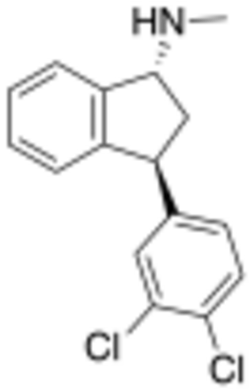
Chemistry:Indatraline
 | |
| Clinical data | |
|---|---|
| Routes of administration | Oral |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| ChEMBL | |
| Chemical and physical data | |
| Formula | C16H15Cl2N |
| Molar mass | 292.20 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Indatraline hydrochloride (Lu 19-005) is an antidepressive agent and non-selective monoamine transporter inhibitor that blocks the reuptake of dopamine, norepinephrine, and serotonin with similar efficacy to cocaine.[1] This compound may be used to treat cocaine addictions as its effects have a slower onset and a longer duration than those of cocaine.[2] Lu 19-005 has been shown to block the action of methamphetamine and MDMA in laboratory experiments.[3]
Methylation
Indatraline is N-alkylated at the amino group, making it possible to slow the onset of action, so that it is not until N-demethylation occurs that the molecules become active. N-methylindatraline has a longer duration than indatraline because norindatraline is inactive, whereas demethylating N-methylindatraline does not terminate the actions of the parent compound.
Effects of N-dimethylindatraline start about 20–30 minutes after administration; it takes a longer time for this chemical to absorb into the body than cocaine.[4]
Synthesis
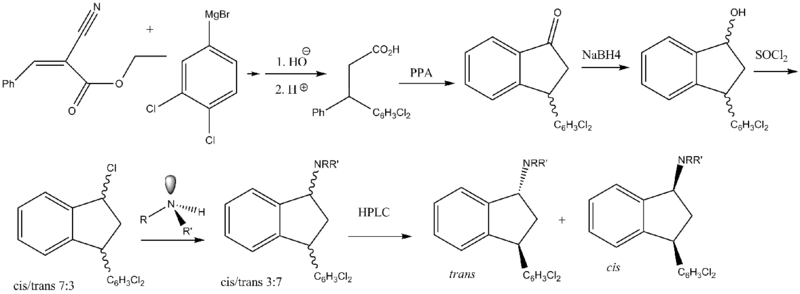
Two main routes have been reported. The first route was reported by Bøgesø and co-workers.[5]
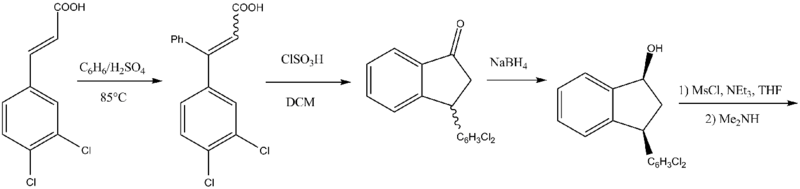
The other has been adapted to scale-up:[6]
Another method involves the contraction of a dihydronaphthalene (6–6 fused system) to form the 6–5 indane skeleton.[7]
Routes based on 1-indanone-type intermediates are not as simple as a direct reduction of an imine or oxime. The undesirable cis diastereomers are formed instead of the desirable trans isomers. This adds an extra step to the synthetic route. First, the ketones are reduced to mostly cis alcohols. Second, the cis alcohols are converted to the corresponding mesylates, conserving stereochemistry. Third, the mesylates can then be reacted, e.g. with, N-methylbenzylamine, causing a Walden inversion (SN2). Finally, the removal of the benzyl affords the product as a racemic mixture.
See also
References
- ↑ "Antidepressant indatraline induces autophagy and inhibits restenosis via suppression of mTOR/S6 kinase signaling pathway". Scientific Reports 6 (1): 34655. October 2016. doi:10.1038/srep34655. PMID 27694974.
- ↑ "Effects of the long-acting monoamine reuptake inhibitor indatraline on cocaine self-administration in rhesus monkeys". The Journal of Pharmacology and Experimental Therapeutics 291 (1): 60–69. October 1999. PMID 10490887.
- ↑ "Neurochemical neutralization of methamphetamine with high-affinity nonselective inhibitors of biogenic amine transporters: a pharmacological strategy for treating stimulant abuse". Synapse 35 (3): 222–227. March 2000. doi:10.1002/(SICI)1098-2396(20000301)35:3<222::AID-SYN7>3.0.CO;2-K. PMID 10657029. https://zenodo.org/record/1235504.
- ↑ "A slow-onset, long-duration indanamine monoamine reuptake inhibitor as a potential maintenance pharmacotherapy for psychostimulant abuse: effects in laboratory rat models relating to addiction". Neuropharmacology 51 (5): 993–1003. October 2006. doi:10.1016/j.neuropharm.2006.06.009. PMID 16901516.
- ↑ "3-Phenyl-1-indanamines. Potential antidepressant activity and potent inhibition of dopamine, norepinephrine, and serotonin uptake". Journal of Medicinal Chemistry 28 (12): 1817–28. December 1985. doi:10.1021/jm00150a012. PMID 2999402.
- ↑ "Slow-onset, long-duration 3-(3',4'-dichlorophenyl)-1-indanamine monoamine reuptake blockers as potential medications to treat cocaine abuse". Journal of Medicinal Chemistry 43 (26): 4981–92. December 2000. doi:10.1021/jm000201d. PMID 11150168.
- ↑ "Iodine(III)-promoted ring contraction of 1,2-dihydronaphthalenes: a diastereoselective total synthesis of (±)-indatraline". Organic Letters 9 (8): 1433–6. April 2007. doi:10.1021/ol070027o. PMID 17371034.
 |