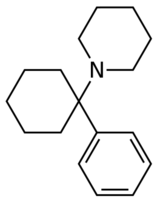
Chemistry:Arylcyclohexylamine
Arylcyclohexylamines, also known as arylcyclohexamines or arylcyclohexanamines, are a chemical class of pharmaceutical, designer, and experimental drugs.
History
Phencyclidine (PCP) is believed to be the first arylcyclohexylamine with recognized anesthetic properties, but several arylcyclohexylamines were described before PCP in the scientific literature, beginning with PCA (1-phenylcyclohexan-1-amine) the synthesis of which was first published in 1907. PCE was reported in 1953 and PCMo (4-(1-phenyl-cyclohexyl)-morpholine[1] see chart below for figure) in 1954, with PCMo described as a potent sedative.[2] Arylcyclohexylamine anesthetics were intensively investigated at Parke-Davis, beginning with the 1956 synthesis of phencyclidine and later the related compound ketamine.[2] The 1970s saw the debut of these compounds, especially PCP and its analogues, as illicitly used recreational drugs due to their dissociative hallucinogenic and euphoriant effects. Since that time, the class has been expanded by scientific research into stimulant, analgesic, and neuroprotective agents, and also by clandestine chemists in search of novel recreational drugs.[3][4][5]
Structure
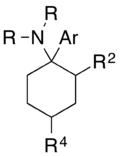
An arylcyclohexylamine is composed of a cyclohexylamine unit with an aryl moiety attachment. The aryl group is positioned geminal to the amine. In the simplest cases, the aryl moiety is typically a phenyl ring, sometimes with additional substitution. The amine is usually not primary; secondary amines such as methylamine or ethylamine, or tertiary cycloalkylamines such as piperidine and pyrrolidine, are the most commonly encountered N-substituents.
Pharmacology
Arylcyclohexylamines varyingly possess NMDA receptor antagonistic,[6][7] dopamine reuptake inhibitory,[8] and μ-opioid receptor agonistic[9] properties. Additionally, σ receptor agonistic,[10] nACh receptor antagonistic,[11] and D2 receptor agonistic[12] actions have been reported for some of these agents. Antagonism of the NMDA receptor confers anesthetic, anticonvulsant, neuroprotective, and dissociative effects; blockade of the dopamine transporter mediates stimulant and euphoriant effects as well as psychosis in high amounts; and activation of the μ-opioid receptor causes analgesic and euphoriant effects. Stimulation of the σ and D2 receptors may also contribute to hallucinogenic and psychotomimetic effects.[12]
These are versatile agents with a wide range of possible pharmacological activities depending on the extent and range to which chemical modifications are implemented.[13][14][15][16][17][18][19][20][21] The various choice of substitutions that are made allows for "fine-tuning" of the pharmacological profile that results. As examples, BTCP is a selective dopamine reuptake inhibitor,[8] PCP is primarily an NMDA antagonist,[6] and BDPC is a potent μ-opioid agonist,[22] while PRE-084 is a selective sigma receptor agonist.[23] Thus, radically different pharmacology is possible through different structural combinations.
Notes on numbering
PCP itself is composed of three six-membered rings, which can each be substituted by a variety of groups. These are traditionally numbered in the older research as first the cyclohexyl ring, then the phenyl, and finally the piperidine ring, with the different rings represented by prime notation (') next to the number. For instance, 4-methyl-PCP, 4'-methyl-PCP and 4''-methyl-PCP are all known compounds, with similar activity but quite different potencies.
However, since the widespread sale of these compounds as grey-market designer drugs, nearly all such compounds that have come to prominence either have a bare cyclohexyl ring or a 2-ketocyclohexyl ring, while the piperidine is replaced by a variety of alkyl or cycloalkyl amines and most substitution has taken place on the phenyl ring. Consequently, it is common for widely used phenyl substituted analogues such as 3'-MeO-PCP and 3'-MeO-PCE to be referred to as 3-MeO-PCP and 3-MeO-PCE without the prime, even though this is technically incorrect and could lead to confusion.
List of arylcyclohexylamines
| Structures | Compound | Aryl Substituent | N Group | Cyclohexyl ring | CAS number |
|---|---|---|---|---|---|
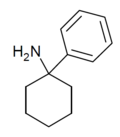 |
PCA[24] | Phenyl | NH2 | - | 1934-71-0 |
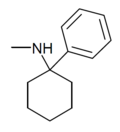 |
PCM[24] | Phenyl | Methylamino | - | 2201-16-3 |
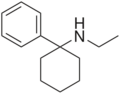 |
Eticyclidine | Phenyl | Ethylamino | - | 2201-15-2 |
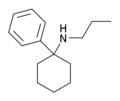 |
PCPr[25] | Phenyl | n-Propylamino | - | 18949-81-0 |
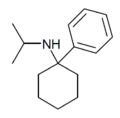 |
PCiP | Phenyl | Isopropylamino | - | 1195-42-2 |
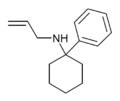 |
PCAL [26] | Phenyl | Allylamino | - | 2185-95-7 |
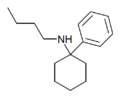 |
PCBu | Phenyl | n-Butylamino | - | 73166-29-7 |
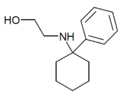 |
PCEOH | Phenyl | Hydroxyethylamino | - | 2201-22-1 |
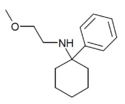 |
PCMEA[27] | Phenyl | Methoxyethylamino | - | 2201-57-2 |
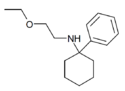 |
PCEEA | Phenyl | Ethoxyethylamino | - | 1072895-05-6 |
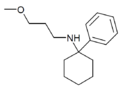 |
PCMPA | Phenyl | Methoxypropylamino | - | 2201-58-3 |
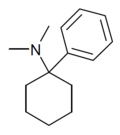 |
PCDM[24] | Phenyl | Dimethylamino | - | 2201-17-4 |
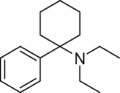 |
Dieticyclidine | Phenyl | Diethylamino | - | 2201-19-6 |
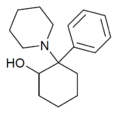 |
2-HO-PCP[6] | Phenyl | Piperidine | 2-Hydroxy | 94852-58-1 |
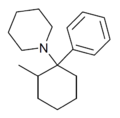 |
2-Me-PCP[28] | Phenyl | Piperidine | 2-Methyl | 59397-29-4 |
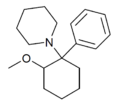 |
2-MeO-PCP[29] | Phenyl | Piperidine | 2-Methoxy | 78636-34-7 |
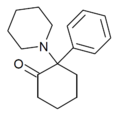 |
2-Keto-PCP | Phenyl | Piperidine | 2-Keto | 101688-16-8 |
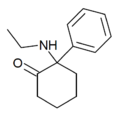 |
Eticyclidone ("O-PCE") | Phenyl | Ethylamino | 2-Keto | 6740-82-5 |
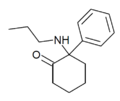 |
2-Keto-PCPr | Phenyl | n-Propylamino | 2-Keto | |
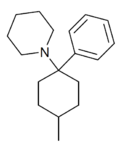 |
4-Methyl-PCP | Phenyl | Piperidine | 4-Methyl | 19420-52-1 |
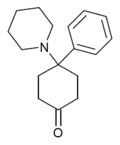 |
4-Keto-PCP[30] | Phenyl | Piperidine | 4-Keto | 65620-13-5 |
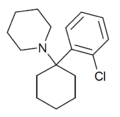 |
2'-Cl-PCP | o-Chlorophenyl | Piperidine | - | 2201-31-2 |
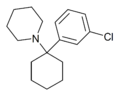 |
3'-Cl-PCP | m-Chlorophenyl | Piperidine | - | 2201-32-3 |
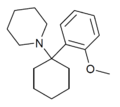 |
2'-MeO-PCP | o-Methoxyphenyl | Piperidine | - | 2201-34-5 |
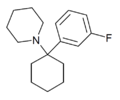 |
3'-F-PCP[31] | m-Fluorophenyl | Piperidine | - | 89156-99-0 |
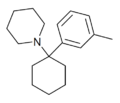 |
3'-Me-PCP[32] | m-Tolyl | Piperidine | - | 2201-30-1 |
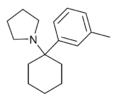 |
3'-Me-PCPy | m-Tolyl | Pyrrolidine | - | 1622348-63-3 |
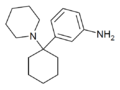 |
3'-NH2-PCP | m-Aminophenyl | Piperidine | - | 72242-00-3 |
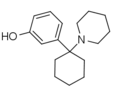 |
3'-HO-PCP | m-Hydroxyphenyl | Piperidine | - | 79787-43-2 |
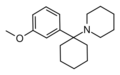 |
3'-MeO-PCP | m-Methoxyphenyl | Piperidine | - | 72242-03-6 |
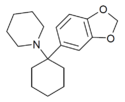 |
3',4'-MD-PCP | 3,4-Methylenedioxyphenyl | Piperidine | - | |
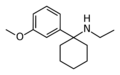 |
3'-MeO-PCE | m-Methoxyphenyl | Ethylamino | - | 1364933-80-1 |
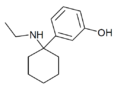 |
3'-HO-PCE | m-Hydroxyphenyl | Ethylamino | - | |
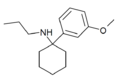 |
3'-MeO-PCPr | m-Methoxyphenyl | n-Propylamino | - | 1364933-81-2 |
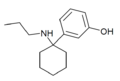 |
3'-HO-PCPr | m-Hydroxyphenyl | n-Propylamino | - | |
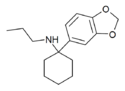 |
3',4'-MD-PCPr | 3,4-Methylenedioxyphenyl | n-Propylamino | - | |
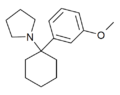 |
3'-MeO-PCPy[32] | m-Methoxyphenyl | Pyrrolidine | - | 1364933-79-8 |
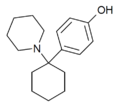 |
4'-HO-PCP | p-Hydroxyphenyl | Piperidine | - | 66568-88-5 |
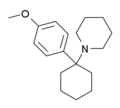 |
Methoxydine (4'-MeO-PCP) | p-Methoxyphenyl | Piperidine | - | 2201-35-6 |
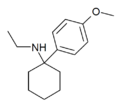 |
4'-MeO-PCE | p-Methoxyphenyl | Ethylamino | - | |
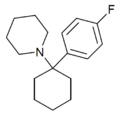 |
4'-F-PCP[31] | p-Fluorophenyl | Piperidine | - | 22904-99-0 |
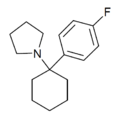 |
4'-F-PCPy | p-Fluorophenyl | Pyrrolidine | - | |
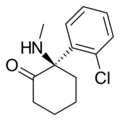 |
Arketamine | o-Chlorophenyl | Methylamino | 2-Keto | 33643-49-1 |
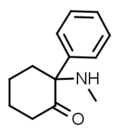 |
Deschloroketamine | Phenyl | Methylamino | 2-Keto | 7063-30-1 |
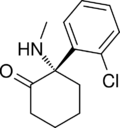 |
Esketamine | o-Chlorophenyl | Methylamino | 2-Keto | 33643-46-8 |
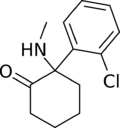 |
Ketamine | o-Chlorophenyl | Methylamino | 2-Keto | 6740-88-1 |
 |
Hydroxynorketamine | o-Chlorophenyl | NH2 | 2-Keto, 6-Hydroxy | 81395-70-2 |
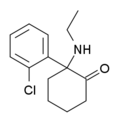 |
Ethketamine | o-Chlorophenyl | Ethylamino | 2-Keto | 1354634-10-8 |
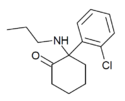 |
NPNK | o-Chlorophenyl | n-Propylamino | 2-Keto | 2749326-65-4 |
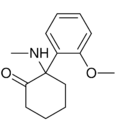 |
Methoxyketamine | o-Methoxyphenyl | Methylamino | 2-Keto | 7063-51-6 |
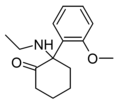 |
2-MeO-NEK[33] | o-Methoxyphenyl | Ethylamino | 2-Keto | |
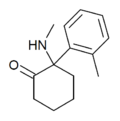 |
oMDCK[34] | o-Tolyl | Methylamino | 2-Keto | 7063-37-8 |
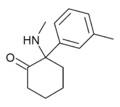 |
mMDCK | m-Tolyl | Methylamino | 2-Keto | |
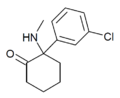 |
meta-Ketamine | m-Chlorophenyl | Methylamino | 2-Keto | 7063-53-8 |
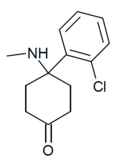 |
iso-Ketamine | o-Chlorophenyl | Methylamino | 4-Keto | |
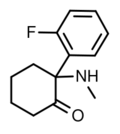 |
2-Fluorodeschloroketamine | o-Fluorophenyl | Methylamino | 2-Keto | 111982-50-4 |
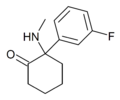 |
3-Fluorodeschloroketamine | m-Fluorophenyl | Methylamino | 2-Keto | 2657761-23-2 |
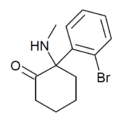 |
Bromoketamine | o-Bromophenyl | Methylamino | 2-Keto | 120807-70-7 |
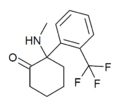 |
TFMDCK | o-Trifluoromethylphenyl | Methylamino | 2-Keto | 1782149-73-8 |
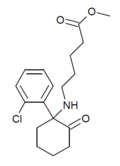 |
SN 35210[35] | o-Chlorophenyl | Carbomethoxybutylamino | 2-Keto | 1450615-41-4 |
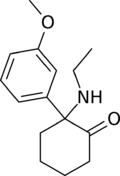 |
Methoxetamine | m-Methoxyphenyl | Ethylamino | 2-Keto | 1239943-76-0 |
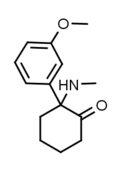 |
Methoxmetamine | m-Methoxyphenyl | Methylamino | 2-Keto | 1781829-56-8 |
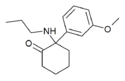 |
Methoxpropamine | m-Methoxyphenyl | n-Propylamino | 2-Keto | 2504100-71-2 |
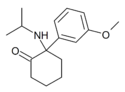 |
MXiPr | m-Methoxyphenyl | i-Propylamino | 2-Keto | |
 |
Ethoxetamine | m-Ethoxyphenyl | Ethylamino | 2-Keto | |
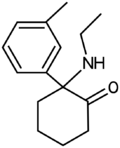 |
Deoxymethoxetamine (3-Me-2'-Oxo-PCE) | m-Tolyl | Ethylamino | 2-Keto | 2666932-45-0 |
 |
Br-MXE | 2-bromo-5-methoxyphenyl | Ethylamino | 2-Keto | |
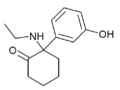 |
Hydroxetamine (HXE) | m-Hydroxyphenyl | Ethylamino | 2-Keto | 1620054-73-0 |
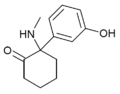 |
HXM | m-Hydroxyphenyl | Methylamino | 2-Keto | |
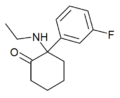 |
Fluorexetamine (FXE) | m-Fluorophenyl | Ethylamino | 2-Keto | |
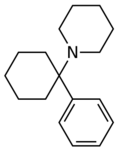 |
Phencyclidine (PCP) | Phenyl | Piperidine | - | 77-10-1 |
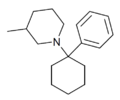 |
PC3MP | Phenyl | 3-Methylpiperidine | - | 2201-41-4 |
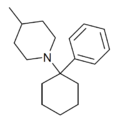 |
PC4MP | Phenyl | 4-Methylpiperidine | - | 2201-42-5 |
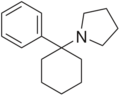 |
Rolicyclidine (PCPy) | Phenyl | Pyrrolidine | - | 2201-39-0 |
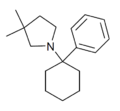 |
PCDMPy | Phenyl | 3,3-Dimethylpyrrolidine | - | |
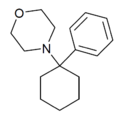 |
PCMo | Phenyl | Morpholine | - | 2201-40-3 |
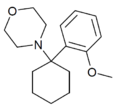 |
Methoxy-PCM[7] (2'-MeO-PCMo) | o-Methoxyphenyl | Morpholine | - | 1314323-88-0 |
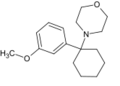 |
3'-MeO-PCMo | m-Methoxyphenyl | Morpholine | - | 138873-80-0 |
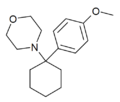 |
4'-MeO-PCMo | p-Methoxyphenyl | Morpholine | - | |
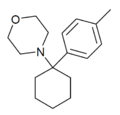 |
Methyl-PCM[36] (4'-Me-PCMo) | p-Tolyl | Morpholine | - | 120803-52-3 |
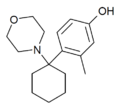 |
Hydroxy-methyl-PCM | 2-Methyl-4-hydroxyphenyl | Morpholine | - | 1314323-89-1 |
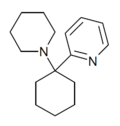 |
PYCP [37] | 2-Pyridinyl | Piperidine | - | |
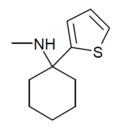 |
TCM | 2-Thienyl | Methylamino | - | 139401-07-3 |
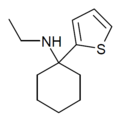 |
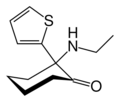
TCE | 2-Thienyl | Ethylamino | - | 101589-62-2 |
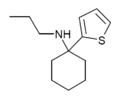 |
TCPr [38] | 2-Thienyl | Propylamino | - | |
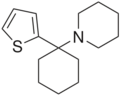 |
Tenocyclidine (TCP) | 2-Thienyl | Piperidine | - | 21500-98-1 |
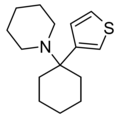 |
T3CP | 3-Thienyl | Piperidine | - | 19420-50-9 |
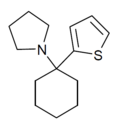 |
TCPy | 2-Thienyl | Pyrrolidine | - | 22912-13-6 |
 |
Tiletamine | 2-Thienyl | Ethylamino | 2-Keto | 14176-49-9 |
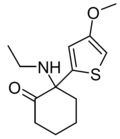 |
MXTE | 4-Methoxy-2-thienyl | Ethylamino | 2-Keto | |
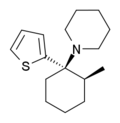 |
Gacyclidine | 2-Thienyl | Piperidine | 2-Methyl | 68134-81-6 |
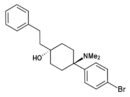 |
BDPC | p-Bromophenyl | Dimethylamino | 4-Phenethyl-4-hydroxy | 77239-98-6 |
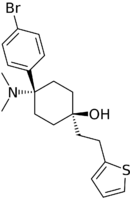 |
C-8813 | p-Bromophenyl | Dimethylamino | 4-(thiophen-2-yl)ethyl-4-hydroxy | 616898-54-5 |
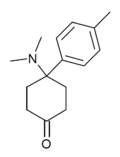 |
Dimetamine[39] | p-Tolyl | Dimethylamino | 4-Keto | 65619-06-9 |
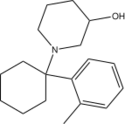 |
3''-OH-2'-Me-PCP [40] | o-Tolyl | 3-Hydroxypiperidine | - | |
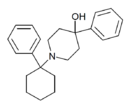 |
4''-Ph-4''-OH-PCP [41] | Phenyl | 4-Phenyl-4-hydroxypiperidine | - | 77179-39-6 |
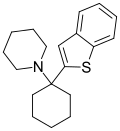 |
BTCP[42] | Benzothiophen-2-yl | Piperidine | - | 112726-66-6 |
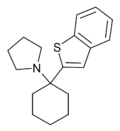 |
BTCPy[10] | Benzothiophen-2-yl | Pyrrolidine | - | |
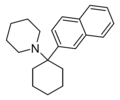 |
GK-189[43] | Naphthalen-2-yl | Piperidine | - | 81490-58-6 |
Related compounds
Other similar compounds exist where the base ring has been varied, or the amine chain replaced with other groups.[44] More cycloalkane ring sizes have been experimented with than just purely thinking in terms of the cyclohexylamine. The cyclopentyl homologue of PCP is active with around one-tenth the potency,[45] while the cycloheptyl and cyclooctyl derivatives are inactive, though some substituted arylcycloheptylamines retain activity.[46] The requisite cycloalkylketone is reacted with PhMgBr; 3° alcohol is then reacted with NaN3; azide then reduced with LAH. Then in the final step the piperidine ring is constructed with 1-5-dibromo-pentane.[47] Other compounds are known where the cyclohexyl base ring is replaced by rings such as norbornyl, adamantyl,[48] tetralin, oxane, thiane [49] or piperidine.[50] Conformationally constrained analogs have been prepared and researched by Morieti et al.[51]
| Structure | Compound | Aryl Substituent | N Group | Base ring | CAS number |
|---|---|---|---|---|---|
 |
PCPEP | Phenyl | Piperidine | Cyclopentyl | 23036-19-3 |
 |
3F-PCHEPy | 3-Fluorophenyl | Pyrrolidine | Cycloheptyl | |
 |
3-MeO-PBCHP | 3-Methoxyphenyl | Piperidine | Bicyclo[2.2.1]heptane | |
 |
PADP (P2AP) | Phenyl | Piperidine | Adamantyl | 72241-99-7 |
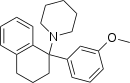 |
3-MeO-PTP | 3-Methoxyphenyl | Piperidine | Tetralin | |
 |
HHFA | Fused phenyl | Amino | Hexahydrofluorene | |
 |
DHPQ | Phenyl | Decahydroquinoline | ||
 |
POXP | Phenyl | Piperidine | Oxane | |
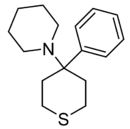 |
PTHP | Phenyl | Piperidine | Thiane | |
 |
MPBPip | Phenyl | Piperidine | N-Methylpiperidine | 36882-04-9 |
 |
BnCP | Benzyl | Piperidine | Cyclohexyl | 22912-07-8 |
 |
YNCP | Ethynyl | Piperidine | Cyclohexyl | 51165-02-7 |
 |
ALCP | Allyl | Piperidine | Cyclohexyl | 7418-80-6 |
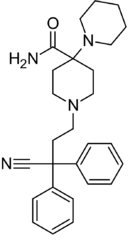 |
Piritramide | Replaced by carboxamide | Piperidine | N-(3-cyano-3,3-diphenylpropyl)piperidine | 302-41-0 |
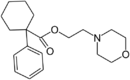 |
PRE-084 | Phenyl | Morpholinylethylcarboxylate | Cyclohexyl | 138847-85-5 |
 |
Clofenciclan | p-Chlorophenyl | Diethylaminoethoxy | Cyclohexyl | 5632-52-0 |
References
- ↑ "4-(1-phenyl-cyclohexyl)-morpholine". chemsrc. https://www.chemsrc.com/cas/2201-40-3_1255636.html.
- ↑ 2.0 2.1 "From PCP to MXE: a comprehensive review of the non-medical use of dissociative drugs". Drug Testing and Analysis 6 (7–8): 614–32. 2014. doi:10.1002/dta.1620. PMID 24678061.
- ↑ Designer Drugs Directory. Elsevier. 1998. ISBN 0-444-20525-X.
- ↑ "Phencyclidine-Based New Psychoactive Substances". New Psychoactive Substances. Handbook of Experimental Pharmacology. 252. August 2018. pp. 261–303. doi:10.1007/164_2018_124. ISBN 978-3-030-10560-0.
- ↑ "1,2-Diarylethylamine- and Ketamine-Based New Psychoactive Substances". New Psychoactive Substances. Handbook of Experimental Pharmacology. 252. 2018. pp. 305–352. doi:10.1007/164_2018_148. ISBN 978-3-030-10560-0.
- ↑ 6.0 6.1 6.2 "Synthesis and biological properties of 2-hydroxy-1-(1-phenyltetralyl)piperidine and some of its intermediates as derivatives of phencyclidine". Arzneimittel-Forschung 55 (9): 528–32. 2005. doi:10.1055/s-0031-1296900. PMID 16229117.
- ↑ 7.0 7.1 "New morpholine analogues of phencyclidine: chemical synthesis and pain perception in rats". Pharmacology, Biochemistry, and Behavior 98 (2): 227–33. April 2011. doi:10.1016/j.pbb.2010.12.019. PMID 21215770.
- ↑ 8.0 8.1 "Role of the aromatic group in the inhibition of phencyclidine binding and dopamine uptake by PCP analogs". Pharmacology, Biochemistry, and Behavior 32 (3): 699–705. March 1989. doi:10.1016/0091-3057(89)90020-8. PMID 2544905.
- ↑ "A novel phencyclidine analog interacts selectively with mu opioid receptors". The Journal of Pharmacology and Experimental Therapeutics 230 (2): 383–6. August 1984. PMID 6086884.
- ↑ 10.0 10.1 "Synthesis and biological evaluation of 1-[1-(2-benzo[b]thienyl)cyclohexyl]piperidine homologues at dopamine-uptake and phencyclidine- and sigma-binding sites". Journal of Medicinal Chemistry 36 (9): 1188–93. April 1993. doi:10.1021/jm00061a009. PMID 8098066.
- ↑ "Determinants of phencyclidine potency on the nicotinic acetylcholine receptors from muscle and electric organ". Cellular and Molecular Neurobiology 19 (6): 745–57. December 1999. doi:10.1023/A:1006905106834. PMID 10456235.
- ↑ 12.0 12.1 "Dopamine receptor contribution to the action of PCP, LSD and ketamine psychotomimetics". Molecular Psychiatry 10 (9): 877–83. September 2005. doi:10.1038/sj.mp.4001682. PMID 15852061.
- ↑ "New analgesics derived from the phencyclidine analogue thienylcyclidine". Arzneimittel-Forschung 46 (5): 505–8. May 1996. PMID 8737636.
- ↑ "Synthesis and study the analgesic effects of new analogues of ketamine on female wistar rats". Medicinal Chemistry 8 (2): 246–51. March 2012. doi:10.2174/157340612800493683. PMID 22385170.
- ↑ "Synthesis and study on analgesic effects of 1-[1-(4-methylphenyl) (cyclohexyl)] 4-piperidinol and 1-[1-(4-methoxyphenyl) (cyclohexyl)] 4-piperidinol as two new phencyclidine derivatives". Arzneimittel-Forschung 59 (4): 202–6. 2009. doi:10.1055/s-0031-1296386. PMID 19517897.
- ↑ "Synthesis and pain perception of new analogues of phencyclidine in NMRI male mice". Mini Reviews in Medicinal Chemistry 14 (1): 64–71. January 2014. doi:10.2174/1389557513666131119203551. PMID 24251803.
- ↑ "Synthesis and analgesic effects of new pyrrole derivatives of phencyclidine in mice". Arzneimittel-Forschung 61 (5): 296–300. 2011. doi:10.1055/s-0031-1296202. PMID 21755813.
- ↑ "Synthesis and determination of chronic and acute thermal and chemical pain activities of a new derivative of phencyclidine in rats". Iranian Journal of Pharmaceutical Research 9 (4): 379–85. 2010. PMID 24381602.
- ↑ "Synthesis and determination of acute and chronic pain activities of 1-[1-(3-methylphenyl) (tetralyl)]piperidine as a new derivative of phencyclidine via tail immersion and formalin tests". Arzneimittel-Forschung 60 (1): 30–5. 2010. doi:10.1055/s-0031-1296245. PMID 20184224.
- ↑ "Effect of phencyclidine derivatives on anxiety-like behavior using an elevated-plus maze test in mice". Advances in Clinical and Experimental Medicine 21 (3): 307–12. 2012. PMID 23214193.
- ↑ "Synthesis and Antinociception Activities of Some Novel Derivatives of Phencyclidine with Substituted Aminobenzothiazoles". Mini Reviews in Medicinal Chemistry 17 (1): 78–84. 2017. doi:10.2174/1389557516666160428112532. PMID 27121715.
- ↑ "4-(p-Bromophenyl)-4-(dimethylamino)-1-phenethylcyclohexanol, an extremely potent respresentative of a new analgesic series". Journal of Medicinal Chemistry 22 (10): 1157–8. October 1979. doi:10.1021/jm00196a001. PMID 513062.
- ↑ "PRE-084, a sigma selective PCP derivative, attenuates MK-801-induced impairment of learning in mice". Pharmacology, Biochemistry, and Behavior 49 (4): 859–69. December 1994. doi:10.1016/0091-3057(94)90235-6. PMID 7886099. https://zenodo.org/record/1258299.
- ↑ 24.0 24.1 24.2 "Synthesis and anticonvulsant activity of 1-phenylcyclohexylamine analogues". Journal of Medicinal Chemistry 33 (5): 1452–8. May 1990. doi:10.1021/jm00167a027. PMID 2329567.
- ↑ "Metabolism and toxicological detection of a new designer drug, N-(1-phenylcyclohexyl)propanamine, in rat urine using gas chromatography-mass spectrometry". Journal of Chromatography A 1186 (1–2): 380–90. April 2008. doi:10.1016/j.chroma.2007.11.002. PMID 18035363.
- ↑ "N-allyl analogues of phencyclidine: chemical synthesis and pharmacological properties". Journal of Medicinal Chemistry 27 (10): 1267–71. October 1984. doi:10.1021/jm00376a006. PMID 6481761.
- ↑ "Investigations on the cytochrome P450 (CYP) isoenzymes involved in the metabolism of the designer drugs N-(1-phenyl cyclohexyl)-2-ethoxyethanamine and N-(1-phenylcyclohexyl)-2-methoxyethanamine". Biochemical Pharmacology 77 (3): 444–50. February 2009. doi:10.1016/j.bcp.2008.10.024. PMID 19022226.
- ↑ "Synthesis, stereochemistry, and biological activity of the 1-(1-phenyl-2-methylcyclohexyl)piperidines and the 1-(1-phenyl-4-methylcyclohexyl)piperidines. Absolute configuration of the potent trans-(-)-1-(1-phenyl-2-methylcyclohexyl)piperidine". Journal of Medicinal Chemistry 34 (8): 2615–23. August 1991. doi:10.1021/jm00112a041. PMID 1875352.
- ↑ "Synthesis with improved yield and study on the analgesic effect of 2-methoxyphencyclidine". Arzneimittel-Forschung 56 (5): 346–50. 2006. doi:10.1055/s-0031-1296732. PMID 16821645.
- ↑ "The dopaminergic alterations induced by 4-F-PCP and 4-Keto-PCP may enhance their drug-induced rewarding and reinforcing effects: Implications for abuse". Addiction Biology 26 (4): e12981. July 2021. doi:10.1111/adb.12981. PMID 33135332.
- ↑ 31.0 31.1 "Syntheses of fluorinated phencyclidine analogs". Journal of Fluorine Chemistry 114: 39–42. 2002. doi:10.1016/S0022-1139(01)00565-6.
- ↑ 32.0 32.1 "Preparation and analytical characterization of 1-(1-phenylcyclohexyl)piperidine (PCP) and 1-(1-phenylcyclohexyl)pyrrolidine (PCPy) analogues". Drug Testing and Analysis 6 (7–8): 633–50. 2013. doi:10.1002/dta.1468. PMID 23554350.
- ↑ "The novel methoxetamine analogs N-ethylnorketamine hydrochloride (NENK), 2-MeO-N-ethylketamine hydrochloride (2-MeO-NEK), and 4-MeO-N-ethylketamine hydrochloride (4-MeO-NEK) elicit rapid antidepressant effects via activation of AMPA and 5-HT2 receptors". Psychopharmacology 236 (7): 2201–2210. July 2019. doi:10.1007/s00213-019-05219-x. PMID 30891619.
- ↑ Kruegel AC, Sames D, Hashimoto K, "Arylcyclohexylamine derivatives and their use in the treatment of psychiatric disorders", WO patent 2021134086, published 1 July 2021, assigned to Gilgamesh Pharmaceuticals, Inc.and The Trustees Of Columbia University In The City Of New York.
- ↑ "Determination of the Hypnotic Potency in Rats of the Novel Ketamine Ester Analogue SN 35210". Pharmacology 96 (5–6): 226–32. 2015. doi:10.1159/000439598. PMID 26352278.
- ↑ "Synthesis and determination of acute and chronic pain activities of 1-[1-(4-methylphenyl) (cyclohexyl)] morpholine as a new phencyclidine derivative in rats". Arzneimittel-Forschung 61 (2): 92–7. 2011. doi:10.1055/s-0031-1296173. PMID 21428243.
- ↑ "Novel analogues of ketamine and phencyclidine as NMDA receptor antagonists". Bioorganic & Medicinal Chemistry Letters 21 (7): 2059–63. April 2011. doi:10.1016/j.bmcl.2011.02.009. PMID 21334205.
- ↑ "Syntheses and analytical characterizations of N-alkyl-arylcyclohexylamines". Drug Testing and Analysis 8 (8): 801–15. August 2016. doi:10.1002/dta.1861. PMID 26360516. http://researchonline.ljmu.ac.uk/id/eprint/3240/1/%EF%BF%BC%EF%BF%BC%EF%BF%BCDTA-15-0185.R1.pdf.
- ↑ "4-Amino-4-arylcyclohexanones and their derivatives, a novel class of analgesics. 1. Modification of the aryl ring". Journal of Medicinal Chemistry 23 (4): 424–30. April 1980. doi:10.1021/jm00178a014. PMID 7381841.
- ↑ "Synthesis and analgesic effects of 1-[1-(2-methylphenyl)(cyclohexyl)]-3-piperidinol as a new derivative of phencyclidine in mice". Arzneimittel-Forschung 60 (8): 492–6. 2010. doi:10.1055/s-0031-1296317. PMID 20863005.
- ↑ "New analgesic drugs derived from phencyclidine". Journal of Medicinal Chemistry 24 (5): 496–9. May 1981. doi:10.1021/jm00137a004. PMID 7241506.
- ↑ "[3H]N-[1-(2-benzo(b)thiophenyl)cyclohexyl]piperidine ([3H]BTCP): a new phencyclidine analog selective for the dopamine uptake complex". European Journal of Pharmacology 148 (3): 427–36. April 1988. doi:10.1016/0014-2999(88)90122-7. PMID 3384005.
- ↑ Kamenka JM, et al. Substituted cyclic amines and pharmaceutical composition containing them. Patent US5248686, 28 September 1993
- ↑ Wallach JV. Structure activity relationship (SAR) studies of arylcycloalkylamines as N-methyl-D-aspartate receptor antagonists. PhD. Thesis, University of the Sciences in Philadelphia, 19 Dec 2014.
- ↑ "Illicit synthesis of phencyclidine (PCP) and several of its analogs". Clinical Toxicology 9 (4): 553–60. 1976. doi:10.3109/15563657608988157. PMID 975751.
- ↑ "Syntheses and N-methyl-D-aspartate receptor antagonist pharmacology of fluorinated arylcycloheptylamines". Medicinal Chemistry (Shariqah (United Arab Emirates)) 10 (8): 843–52. 2014. doi:10.2174/1573406410666140428104444. PMID 24773376.
- ↑ "Structure-activity relationships of the cycloalkyl ring of phencyclidine". Journal of Medicinal Chemistry 24 (12): 1429–32. December 1981. doi:10.1021/jm00144a011. PMID 7310819.
- ↑ "Geometries and conformational processes in phencyclidine and a rigid adamantyl analogue: variable-temperature NMR, X-ray crystallographic, and molecular mechanics studies". Journal of Medicinal Chemistry 26 (4): 479–86. April 1983. doi:10.1021/jm00358a005. PMID 6834381.
- ↑ Sisco E, Urbas A. Identification and Characterization of Designer Phencyclidines (PCPs) in Forensic Casework
- ↑ Gerhard O, Eberhard E. 4-amino-piperidines. US3311624A
- ↑ "Rigid phencyclidine analogues. Binding to the phencyclidine and sigma 1 receptors". Journal of Medicinal Chemistry 41 (4): 468–77. February 1998. doi:10.1021/jm970059p. PMID 9484497.
Further reading
- "From PCP to MXE: a comprehensive review of the non-medical use of dissociative drugs". Drug Testing and Analysis 6 (7–8): 614–32. 2014. doi:10.1002/dta.1620. PMID 24678061.
External links
- Synthesis and Effects of PCP Analogs
- Interview with a Ketamine Chemist
- New Drugs: Designing Novel Arylcyclohexylamines
 |