make removal more difficult. It is possible that insertion in the same site as a previous implant increases the likelihood of migration.[31] Rods can be located only through high-frequency ultrasound or magnetic resonance imaging (MRI).[23] It can be located using traditional X-ray or CT-scan because of the inclusion of barium sulphate. There have been rare reports of implants having reached the lung via the pulmonary artery.[32] Correct subdermal insertion over the triceps muscle reduces the risk of these events.
Possible weight gain: Some women may experience slight weight gain when using the implant.[23] However, current studies are not conclusive because they do not compare the weight of women using implants with a control group of women not using the implant. The average increase in body weight in studies was less than 5 pounds (2,25 kg) over 2 years.[24]
Ovarian cysts: A small portion of women using implants and other contraceptive implants develop ovarian cysts.[23] Usually these cysts will disappear without treatment.[33]
Pregnancy: it is recommended that implants be removed if a pregnancy does occur. However, there is no evidence to suggest that the implant has a negative effect on pregnancy or a developing fetus.[23]
Acne: Acne has been self-reported to be a side effect, and is listed as a side effect by the FDA. However, a study of users found that a majority of users with acne before their insertion reported that their acne had decreased, and only 16% of those who did not have acne before insertion developed acne.[24]
Other possible symptoms: Other symptoms that have been reported in trials of implants include headache, emotional lability, depression, abdominal pain, loss of libido, and vaginal dryness.[23] However, there have been no studies that conclusively determine that these symptoms are caused by the implant.[24][25]
Overdose
No serious side effects are expected when overdosing contraceptives in general.[34]
Interactions
Efavirenz, an inducer of the liver enzyme CYP3A4, appears to decrease etonogestrel levels[35] and increase rates of undesired pregnancy among implant users.
Similar effects are expected for other CYP3A4 inducers, but it is not known whether these are clinically relevant. The opposite is true of CYP3A4 inhibitors such as ketoconazole, itraconazole and clarithromycin: they might increase etonogestrel concentrations in the body.[34]
Device description
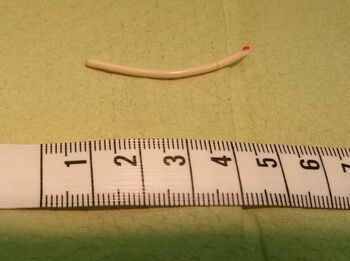
Nexplanon/Implanon consists of a single rod made of ethylene vinylacetate copolymer that is 4 cm long and 2 mm in diameter.[30] It is similar to a matchstick in size. The rod contains 68 mg of etonogestrel (sometimes called 3-keto-destrogestrel), a type of progestin.[23] Peak serum etonogestrel concentrations have been found to reach 781–894 pg/mL in the first few weeks, gradually decreasing to 192–261 pg/mL after one year, 154–194 pg/mL after two years, and 156–177 pg/mL after three years, maintaining ovulation suppression and contraceptive efficacy.[36] Serum levels maintain relatively stable through 36 months, which implies that the method may be effective for longer than three years.[37]
Although not formally approved by the manufacturer for more than three years, studies have shown it remains a highly effective contraceptive for five years.[27]
It is a type of progestogen-only contraception.
Insertion and removal
File:Implanon implantation.webm An experienced clinician must perform the insertion of implants to ensure proper insertion and minimize the risk of nerve damage or misplacement, which could result in pregnancy.[38] Before insertion, the arm is washed with a cleaning solution and a local anesthetic is applied to the upper arm around the insertion area.[23] A needle-like applicator is used to insert the rod under the skin into the subdermal tissue on the inner side of the arm posterior to the groove between the biceps and triceps muscles.[39] The average time for insertion is 0.5 to 1 minute.[24][25] A bandage should be kept on the insertion site for 24 hours afterwards. Bruising and mild discomfort are common after insertion.[23] Serious insertion site complications such as infection can occur very rarely, in less than 1% of patients. If a woman receives an implant outside the first five days of her period, she should wait to have sex or use a backup method of contraception (such as a condom, female condom, diaphragm, sponge, or emergency contraception) for the following week after insertion to prevent pregnancy. However, if the implant is inserted during the first five days of a woman's period, she is protected for that cycle and beyond.[40] File:Removal Implanon.webm Implants can be removed at any time if pregnancy is desired. The rod must also be removed by an experienced clinician. At removal, a local anesthetic is again used around the implant area at the distal end.[23] If the provider cannot feel the implant, imaging tests may be necessary to locate the rod before it can be removed. A small incision is made in the skin over the end of the implant site. In some cases, a fibrous sheath may have formed around the implant, in which case the sheath must be incised.[23] The implant is removed using forceps. The removal procedure lasts, on average, 3 to 3.5 minutes.[24][25]
Fertility after removal
Within a week of removal, the hormones from the device leave the body and etonogestrel is undetectable in most users.[23] Most women will begin to ovulate within six weeks of removal.[37][41] Fertility levels will return to what they were before implant insertion.[14]
Differences
Nexplanon and Implanon NXT are essentially identical to Implanon except Nexplanon and Implanon NXT have 15 mg of barium sulphate added to the core, so it is detectable by x-ray.[42][27] Nexplanon and Implanon NXT also has a pre-loaded applicator for easier insertion.[43]
Pharmacology
The mechanism of action of progestin-only contraceptives depends on the progestin activity and dose.[44] Intermediate dose progestin-only contraceptives like Nexplanon or Implanon allow some follicular development but inhibit ovulation in almost all cycles as the primary mechanism of action. Ovulation was not observed in studies of Implanon in the first two years of use and only rarely in the third year with no pregnancies. A secondary mechanism of action is the progestogenic increase in cervical mucus viscosity which inhibits sperm penetration.[45] Hormonal contraceptives also have effects on the endometrium that theoretically could affect implantation, however no scientific evidence indicates that prevention of implantation actually results from their use.[46]
Pharmacodynamics
Etonogestrel is a progestogen, or an agonist of the progesterone receptor.[15] It is less androgenic than levonorgestrel and norethisterone,[47][48] and it does not cause a decrease in sex hormone-binding globulin levels.[49] However, it is still associated with acne in up to 13.5% of patients when used as an implant, though this side effect only accounts for 1.3% of premature removals of the implant.[50] In addition to its progestogenic and weak androgenic activity, etonogestrel binds to the glucocorticoid receptor with about 14% of the affinity of dexamethasone (relative to 1% for levonorgestrel) and has very weak glucocorticoid activity.[15] Etonogestrel has no other hormonal activity (e.g., estrogenic, antimineralocorticoid).[15] Some inhibition of 5α-reductase and hepatic cytochrome P450 enzymes has been observed with etonogestrel in vitro, similarly to other 19-nortestosterone progestins.[15]
| Compound | PR | AR | ER | GR | MR | SHBG | CBG |
|---|---|---|---|---|---|---|---|
| Etonogestrel | 150 | 20 | 0 | 14 | 0 | 15 | 0 |
| 5α-Dihydroetonogestrel | 9 | 17 | 0 | ? | ? | ? | ? |
Pharmacokinetics
The bioavailability of etonogestrel when given as a subcutaneous implant or as a vaginal ring is 100%.[3][4] Steady-state levels of etonogestrel are achieved within one week upon insertion as an implant or vaginal ring.[3][4] The mean volume of distribution of etonogestrel is 201 L.[3] The plasma protein binding of the medication is at least 98%, with 66% bound to albumin and 32% bound to sex hormone-binding globulin.[3][4] Etonogestrel is metabolized in the liver by CYP3A4.[3][4] The biological activity of its metabolites is unknown.[3][4] The elimination half-life of etonogestrel is about 25 to 29 hours.[3][4] Following removal of an etonogestrel-containing implant, levels of the medication were below the limits of assay detection by one week.[3] The major portion of etonogestrel is eliminated in urine and a minor portion is eliminated in feces.[3][4]
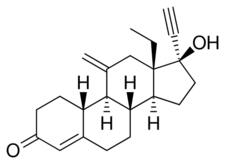
Chemistry
Etonogestrel, also known as 11-methylene-17α-ethynyl-18-methyl-19-nortestosterone or as 11-methylene-17α-ethynyl-18-methylestr-4-en-17β-ol-3-one, is a synthetic estrane steroid and a derivative of testosterone.[11][13] It is more specifically a derivative of norethisterone (17α-ethynyl-19-nortestosterone) and is a member of the gonane (18-methylestrane) subgroup of the 19-nortestosterone family of progestins.[51][52] Etonogestrel is the C3 ketone derivative of desogestrel and the C11 methylene derivative of levonorgestrel and is also known as 3-ketodesogestrel and as 11-methylenelevonorgestrel.[53]
History
The possibility of the subdermal contraceptive implant began when silicone was discovered in the 1940s and found to be bio-compatible with the human body.[54] In 1964, Folkman and Long published the first study demonstrating that such a rod could be used to deliver drugs.[55] In 1966 Dziuk and Cook published a study that looked at release rates and suggested that the rods could be well suited for contraception.[56] After a study that used implants with progestogens for contraception, the Population Council developed and patented Norplant and Jadelle.[57] Norplant has six rods and is considered a first-generation implant. Jadelle (Norplant II), a two-rod implant, and other single rod implants that followed, were developed because of complications resulting from Norplant's six-rod system. The Jadelle system contains two silicone rods mixed with levonorgestrel. In 1990 De Nijs patented a co-axial extrusion technique of ethylene vinylacetate copolymers and 3-keto-desogestrel (etonogestrel) for the preparation of long-acting contraceptive devices, such as Implanon, Nexplanon and Nuvaring.[58] The single rods were less visible under the skin and used etonogestrel as opposed to levonorgestrel in the hopes that it would reduce side effects.[54]
Desogestrel (3-deketoetonogestrel), a prodrug of etonogestrel, was introduced for medical use in 1981.[5][59]
Norplant was used internationally beginning in 1983 and was marketed in the United States and the United Kingdom in 1993. There were many complications associated with Norplant removal in the United States and it was taken off the market in 2002. Although Jadelle was approved by the FDA, it has never been marketed in the United States, but it is widely used in Africa and Asia.[57]
Etonogestrel itself was first introduced as Implanon in Indonesia in 1998,[17][18] was marketed in the United Kingdom shortly thereafter,[60] and approved for use in the United States in 2006.[17][18] Nexplanon was developed to eliminate the problem of non-insertion and localization of Implanon by changing the inserter device and making the rod radiopaque.[42] As of January 2012, Implanon is no longer being marketed and Nexplanon is the only available single-rod implant.
Society and culture
Generic names
Etonogestrel is the generic name of the drug and its INN, USAN, and BAN.[11][13] It is also known by its developmental code name ORG-3236.[11][13]
Brand names
Etonogestrel is marketed under the brand names Circlet, Implanon, Nexplanon, and NuvaRing.[11][13]
Availability
Etonogestrel is available widely throughout the world, including in the United States, Canada, the United Kingdom, Ireland, elsewhere throughout Europe, South Africa, Latin America, South, East, and Southeast Asia, and elsewhere in the world.[13]
Research
An etonogestrel-releasing intrauterine device was under development for use as a form of birth control for women but development was discontinued in 2015.[61]
Etonogestrel has been studied for use as a potential male contraceptive.[62]
See also
- Ethinylestradiol/etonogestrel
References
- ↑ "NEXPLANON : Etonogestrel extended release subdermal implant". https://pdf.hres.ca/dpd_pm/00060728.PDF.
- ↑ "List of nationally authorised medicinal products : Active substance: etonogestrel : Procedure no.: PSUSA/00001331/202109". https://www.ema.europa.eu/documents/psusa/etonogestrel-list-nationally-authorised-medicinal-products-psusa/00001331/202109_en.pdf.
- ↑ 3.00 3.01 3.02 3.03 3.04 3.05 3.06 3.07 3.08 3.09 3.10 3.11 3.12 3.13 3.14 3.15 3.16 "Nexplanon- etonogestrel implant". 18 November 2019. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=b03a3917-9a65-45c2-bbbb-871da858ef34.
- ↑ 4.00 4.01 4.02 4.03 4.04 4.05 4.06 4.07 4.08 4.09 4.10 4.11 4.12 "NuvaRing- etonogestrel and ethinyl estradiol insert, extended release". 24 January 2020. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=017343fb-86c4-45ab-9c47-52cc5b9f3a02.
- ↑ 5.0 5.1 Female Contraception: Update and Trends. Springer Science & Business Media. 6 December 2012. pp. 156–163. ISBN 978-3-642-73790-9. https://books.google.com/books?id=LtT6CAAAQBAJ&pg=PA156.
- ↑ Mosby's GenRx: A Comprehensive Reference for Generic and Brand Prescription Drugs. Mosby. 2001. p. 687. ISBN 978-0-323-00629-3. https://books.google.com/books?id=QxsobYYgm8oC. "The elimination half-life for 3-keto-desogestrel is approximately 38 ± 20 hours at steady state."
- ↑ 7.0 7.1 "Contraceptive efficacy". Contraceptive technology (20th revised ed.). New York: Ardent Media. 2011. pp. 779–863. ISBN 978-1-59708-004-0. OCLC 781956734. http://www.contraceptivetechnology.org/wp-content/uploads/2013/09/CTFailureTable.pdf.
- ↑ 8.0 8.1 Tarascon Pocket Pharmacopoeia 2016 Deluxe Lab-Coat Edition. Jones & Bartlett Publishers. 2016. p. 392. ISBN 9781284095289. https://books.google.com/books?id=YISkDAAAQBAJ&pg=PA392. Retrieved 2021-02-18.
- ↑ Sexual and Reproductive Health at a Glance. John Wiley & Sons. 2015. p. 21. ISBN 9781118460757. https://books.google.com/books?id=5r6SCgAAQBAJ&pg=PA21. Retrieved 2021-02-18.
- ↑ 10.0 10.1 10.2 (in en) Contraception, An Issue of Obstetrics and Gynecology Clinics, E-Book. Elsevier Health Sciences. 2016. p. 634. ISBN 9780323402590. https://books.google.com/books?id=gtqZCwAAQBAJ&pg=PA634. Retrieved 2021-02-18.
- ↑ 11.0 11.1 11.2 11.3 11.4 Index Nominum 2000: International Drug Directory. Taylor & Francis. January 2000. pp. 420. ISBN 978-3-88763-075-1. https://books.google.com/books?id=5GpcTQD_L2oC&pg=PA420. Retrieved 2018-02-21.
- ↑ Foye's Principles of Medicinal Chemistry. Lippincott Williams & Wilkins. 24 January 2012. pp. 1409–. ISBN 978-1-60913-345-0. https://books.google.com/books?id=Sd6ot9ul-bUC&pg=PA1409. Retrieved 11 October 2016.
- ↑ 13.0 13.1 13.2 13.3 13.4 13.5 "Etonogestrel". https://www.drugs.com/international/etonogestrel.html.
- ↑ 14.0 14.1 The selection and use of essential medicines. Twentieth report of the WHO Expert Committee 2015 (including 19th WHO Model List of Essential Medicines and 5th WHO Model List of Essential Medicines for Children). Geneva: World Health Organization. 2015. pp. 332–36. WHO technical report series;994. ISBN 9789241209946.
- ↑ 15.0 15.1 15.2 15.3 15.4 15.5 15.6 "Pharmacology of estrogens and progestogens: influence of different routes of administration". Climacteric 8 (Suppl 1): 3–63. 2005. doi:10.1080/13697130500148875. PMID 16112947. http://hormonebalance.org/images/documents/Kuhl%2005%20%20Pharm%20Estro%20Progest%20Climacteric_1313155660.pdf. Retrieved 2018-02-21.
- ↑ 16.0 16.1 Oxford Handbook of Genitourinary Medicine, HIV, and Sexual Health. OUP Oxford. 2010. p. 368. ISBN 9780199571666. https://books.google.com/books?id=wuZIChOf3A8C&pg=PA368.
- ↑ 17.0 17.1 17.2 17.3 Advanced Health Assessment of Women, Third Edition: Clinical Skills and Procedures. Springer Publishing Company. 10 October 2014. pp. 411–. ISBN 978-0-8261-2308-4. https://books.google.com/books?id=P1uFBQAAQBAJ&pg=PA411. Retrieved 11 October 2016.
- ↑ 18.0 18.1 18.2 18.3 The Essential Guide to Primary Care Procedures. Lippincott Williams & Wilkins. 28 March 2012. pp. 589–. ISBN 978-1-4511-5286-9. https://books.google.com/books?id=d-wNb7eyonMC&pg=PA589. Retrieved 11 October 2016.
- ↑ (in en) Analogue-based Drug Discovery. John Wiley & Sons. 2006. p. 480. ISBN 9783527607495. https://books.google.com/books?id=FjKfqkaKkAAC&pg=PA480. Retrieved 2020-06-06.
- ↑ Atlas of Contraception, Second Edition (2 ed.). CRC Press. 2008. p. 53. ISBN 9780203347324. https://books.google.com/books?id=7dDKBQAAQBAJ&pg=PA53.
- ↑ "Effectiveness of long-acting reversible contraception". The New England Journal of Medicine 366 (21): 1998–2007. May 2012. doi:10.1056/nejmoa1110855. PMID 22621627. https://digitalcommons.wustl.edu/cgi/viewcontent.cgi?article=3776&context=open_access_pubs. Retrieved 2021-02-18.
- ↑ 22.0 22.1 Guttmacher (2012). "Contraceptive Use in the United States". https://www.guttmacher.org/fact-sheet/contraceptive-use-united-states.
- ↑ 23.00 23.01 23.02 23.03 23.04 23.05 23.06 23.07 23.08 23.09 23.10 23.11 23.12 23.13 23.14 "Contraceptive Implants". Contraceptive technology (19th revised ed.). New York: Ardent Media. 2011. pp. 144–156.
- ↑ 24.0 24.1 24.2 24.3 24.4 24.5 24.6 "Safety and efficacy of Implanon, a single-rod implantable contraceptive containing etonogestrel". Contraception 71 (5): 319–26. May 2005. doi:10.1016/j.contraception.2004.11.007. PMID 15854630.
- ↑ 25.0 25.1 25.2 25.3 25.4 "Clinical experience and acceptability of the etonogestrel subdermal contraceptive implant". International Journal of Gynaecology and Obstetrics 90 (3): 228–33. September 2005. doi:10.1016/j.ijgo.2005.06.007. PMID 16043175.
- ↑ 26.0 26.1 "Unintended pregnancies with the etonogestrel implant (Implanon): a case series from postmarketing experience in Australia". Contraception 71 (4): 306–8. April 2005. doi:10.1016/j.contraception.2004.10.005. PMID 15792651.
- ↑ 27.0 27.1 27.2 Contraceptive technology. Hatcher, Robert A. (Robert Anthony), 1937- (21st ed.). New York, NY. September 2018. pp. Chapter 4, specifically pages 129–134. ISBN 978-1732055605. OCLC 1048947218.
- ↑ "US CDC Medical Eligibility Criteria for Contraceptive Use". 2016. https://www.cdc.gov/reproductivehealth/contraception/mmwr/mec/appendixc_tableC1.html#mec_personal.
- ↑ "Hormonal contraception in women with migraine: is progestogen-only contraception a better choice?". The Journal of Headache and Pain 14 (1): 66. August 2013. doi:10.1186/1129-2377-14-66. PMID 24456509.
- ↑ 30.0 30.1 "Implanon: a review of the literature with recommendations for clinical management". Journal of Midwifery & Women's Health 54 (2): 142–9. 2009. doi:10.1016/j.jmwh.2008.09.004. PMID 19249660.
- ↑ 31.0 31.1 "An assessment of the use of Implanon in three community services". The Journal of Family Planning and Reproductive Health Care 28 (4): 193–6. October 2002. doi:10.1783/147118902101196540. PMID 12419059.
- ↑ "Nexplanon (etonogestrel) contraceptive implants: Reports of device in vasculature and lung". https://www.gov.uk/drug-safety-update/nexplanon-etonogestrel-contraceptive-implants-reports-of-device-in-vasculature-and-lung.
- ↑ "Nonmenstrual adverse events during use of implantable contraceptives for women: data from clinical trials". Contraception 65 (1): 63–74. January 2002. doi:10.1016/s0010-7824(01)00289-x. PMID 11861056.
- ↑ 34.0 34.1 (in German) Austria-Codex. Vienna: Österreichischer Apothekerverlag. 2020. Implanon NXT 68 mg Implantat zur subkutanen Anwendung.
- ↑ "Effect of antiretroviral therapy including lopinavir/ritonavir or efavirenz on etonogestrel-releasing implant pharmacokinetics in HIV-positive women". Journal of Acquired Immune Deficiency Syndromes 66 (4): 378–385. August 2014. doi:10.1097/QAI.0000000000000189. PMID 24798768.
- ↑ "Implanon label" (PDF). FDA. 2010-10-26. http://dailymed.nlm.nih.gov/dailymed/fda/fdaDrugXsl.cfm?id=9311&type=display.
- ↑ 37.0 37.1 "Ovarian function during the use of a single contraceptive implant: Implanon compared with Norplant". Fertility and Sterility 69 (4): 714–21. April 1998. doi:10.1016/s0015-0282(98)00015-6. PMID 9548163.
- ↑ "Nerve injury caused by removal of an implantable hormonal contraceptive". American Journal of Obstetrics and Gynecology 195 (1): 323–6. July 2006. doi:10.1016/j.ajog.2005.09.016. PMID 16813761.
- ↑ "Nexplanon Prescribing Information". https://www.merck.com/product/usa/pi_circulars/n/nexplanon/nexplanon_pi.pdf.
- ↑ Bedsider (2010). "Implant." Retrieved from http://bedsider.org/methods/implant#how_to_tab on March 17, 2011.
- ↑ "Release characteristics, ovarian activity and menstrual bleeding pattern with a single contraceptive implant releasing 3-ketodesogestrel". Contraception 47 (3): 251–61. March 1993. doi:10.1016/0010-7824(93)90042-6. PMID 8462316.
- ↑ 42.0 42.1 "Nexplanon: what Implanon did next". The Journal of Family Planning and Reproductive Health Care 36 (4): 187–9. October 2010. doi:10.1783/147118910793048629. PMID 21067632.
- ↑ "Contraceptive alert after women fall pregnant". Reuters. 5 Jan 2011. https://www.reuters.com/article/britain-contraceptive-idUSLDE7040T320110105.
- ↑ "Contraception". Endocrinology (5th ed.). Philadelphia: Elsevier Saunders. 2006. pp. 3000–1. ISBN 978-0-7216-0376-6. https://archive.org/details/endocrinology0003unse/page/3000.
- ↑ Organon (April 2006). "Implanon SPC (Summary of Product Characteristics)". http://emc.medicines.org.uk/emc/assets/c/html/displaydoc.asp?documentid=5382.
- ↑ "The mechanism of action of hormonal contraceptives and intrauterine contraceptive devices". American Journal of Obstetrics and Gynecology 181 (5 Pt 1): 1263–9. November 1999. doi:10.1016/S0002-9378(99)70120-1. PMID 10561657.
- ↑ Acne: Causes and Practical Management. John Wiley & Sons. 27 January 2015. pp. 77–. ISBN 978-1-118-23277-4. https://books.google.com/books?id=Z1yFBQAAQBAJ&pg=PA77. Retrieved 11 October 2016.
- ↑ Principles of Pharmacology: The Pathophysiologic Basis of Drug Therapy. Lippincott Williams & Wilkins. 2008. pp. 521–. ISBN 978-0-7817-8355-2. https://books.google.com/books?id=az8uSDkB0mgC&pg=PA521. Retrieved 2016-10-11.
- ↑ A Clinical Guide for Contraception. Lippincott Williams & Wilkins. 22 November 2010. pp. 365–. ISBN 978-1-60831-610-6. https://books.google.com/books?id=f5XJtYkiJ0YC&pg=PT365. Retrieved 11 October 2016.
- ↑ Comprehensive Gynecology. Elsevier Health Sciences. 21 February 2012. pp. 256–. ISBN 978-0-323-09131-2. https://books.google.com/books?id=X5KT_w6Nye8C&pg=PA256. Retrieved 11 October 2016.
- ↑ Pharmacology for Women's Health. Jones & Bartlett Publishers. 8 September 2015. pp. 368–. ISBN 978-1-284-05748-5. https://books.google.com/books?id=AniUCgAAQBAJ&pg=PA368. Retrieved 3 August 2017.
- ↑ The Handbook of Contraception: A Guide for Practical Management. Springer Science & Business Media. 7 November 2007. pp. 16–. ISBN 978-1-59745-150-5. https://books.google.com/books?id=sczb0Tk_2IwC&pg=PA16. Retrieved 3 August 2017.
- ↑ Cite error: Invalid
<ref>tag; no text was provided for refs namedRyan1999 - ↑ 54.0 54.1 "Contraceptive implants". African Journal of Reproductive Health 9 (1): 16–23. April 2005. doi:10.2307/3583156. PMID 16104651.
- ↑ "The use of silicone rubber as a carrier for prolonged drug therapy". The Journal of Surgical Research 4 (3): 139–42. March 1964. doi:10.1016/s0022-4804(64)80040-8. PMID 14130164.
- ↑ "Passage of steroids through silicone rubber". Endocrinology 78 (1): 208–11. January 1966. doi:10.1210/endo-78-1-208. PMID 5948426.
- ↑ 57.0 57.1 Association of Reproductive Health Professionals (July 2008). "The Single-Rod Contraceptive Implant". http://www.arhp.org/publications-and-resources/clinical-proceedings/Single-Rod/History.
- ↑ De Nijs H, "Contraceptive Implant", US patent granted 4957119, issued 18 September 1990, assigned to Akzo NV
- ↑ Progress Towards the Total Synthesis of Desogestrel and the Development of a New Chiral Dihydroimidazol-2-ylidene Ligand. University of Michigan.. 2007. p. 25. https://books.google.com/books?id=-lUeAQAAMAAJ. "In 1981, desogestrel was marketed as a new low dose oral contraceptive under the trade names Marvelon and Desogen.32"
- ↑ Contraception. Health Press. December 1999. p. 41. ISBN 978-1-899541-18-8. https://books.google.com/books?id=QYPiaMbHVCsC. Retrieved 2016-10-11.
- ↑ "Etonogestrel-releasing intrauterine system - Merck & Co.". http://adisinsight.springer.com/drugs/800032917.
- ↑ "Clinical trials in male hormonal contraception". Contraception 82 (5): 457–70. 2010. doi:10.1016/j.contraception.2010.03.020. PMID 20933120. http://www.kup.at/kup/pdf/10172.pdf. Retrieved 2020-09-05.
Further reading
- "The pharmacokinetics and pharmacodynamics of Implanon, a single-rod etonogestrel contraceptive implant". Eur J Contracept Reprod Health Care 5 (Suppl 2): 12–20. September 2000. doi:10.1080/14730782.2000.12288981. PMID 11246602.
- "Endometrial effects of etonogestrel (Implanon) contraceptive implant". Curr. Opin. Obstet. Gynecol. 13 (3): 335–41. June 2001. doi:10.1097/00001703-200106000-00015. PMID 11396660.
- "Etonogestrel implant (Implanon) for contraception". Drug Ther Bull 39 (8): 57–9. August 2001. PMID 11526801.
- "Medical eligibility criteria for new contraceptive methods: combined hormonal patch, combined hormonal vaginal ring and the etonogestrel implant". Contraception 73 (2): 134–44. February 2006. doi:10.1016/j.contraception.2005.08.002. PMID 16413844.
- "The combined etonogestrel/ethinyl estradiol contraceptive vaginal ring". Expert Opin Pharmacother 8 (11): 1769–77. August 2007. doi:10.1517/14656566.8.11.1769. PMID 17685892.
- "The management of unacceptable bleeding patterns in etonogestrel-releasing contraceptive implant users". Contraception 83 (3): 202–10. March 2011. doi:10.1016/j.contraception.2010.08.001. PMID 21310280.
- "In brief: etonogestrel (nexplanon) contraceptive implant". The Medical Letter on Drugs and Therapeutics 54 (1383): 12. February 2012. PMID 22354222.
- "Association of Etonogestrel-Releasing Contraceptive Implant with Reduced Weight Gain in an Exclusively Breastfed Infant: Report and Literature Review". Breastfeed Med 11 (4): 203–6. May 2016. doi:10.1089/bfm.2016.0017. PMID 27032034.
- "Efficacy and side-effects profile of the ethinylestradiol and etonogestrel contraceptive vaginal ring: a systematic review and meta-analysis". Eur J Contracept Reprod Health Care 22 (2): 131–146. April 2017. doi:10.1080/13625187.2017.1287351. PMID 28256919.
External links
 |