Chemistry:5-MeO-MPMI
From HandWiki
Short description: Chemical compound
 | |
| Legal status | |
|---|---|
| Legal status | |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| ChEMBL | |
| Chemical and physical data | |
| Formula | C15H20N2O |
| Molar mass | 244.338 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
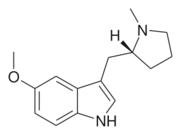
5-MeO-MPMI (also known as 5-Methoxy-N-methyl-(α,N-trimethylene)tryptamine) is a tryptamine derivative that is a psychedelic drug. It was first developed by the team led by JE Macor in 1992,[1] and subsequently investigated by the team led by David Nichols from Purdue University in the late 1990s. This compound produces psychedelic-appropriate responding in animal tests with a similar potency to the amphetamine-derived psychedelic DOI, and has two enantiomers, with only the (R)-enantiomer being active.[2]
See also
- 5-MeO-pyr-T
- 5F-MPMI
- 4-HO-MPMI
- 4-HO-pyr-T
- CP-135,807
- MPMI
References
- ↑ "Synthesis and serotonergic pharmacology of the enantiomers of 3-[(N-methylpyrrolidin-2-yl)methyl]-5-methoxy-1H-indole: discovery of stereogenic differentiation in the aminoethyl side chain of the neurotransmitter serotonin". Journal of Medicinal Chemistry 35 (23): 4503–5. 1992. doi:10.1021/jm00101a032. PMID 1447752.
- ↑ "Further studies on oxygenated tryptamines with LSD-like activity incorporating a chiral pyrrolidine moiety into the side chain". Journal of Medicinal Chemistry 42 (20): 4257–4263. 1999. doi:10.1021/jm990325u. PMID 10514296.
 |
