Chemistry:RTI-112
This article may be too technical for most readers to understand. Please help improve it to make it understandable to non-experts, without removing the technical details. (August 2009) (Learn how and when to remove this template message) |
 | |
| Identifiers | |
|---|---|
| |
| CAS Number |
|
| PubChem CID | |
| ChemSpider | |
| UNII | |
| ChEMBL | |
| Chemical and physical data | |
| Formula | C17H22ClNO2 |
| Molar mass | 307.82 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
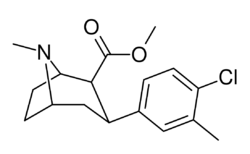
RTI(-4229)-112 (2β-carbomethoxy-3β-(3-methyl-4-chlorophenyl)tropane) is a synthetic stimulant drug from the phenyltropane family. It is primarily used in scientific research to study the brain's reward system. In contrast to more well-known stimulants that primarily affect one type of brain cell communication, RTI-112 is a nonselective triple reuptake inhibitor. This means it simultaneously affects three important brain chemicals: serotonin, dopamine, and norepinephrine.[1]
In vitro tests show a very similar serotonin transporter (SERT)/dopamine transporter (DAT)/norepinephrine transporter (NET) selectivity to cocaine,[2] although in vivo behaviour is different:
"The nonselective monoamine transporter inhibitor RTI-126 and the DAT-selective inhibitors RTI-150 and RTI-336 both had a faster rate of onset (30 min) and a short duration of action (4h). In contrast, the nonselective monoamine transporter inhibitor RTI-112 had a slower rate of onset (30–60 min) and a longer duration of action (10h). The DAT-selective inhibitors RTI-171 and RTI-177 also had slower rates of onset (30–120 min), but RTI-171 had a short duration of action (2.5 h) while RTI-177 had a very long duration of action (20 h)."[3]
The efficacy of cocaine analogs to elicit self-administration is related to the rate at which they are administered.[clarification needed] Slower onset analogs are less likely to function as behavioral stimulants than analogs eliciting a faster rate of onset.[4] Nonselective analogs are less likely to function as "reinforcers" than reuptake inhibitors that have DAT specificity.[3]
In order for a dopamine reuptake inhibitor (DRI) such as cocaine to induce euphoria, PET scans on primates reveal that the DAT occupancy needs to be >60%.[5]
RTI-112 has equipotent in vitro affinity at the SERT, NET and DAT, respectively.[2] RTI-112 was not reliably self-administered, in contrast to the DAT selective reuptake inhibitors that were used in this study.[2]
In vivo at the ED50, RTI-112 had no DAT occupancy at all.[2] At the ED50, almost all of the RTI-112 occupied the SERT at this dose.[2] A significantly higher dose was required to get >70% DAT occupancy in the case of RTI-112;[2] however, RTI-112 was still able to suppress cocaine administration at the ED50, suggesting a serotonergic mechanism was responsible for this.[2]
References
- ↑ "Interaction of cocaine and dopamine transporter inhibitors on behavior and neurochemistry in monkeys". Pharmacology, Biochemistry, and Behavior 80 (3): 481–491. March 2005. doi:10.1016/j.pbb.2005.01.004. PMID 15740791. http://research.yerkes.emory.edu/Howell/PB&B80.pdf.
- ↑ 2.0 2.1 2.2 2.3 2.4 2.5 2.6 "Effects of dopamine transporter inhibitors on cocaine self-administration in rhesus monkeys: relationship to transporter occupancy determined by positron emission tomography neuroimaging". The Journal of Pharmacology and Experimental Therapeutics 309 (3): 959–969. June 2004. doi:10.1124/jpet.103.060293. PMID 14982963. http://research.yerkes.emory.edu/Howell/JPET309.pdf. Retrieved 2009-07-15.
- ↑ 3.0 3.1 "Faster onset and dopamine transporter selectivity predict stimulant and reinforcing effects of cocaine analogs in squirrel monkeys". Pharmacology, Biochemistry, and Behavior 86 (1): 45–54. January 2007. doi:10.1016/j.pbb.2006.12.006. PMID 17258302.
- ↑ "A reduced rate of in vivo dopamine transporter binding is associated with lower relative reinforcing efficacy of stimulants". Neuropsychopharmacology 31 (2): 351–362. February 2006. doi:10.1038/sj.npp.1300795. PMID 15957006.
- ↑ "The dopamine transporter and cocaine medication development: drug self-administration in nonhuman primates". The Journal of Pharmacology and Experimental Therapeutics 298 (1): 1–6. July 2001. doi:10.1016/S0022-3565(24)29344-7. PMID 11408518. http://research.yerkes.emory.edu/Howell/JPET298.pdf.
 |
