Chemistry:7α-Hydroxy-DHEA
| |
| Names | |
|---|---|
| IUPAC name
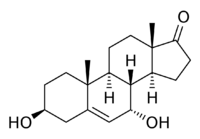
3β,7α-Dihydroxyandrost-5-ene-17-one
| |
| Systematic IUPAC name
(3aS,3bR,4S,7S,9aR,9bS,11aS)-4,7-Dihydroxy-9a,11a-dimethyl-2,3,3a,3b,4,6,7,8,9,9a,9b,10,11,11a-tetradecahydro-1H-cyclopenta[a]phenanthren-1-one | |
| Other names
7α-OH-DHEA; Androst-5-en-3β,7α-diol-17-one
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| KEGG | |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C19H28O3 | |
| Molar mass | 304.430 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
7α-Hydroxydehydroepiandrosterone (7α-hydroxy-DHEA; 7α-OH-DHEA), also known as 3β,7α-dihydroxyandrost-5-ene-17-one, is an endogenous, naturally occurring steroid and a major metabolite of dehydroepiandrosterone (DHEA) that is formed by CYP7B1 (steroid 7α-hydroxylase) in tissues such as the prostate gland and by CYP3A4 in the liver.[1][2] The major metabolic pathway of DHEA outside the liver is via 7-hydroxylation into 7α-OH-DHEA and 7β-OH-DHEA.[3] 7α-OH-DHEA has weak estrogenic activity, selectively activating the estrogen receptor ERβ.[2] In addition, 7α-OH-DHEA may be responsible for the known antiglucocorticoid effects of DHEA.[4][5]
Serum levels of 7α-OH-DHEA have been found to be significantly elevated in patients with Alzheimer's disease.[4] It is unclear what significance this may have, if any.[6]
7α-OH-DHEA is on the World Anti-Doping Agency list of prohibited substances in sporting.[7]
See also
References
- ↑ "Metabocard for 7α-Hydroxydehydroepiandrosterone (HMDB04611)". Human Metabolome Database. http://www.hmdb.ca/metabolites/HMDB04611.
- ↑ 2.0 2.1 "DHEA metabolites activate estrogen receptors alpha and beta". Steroids 78 (1): 15–25. 2013. doi:10.1016/j.steroids.2012.10.002. PMID 23123738.
- ↑ "Synthesis of 7alpha-hydroxy-dehydroepiandrosterone and 7beta-hydroxy-dehydroepiandrosterone". Steroids 70 (14): 970–3. 2005. doi:10.1016/j.steroids.2005.07.006. PMID 16143359. "he major metabolic pathway for DHEA in extra-hepatic tissues is via 7-hydroxylation [18], [19] and [20].".
- ↑ 4.0 4.1 "Increased total 7 alpha-hydroxy-dehydroepiandrosterone in serum of patients with Alzheimer's disease". J. Gerontol. A Biol. Sci. Med. Sci. 53 (2): B125–32. 1998. doi:10.1093/gerona/53a.2.b125. PMID 9520908.
- ↑ Neurosteroids and Brain Function. Academic Press. 12 December 2001. pp. 84–. ISBN 978-0-08-054423-6. https://books.google.com/books?id=BJumUEbiaPYC&pg=PA84.
- ↑ Ronald Ross Watson (22 July 2011). DHEA in Human Health and Aging. CRC Press. pp. 437–. ISBN 978-1-4398-3883-9. https://books.google.com/books?id=COZH22RYMT4C&pg=PA437.
- ↑ "World Anti-Doping Agency Prohibited List 2019". https://www.wada-ama.org/sites/default/files/wada_2019_english_prohibited_list.pdf.
External links
 |

