Chemistry:Diazepam
 | |
 | |
| Clinical data | |
|---|---|
| Pronunciation | /daɪˈæzɪpæm/ dy-AZ-ip-am |
| Trade names | Valium, Vazepam, Valtoco, others[1] |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a682047 |
| License data | |
| Pregnancy category |
|
| Dependence liability | High[3] |
| Addiction liability | Moderate[4][5] |
| Routes of administration | oral, intramuscular, intravenous, rectal, nasal[6] |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | 76% (64–97%) oral, 81% (62–98%) rectal[8] |
| Metabolism | Liver – CYP2B6 (minor route) to desmethyldiazepam, CYP2C19 (major route) to inactive metabolites, CYP3A4 (major route) to desmethyldiazepam |
| Elimination half-life | (50 h); 20–100 h (32–200 h for main active metabolite desmethyldiazepam)[7][9] |
| Excretion | Kidney |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| Chemical and physical data | |
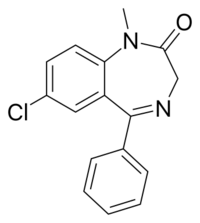
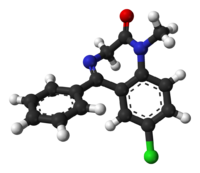
| Formula | C16H13ClN2O |
| Molar mass | 284.74 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Diazepam, first marketed as Valium, is a medicine of the benzodiazepine family that acts as an anxiolytic.[10] It is commonly used to treat a range of conditions, including anxiety, seizures, alcohol withdrawal syndrome, muscle spasms, insomnia, and restless legs syndrome.[10] It may also be used to cause memory loss during certain medical procedures.[11][12] It can be taken orally (by mouth), as a suppository inserted into the rectum, intramuscularly (injected into muscle), intravenously (injection into a vein) or used as a nasal spray.[6][12] When injected intravenously, effects begin in one to five minutes and last up to an hour.[12] Orally, effects begin after 15 to 60 minutes.[13]
Common side effects include sleepiness and trouble with coordination.[7][12] Serious side effects are rare.[10] They include increased risk of suicide, decreased breathing, and an increased risk of seizures if used too frequently in those with epilepsy.[10][12][14] Occasionally, excitement or agitation may occur.[15][16] Long-term use can result in tolerance, dependence, and withdrawal symptoms on dose reduction.[10] Abrupt stopping after long-term use can be potentially dangerous.[10] After stopping, cognitive problems may persist for six months or longer.[15] It is not recommended during pregnancy or breastfeeding.[12] Its mechanism of action works by increasing the effect of the neurotransmitter gamma-aminobutyric acid (GABA).[15]
Diazepam was patented in 1959 by Hoffmann-La Roche.[10][17][18] It has been one of the most frequently prescribed medications in the world since its launch in 1963.[10] In the United States it was the best-selling medication between 1968 and 1982, selling more than 2 billion tablets in 1978 alone.[10] In 2020, it was the 128th most commonly prescribed medication in the United States, with more than 4 million prescriptions.[19][20] In 1985 the patent ended, and there are now more than 500 brands available on the market.[10] It is on the World Health Organization's List of Essential Medicines.[21]
Medical uses
Diazepam is mainly used to treat anxiety, insomnia, panic attacks and symptoms of acute alcohol withdrawal. It is also used as a premedication for inducing sedation, anxiolysis, or amnesia before certain medical procedures (e.g., endoscopy).[22][23] In 2020, it was approved for use in the United States as a nasal spray to interrupt seizure activity in people with epilepsy.[6][24] Diazepam is the most commonly used benzodiazepine for "tapering" benzodiazepine dependence due to the drug's comparatively long half-life, allowing for more efficient dose reduction. Benzodiazepines have a relatively low toxicity in overdose.[15]
Diazepam has a number of uses, including:
- Treatment of anxiety, panic attacks, and states of agitation[22][25]
- Treatment of neurovegetative symptoms associated with vertigo[26]
- Treatment of the symptoms of alcohol, opiate, and benzodiazepine withdrawal[22][27]
- Short-term treatment of insomnia[22]
- Treatment of muscle spasms
- Treatment of tetanus, together with other measures of intensive treatment[28]
- Adjunctive treatment of spastic muscular paresis (paraplegia/tetraplegia) caused by cerebral or spinal cord conditions such as stroke, multiple sclerosis, or spinal cord injury (long-term treatment is coupled with other rehabilitative measures)[29]
- Palliative treatment of stiff person syndrome[30]
- Pre- or postoperative sedation, anxiolysis or amnesia (e.g., before endoscopic or surgical procedures)[29]
- Treatment of complications with stimulant overdoses and psychosis, such as cocaine or methamphetamine[31]
Used in treatment of organophosphate poisoning and reduces the risk of seizure induced brain and cardiac damage.
- Preventive treatment of oxygen toxicity during hyperbaric oxygen therapy[32]
Dosages should be determined on an individual basis, depending on the condition being treated, severity of symptoms, patient body weight, and any other conditions the person may have.[31]
Seizures
Intravenous diazepam or lorazepam are first-line treatments for status epilepticus.[15][33] However, intravenous lorazepam has advantages over intravenous diazepam, including a higher rate of terminating seizures and a more prolonged anticonvulsant effect. Diazepam gel was better than placebo gel in reducing the risk of non-cessation of seizures.[34] Diazepam is rarely used for the long-term treatment of epilepsy because tolerance to its anticonvulsant effects usually develops within six to twelve months of treatment, effectively rendering it useless for that purpose.[31][35]
The anticonvulsant effects of diazepam can help in the treatment of seizures due to a drug overdose or chemical toxicity as a result of exposure to sarin, VX, or soman (or other organophosphate poisons), lindane, chloroquine, physostigmine, or pyrethroids.[31][36]
Diazepam is sometimes used intermittently for the prevention of febrile seizures that may occur in children under five years of age.[15] Recurrence rates are reduced, but side effects are common so the decision to treat febrile seizures (which are benign in nature) with medication should use this as part of the evaluation.[37] Long-term use of diazepam for the management of epilepsy is not recommended; however, a subgroup of individuals with treatment-resistant epilepsy benefit from long-term benzodiazepines, and for such individuals, clorazepate has been recommended due to its slower onset of tolerance to the anticonvulsant effects.[15]
Alcohol withdrawal
Because of its relatively long duration of action, and evidence of safety and efficacy, diazepam is preferred over other benzodiazepines for treatment of persons experiencing moderate to severe alcohol withdrawal.[38] An exception to this is when a medication is required intramuscular in which case either lorazepam or midazolam is recommended.[38]
Other
Diazepam is used for the emergency treatment of eclampsia, when IV magnesium sulfate and blood-pressure control measures have failed.[39][40] Benzodiazepines do not have any pain-relieving properties themselves, and are generally recommended to avoid in individuals with pain.[41] However, benzodiazepines such as diazepam can be used for their muscle-relaxant properties to alleviate pain caused by muscle spasms and various dystonias, including blepharospasm.[42][43] Tolerance often develops to the muscle relaxant effects of benzodiazepines such as diazepam.[44] Baclofen is sometimes used as an alternative to diazepam.[45]
Availability
Diazepam is marketed in over 500 brands throughout the world.[46] It is supplied in oral, injectable, inhalation, and rectal forms.[31][47][48]
The United States military employs a specialized diazepam preparation known as Convulsive Antidote, Nerve Agent (CANA), which contains diazepam. One CANA kit is typically issued to service members, along with three Mark I NAAK kits, when operating in circumstances where chemical weapons in the form of nerve agents are considered a potential hazard. Both of these kits deliver drugs using autoinjectors. They are intended for use in "buddy aid" or "self aid" administration of the drugs in the field prior to decontamination and delivery of the patient to definitive medical care.[49]
Contraindications
Use of diazepam should be avoided, when possible, in individuals with:[50]
- Ataxia
- Severe hypoventilation
- Acute narrow-angle glaucoma
- Severe hepatic deficiencies (hepatitis and liver cirrhosis decrease elimination by a factor of two)
- Severe renal deficiencies (for example, patients on dialysis)
- Liver disorders
- Severe sleep apnea
- Severe depression, particularly when accompanied by suicidal tendencies
- Psychosis
- Pregnancy or breast feeding[51]
- Caution required in elderly or debilitated patients
- Coma or shock
- Abrupt discontinuation of therapy
- Acute intoxication with alcohol, narcotics, or other psychoactive substances (with the exception of hallucinogens or some stimulants, where it is occasionally used as a treatment for overdose)
- History of alcohol or drug dependence
- Myasthenia gravis, an autoimmune disorder causing marked fatiguability
- Hypersensitivity or allergy to any drug in the benzodiazepine class
Caution
- Benzodiazepine abuse and misuse should be guarded against when prescribed to those with alcohol or drug dependencies or who have psychiatric disorders.[52]
- Pediatric patients
- Less than 18 years of age, this treatment is usually not indicated, except for treatment of epilepsy, and pre- or postoperative treatment. The smallest possible effective dose should be used for this group of patients.[53]
- Under 6 months of age, safety and effectiveness have not been established; diazepam should not be given to those in this age group.[30][53]
- Elderly and very ill patients can possibly experience apnea or cardiac arrest. Concomitant use of other central nervous system depressants increases this risk. The smallest possible effective dose should be used for this group of people.[53][54] The elderly metabolise benzodiazepines much more slowly than younger adults, and are also more sensitive to the effects of benzodiazepines, even at similar blood plasma levels. Doses of diazepam are recommended to be about half of those given to younger people, and treatment limited to a maximum of two weeks. Long-acting benzodiazepines such as diazepam are not recommended for the elderly.[15] Diazepam can also be dangerous in geriatric patients owing to a significant increased risk of falls.[55]
- Intravenous or intramuscular injections in hypotensive people or those in shock should be administered carefully and vital signs should be monitored.[54]
- Benzodiazepines such as diazepam are lipophilic and rapidly penetrate membranes, so rapidly cross over into the placenta with significant uptake of the drug. Use of benzodiazepines including diazepam in late pregnancy, especially high doses, can result in floppy infant syndrome.[56] Diazepam when taken late in pregnancy, during the third trimester, causes a definite risk of a severe benzodiazepine withdrawal syndrome in the neonate with symptoms including hypotonia, and reluctance to suck, to apnoeic spells, cyanosis, and impaired metabolic responses to cold stress. Floppy infant syndrome and sedation in the newborn may also occur. Symptoms of floppy infant syndrome and the neonatal benzodiazepine withdrawal syndrome have been reported to persist from hours to months after birth.[57]
Adverse effects
Benzodiazepines such as diazepam can cause anterograde amnesia, confusion, and sedation. The elderly are more prone to diazepam's confusion, amnesia, ataxia, hangover symptoms, and falls. Long-term use of benzodiazepines such as diazepam induces tolerance, dependency, and withdrawal syndrome.[15] Like other benzodiazepines, diazepam impairs short-term memory and learning new information. Diazepam and other benzodiazepines can produce anterograde amnesia, but not retrograde amnesia. It means information learned before using benzodiazepines is not impaired. Short-term benzodiazepine use does not lead to tolerance, and the elderly are more sensitive to them.[58] Additionally, after stopping benzodiazepines, cognitive problems may last at least six months; it is unclear if these problems last for longer than six months or are permanent. Benzodiazepines may also cause or worsen depression.[15] Infusions or repeated intravenous injections of diazepam when managing seizures, for example, may lead to drug toxicity, including respiratory depression, sedation and hypotension. Drug tolerance may also develop to infusions of diazepam if it is given for longer than 24 hours.[15] Sedatives and sleeping pills, including diazepam, have been associated with an increased risk of death.[59]
In September 2020, the U.S. Food and Drug Administration (FDA) required the boxed warning be updated for all benzodiazepine medicines to describe the risks of abuse, misuse, addiction, physical dependence, and withdrawal reactions consistently across all the medicines in the class.[60]
Diazepam has a range of side effects common to most benzodiazepines, including:
- Suppression of REM sleep and slow wave sleep
- Impaired motor function
- Impaired coordination
- Impaired balance
- Dizziness
- Reflex tachycardia[61]
Less commonly, paradoxical reactions can occur, including nervousness, irritability, excitement, worsening of seizures, insomnia, muscle cramps, changes in libido, and in some cases, rage and violence. These adverse reactions are more likely to occur in children, the elderly, and individuals with a history of a substance use disorder, such as an alcohol use disorder, or a history of aggressive behavior.[15][62][63][64] In some people, diazepam may increase the propensity toward self-harming behavior and, in extreme cases, may provoke suicidal tendencies or acts.[65] Very rarely dystonia can occur.[66]
Diazepam may impair the ability to drive vehicles or operate machinery. The impairment is worsened by consumption of alcohol, because both act as central nervous system depressants.[30]
During the course of therapy, tolerance to the sedative effects usually develops, but not to the anxiolytic and myorelaxant effects.[67]
Patients with severe attacks of apnea during sleep may experience respiratory depression (hypoventilation), leading to respiratory arrest and death.[68]
Diazepam in doses of 5 mg or more causes significant deterioration in alertness performance combined with increased feelings of sleepiness.[69]
Tolerance and withdrawal
Diazepam, as with other benzodiazepine drugs, can cause tolerance, physical dependence, substance use disorder, and benzodiazepine withdrawal syndrome. Withdrawal from diazepam or other benzodiazepines often leads to withdrawal symptoms similar to those seen during barbiturate or alcohol withdrawal. The higher the dose and the longer the drug is taken, the greater the risk of experiencing unpleasant withdrawal symptoms.[70]
Withdrawal symptoms can occur from standard dosages and also after short-term use, and can range from insomnia and anxiety to more serious symptoms, including seizures and psychosis. Withdrawal symptoms can sometimes resemble pre-existing conditions and be misdiagnosed. Diazepam may produce less intense withdrawal symptoms due to its long elimination half-life.[38]
Benzodiazepine treatment should be discontinued as soon as possible by a slow and gradual dose reduction regimen.[15][71] Tolerance develops to the therapeutic effects of benzodiazepines; for example tolerance occurs to the anticonvulsant effects and as a result benzodiazepines are not generally recommended for the long-term management of epilepsy. Dose increases may overcome the effects of tolerance, but tolerance may then develop to the higher dose and adverse effects may increase. The mechanism of tolerance to benzodiazepines includes uncoupling of receptor sites, alterations in gene expression, down-regulation of receptor sites, and desensitisation of receptor sites to the effect of GABA. About one-third of individuals who take benzodiazepines for longer than four weeks become dependent and experience withdrawal syndrome on cessation.[15]
Differences in rates of withdrawal (50–100%) vary depending on the patient sample. For example, a random sample of long-term benzodiazepine users typically finds around 50% experience few or no withdrawal symptoms, with the other 50% experiencing notable withdrawal symptoms. Certain select patient groups show a higher rate of notable withdrawal symptoms, up to 100%.[72]
Rebound anxiety, more severe than baseline anxiety, is also a common withdrawal symptom when discontinuing diazepam or other benzodiazepines.[73] Diazepam is therefore only recommended for short-term therapy at the lowest possible dose owing to risks of severe withdrawal problems from low doses even after gradual reduction.[74] The risk of pharmacological dependence on diazepam is significant, and patients experience symptoms of benzodiazepine withdrawal syndrome if it is taken for six weeks or longer.[75] In humans, tolerance to the anticonvulsant effects of diazepam occurs frequently.[76]
Dependence
Improper or excessive use of diazepam can lead to dependence. At a particularly high risk for diazepam misuse, substance use disorder or dependence are:
- People with a history of a substance use disorder or substance dependence.[30][77] Diazepam increases craving for alcohol in problem alcohol consumers. Diazepam also increases the volume of alcohol consumed by problem drinkers.[78]
- People with severe personality disorders, such as borderline personality disorder[79]
Patients from the aforementioned groups should be monitored very closely during therapy for signs of abuse and development of dependence. Therapy should be discontinued if any of these signs are noted, although if dependence has developed, therapy must still be discontinued gradually to avoid severe withdrawal symptoms. Long-term therapy in such instances is not recommended.[30][77]
People suspected of being dependent on benzodiazepine drugs should be very gradually tapered off the drug. Withdrawals can be life-threatening, particularly when excessive doses have been taken for extended periods of time. Equal prudence should be used whether dependence has occurred in therapeutic or recreational contexts.[80]
Diazepam is a good choice for tapering for those using high doses of other benzodiazepines since it has a long half-life thus withdrawal symptoms are tolerable.[81] The process is very slow (usually from 14 to 28 weeks) but is considered safe when done appropriately.[82]
Overdose
An individual who has consumed too much diazepam typically displays one or more of these symptoms in a period of approximately four hours immediately following a suspected overdose:[30][83]
- Drowsiness
- Mental confusion
- Hypotension
- Impaired motor function
- Impaired reflexes
- Impaired coordination
- Impaired balance
- Dizziness
- Coma
Although not usually fatal when taken alone, a diazepam overdose is considered a medical emergency and generally requires the immediate attention of medical personnel. The antidote for an overdose of diazepam (or any other benzodiazepine) is flumazenil (Anexate). This drug is only used in cases with severe respiratory depression or cardiovascular complications. Because flumazenil is a short-acting drug, and the effects of diazepam can last for days, several doses of flumazenil may be necessary. Artificial respiration and stabilization of cardiovascular functions may also be necessary. Though not routinely indicated, activated charcoal can be used for decontamination of the stomach following a diazepam overdose. Emesis is contraindicated. Dialysis is minimally effective. Hypotension may be treated with levarterenol or metaraminol.[30][31][83][84]
The oral -1">50 (lethal dose in 50% of the population) of diazepam is 720 mg/kg in mice and 1240 mg/kg in rats.[30] D. J. Greenblatt and colleagues reported in 1978 on two patients who had taken 500 mg and 2000 mg of diazepam, respectively, went into moderately-deep comas, and were discharged within 48 hours without having experienced any important complications, in spite of having high concentrations of diazepam and its metabolites desmethyldiazepam, oxazepam, and temazepam, according to samples taken in the hospital and as follow-up.[85]
Overdoses of diazepam with alcohol, opiates, or other depressants may be fatal.[84][86]
Interactions
If diazepam is administered concomitantly with other drugs, attention should be paid to the possible pharmacological interactions. Particular care should be taken with drugs that potentiate the effects of diazepam, such as barbiturates, phenothiazines, opioids, and antidepressants.[30]
Diazepam does not increase or decrease hepatic enzyme activity, and does not alter the metabolism of other compounds. No evidence would suggest diazepam alters its own metabolism with chronic administration.[31]
Agents with an effect on hepatic cytochrome P450 pathways or conjugation can alter the rate of diazepam metabolism. These interactions would be expected to be most significant with long-term diazepam therapy, and their clinical significance is variable.[31]
- Diazepam increases the central depressive effects of alcohol, other hypnotics/sedatives (e.g., barbiturates), other muscle relaxants, certain antidepressants, sedative antihistamines, opioids, and antipsychotics, as well as anticonvulsants such as phenobarbital, phenytoin, and carbamazepine. The euphoriant effects of opioids may be increased, leading to increased risk of psychological dependence.[15][53][87]
- Cimetidine, omeprazole, oxcarbazepine, ticlopidine, topiramate, ketoconazole, itraconazole, disulfiram, fluvoxamine, isoniazid, erythromycin, probenecid, propranolol, imipramine, ciprofloxacin, fluoxetine, and valproic acid prolong the action of diazepam by inhibiting its elimination.[15][31][47]
- Alcohol in combination with diazepam may cause a synergistic enhancement of the hypotensive properties of benzodiazepines and alcohol.[88]
- Oral contraceptives significantly decrease the elimination of desmethyldiazepam, a major metabolite of diazepam.[53][89]
- Rifampin, phenytoin, carbamazepine, and phenobarbital increase the metabolism of diazepam, thus decreasing drug levels and effects.[31] Dexamethasone and St John's wort also increase the metabolism of diazepam.[15]
- Diazepam increases the serum levels of phenobarbital.[90]
- Nefazodone can cause increased blood levels of benzodiazepines.[53]
- Cisapride may enhance the absorption, and therefore the sedative activity, of diazepam.[91]
- Small doses of theophylline may inhibit the action of diazepam.[92]
- Diazepam may block the action of levodopa (used in the treatment of Parkinson's disease).[87]
- Diazepam may alter digoxin serum concentrations.[31]
- Other drugs that may have interactions with diazepam include antipsychotics (e.g. chlorpromazine), MAO inhibitors, and ranitidine.[53]
- Because it acts on the GABA receptor, the herb valerian may produce an adverse effect.[93]
- Foods that acidify the urine can lead to faster absorption and elimination of diazepam, reducing drug levels and activity.[87]
- Foods that alkalinize the urine can lead to slower absorption and elimination of diazepam, increasing drug levels and activity.[31]
- Reports conflict as to whether food in general has any effects on the absorption and activity of orally administered diazepam.[87]
Pharmacology
Diazepam is a long-acting "classical" benzodiazepine. Other classical benzodiazepines include chlordiazepoxide, clonazepam, lorazepam, oxazepam, nitrazepam, temazepam, flurazepam, bromazepam, and clorazepate.[94] Diazepam has anticonvulsant properties.[95] Benzodiazepines act via micromolar benzodiazepine binding sites as calcium channel blockers and significantly inhibit depolarization-sensitive calcium uptake in rat nerve cell preparations.[96]
Diazepam inhibits acetylcholine release in mouse hippocampal synaptosomes. This has been found by measuring sodium-dependent high-affinity choline uptake in mouse brain cells in vitro, after pretreatment of the mice with diazepam in vivo. This may play a role in explaining diazepam's anticonvulsant properties.[97]
Diazepam binds with high affinity to glial cells in animal cell cultures.[98] Diazepam at high doses has been found to decrease histamine turnover in mouse brain via diazepam's action at the benzodiazepine-GABA receptor complex.[99] Diazepam also decreases prolactin release in rats.[100]
Mechanism of action
Benzodiazepines are positive allosteric modulators of the GABA type A receptors (GABAA). The GABAA receptors are ligand-gated chloride-selective ion channels that are activated by GABA, the major inhibitory neurotransmitter in the brain. Binding of benzodiazepines to this receptor complex promotes the binding of GABA, which in turn increases the total conduction of chloride ions across the neuronal cell membrane. This increased chloride ion influx hyperpolarizes the neuron's membrane potential. As a result, the difference between resting potential and threshold potential is increased and firing is less likely. As a result, the arousal of the cortical and limbic systems in the central nervous system is reduced.[1]
The GABAA receptor is a heteromer composed of five subunits, the most common ones being two αs, two βs, and one γ (α2β2γ). For each subunit, many subtypes exist (α1–6, β1–3, and γ1–3). GABAA receptors containing the α1 subunit mediate the sedative, the anterograde amnesic, and partly the anticonvulsive effects of diazepam. GABAA receptors containing α2 mediate the anxiolytic actions and to a large degree the myorelaxant effects. GABAA receptors containing α3 and α5 also contribute to benzodiazepines myorelaxant actions, whereas GABAA receptors comprising the α5 subunit were shown to modulate the temporal and spatial memory effects of benzodiazepines.[101] Diazepam is not the only drug to target these GABAA receptors. Drugs such as flumazenil also bind to GABAA to induce their effects.[102]
Diazepam appears to act on areas of the limbic system, thalamus, and hypothalamus, inducing anxiolytic effects. Benzodiazepine drugs including diazepam increase the inhibitory processes in the cerebral cortex.[103]
The anticonvulsant properties of diazepam and other benzodiazepines may be in part or entirely due to binding to voltage-dependent sodium channels rather than GABAA receptors. Sustained repetitive firing seems limited by benzodiazepines' effect of slowing recovery of sodium channels from inactivation.[104]
The muscle relaxant properties of diazepam are produced via inhibition of polysynaptic pathways in the spinal cord.[105]
Pharmacokinetics
Diazepam can be administered orally, intravenously (must be diluted, as it is painful and damaging to veins), intramuscularly (IM), or as a suppository.[31]
The onset of action is one to five minutes for IV administration and 15–30 minutes for IM administration. The duration of diazepam's peak pharmacological effects is 15 minutes to one hour for both routes of administration.[61] The half-life of diazepam in general is 30–56 hours.[1] Peak plasma levels occur between 30 and 90 minutes after oral administration and between 30 and 60 minutes after intramuscular administration; after rectal administration, peak plasma levels occur after 10 to 45 minutes. Diazepam is highly plasma protein-bound, with 96–99% of the absorbed drug being protein-bound. The distribution half-life of diazepam is two to 13 minutes.[15]
Diazepam is highly lipid-soluble, and is widely distributed throughout the body after administration. It easily crosses both the blood–brain barrier and the placenta, and is excreted into breast milk. After absorption, diazepam is redistributed into muscle and adipose tissue. Continual daily doses of diazepam quickly build to a high concentration in the body (mainly in adipose tissue), far in excess of the actual dose for any given day.[15][31]
Diazepam is stored preferentially in some organs, including the heart. Absorption by any administered route and the risk of accumulation is significantly increased in the neonate, and withdrawal of diazepam during pregnancy and breast feeding is clinically justified.[106]
Diazepam undergoes oxidative metabolism by demethylation (CYP2C9, 2C19, 2B6, 3A4, and 3A5), hydroxylation (CYP3A4 and 2C19) and glucuronidation in the liver as part of the cytochrome P450 enzyme system. It has several pharmacologically active metabolites. The main active metabolite of diazepam is desmethyldiazepam (also known as nordazepam or nordiazepam). Its other active metabolites include the minor active metabolites temazepam and oxazepam. These metabolites are conjugated with glucuronide, and are excreted primarily in the urine. Because of these active metabolites, the serum values of diazepam alone are not useful in predicting the effects of the drug. Diazepam has a biphasic half-life of about one to three days, and two to seven days for the active metabolite desmethyldiazepam.[15] Most of the drug is metabolized; very little diazepam is excreted unchanged.[31] The elimination half-life of diazepam and also the active metabolite desmethyldiazepam increases significantly in the elderly, which may result in prolonged action, as well as accumulation of the drug during repeated administration.[107]
Physical and chemical properties
Diazepam is a 1,4-benzodiazepine.[1] Diazepam occurs as solid white or yellow crystals with a melting point of 131.5 to 134.5 °C. It is odorless, and has a slightly bitter taste. The British Pharmacopoeia lists it as being very slightly soluble in water, soluble in alcohol, and freely soluble in chloroform. The United States Pharmacopoeia lists diazepam as soluble 1 in 16 ethyl alcohol, 1 in 2 of chloroform, 1 in 39 ether, and practically insoluble in water. The pH of diazepam is neutral (i.e., pH = 7). Due to additives such as benzoic acid/benzoate in the injectable form.[clarification needed][108] Diazepam has a shelf life of five years for oral tablets and three years for IV/IM solutions.[31] Diazepam should be stored at room temperature (15–30 °C). The solution for parenteral injection should be protected from light and kept from freezing. The oral forms should be stored in air-tight containers and protected from light.[47]
Diazepam can absorb into plastics, so liquid preparations should not be kept in plastic bottles or syringes, etc. As such, it can leach into the plastic bags and tubing used for intravenous infusions. Absorption appears to depend on several factors, such as temperature, concentration, flow rates, and tube length. Diazepam should not be administered if a precipitate has formed and does not dissolve.[47]
Synthesis
Diazepam can be synthesized from nordazepam by methylating the R1 position using dimethyl sulfate.[109]
Detection in body fluids
Diazepam may be quantified in blood or plasma to confirm a diagnosis of poisoning in hospitalized patients, provide evidence in an impaired driving arrest, or to assist in a medicolegal death investigation. Blood or plasma diazepam concentrations are usually in a range of 0.1–1.0 mg/L in persons receiving the drug therapeutically. Most commercial immunoassays for the benzodiazepine class of drugs cross-react with diazepam, but confirmation and quantitation are usually performed using chromatographic techniques.[110][111][112]
Environmental
Diazepam is a common environmental contamination finding near human settlement.[113]
History
Diazepam was the second benzodiazepine invented by Leo Sternbach of Hoffmann-La Roche at the company's Nutley, New Jersey, facility[114] following chlordiazepoxide (Librium), which was approved for use in 1960. Released in 1963 as an improved version of Librium, diazepam became incredibly popular, helping Roche to become a pharmaceutical industry giant. It is 2.5 times more potent than its predecessor, which it quickly surpassed in terms of sales. After this initial success, other pharmaceutical companies began to introduce other benzodiazepine derivatives.[115]
The benzodiazepines gained popularity among medical professionals as an improvement over barbiturates, which have a comparatively narrow therapeutic index, and are far more sedative at therapeutic doses. The benzodiazepines are also far less dangerous; death rarely results from diazepam overdose, except in cases where it is consumed with large amounts of other depressants (such as alcohol or opioids).[84] Benzodiazepine drugs such as diazepam initially had widespread public support, but with time the view changed to one of growing criticism and calls for restrictions on their prescription.[116]
Marketed by Roche using an advertising campaign conceived by the William Douglas McAdams Agency under the leadership of Arthur Sackler,[117] diazepam was the top-selling pharmaceutical in the United States from 1969 to 1982, with peak annual sales in 1978 of 2.3 billion tablets.[115] Diazepam, along with oxazepam, nitrazepam and temazepam, represents 82% of the benzodiazepine market in Australia.[118] While psychiatrists continue to prescribe diazepam for the short-term relief of anxiety, neurology has taken the lead in prescribing diazepam for the palliative treatment of certain types of epilepsy and spastic activity, for example, forms of paresis. [citation needed] It is also the first line of defense for a rare disorder called stiff-person syndrome.[29]
Society and culture
Recreational use
Diazepam is a medication with a high risk of misuse and can cause drug dependence. Urgent action by national governments has been recommended to improve prescribing patterns of benzodiazepines such as diazepam.[119][120] A single dose of diazepam modulates the dopamine system in similar ways to how morphine and alcohol modulate the dopaminergic pathways.[121] Between 50 and 64% of rats will self-administer diazepam.[122] Diazepam has been shown to be able to substitute for the behavioral effects of barbiturates in a primate study.[123] Diazepam has been found as an adulterant in heroin.[124]
Diazepam drug misuse can occur either through recreational misuse where the drug is taken to achieve a high or when the drug is continued long term against medical advice.[125]
Sometimes, it is used by stimulant users to "come down" and sleep and to help control the urge to binge. These users often escalate dosage from 2 to 25 times the therapeutic dose of 5 mg to 10 mg.[126]
A large-scale study in the US, conducted by SAMHSA, using data from 2011, determined benzodiazepines were present in 28.7% of emergency department visits involving nonmedical use of pharmaceuticals. In this regard, benzodiazepines are second only to opiates, the study found in 39.2% of visits. About 29.3% of drug-related suicide attempts involve benzodiazepines, making them the most frequently represented class in drug-related suicide attempts. Males misuse benzodiazepines as commonly as females.[127]
Diazepam was detected in 26% of cases of people suspected of driving under the influence of drugs in Sweden, and its active metabolite nordazepam was detected in 28% of cases. Other benzodiazepines and zolpidem and zopiclone also were found in high numbers. Many drivers had blood levels far exceeding the therapeutic dose range, suggesting a high degree of potential for misuse for benzodiazepines, zolpidem, and zopiclone.[110] In Northern Ireland, in cases where drugs were detected in samples from impaired drivers who were not impaired by alcohol, benzodiazepines were found in 87% of cases. Diazepam was the most commonly detected benzodiazepine.[128]
Legal status
Diazepam is regulated as a prescription medication:
International
Diazepam is a Schedule IV controlled drug under the Convention on Psychotropic Substances.[129]
UK
Classified as a controlled drug, listed under Schedule IV, Part I (CD Benz POM) of the Misuse of Drugs Regulations 2001, allowing possession with a valid prescription. The Misuse of Drugs Act 1971 makes it illegal to possess the drug without a prescription, and for such purposes it is classified as a Class C drug.[130]
Germany
Classified as a prescription drug, or in high dosage as a restricted drug (Betäubungsmittelgesetz, Anlage III).[131]
Australia
Diazepam is a Schedule 4 substance under the Poisons Standard (June 2018).[132] A Schedule 4 drug is outlined in the Poisons Act 1964 as, "Substances, the use or supply of which should be by or on the order of persons permitted by State or Territory legislation to prescribe and should be available from a pharmacist on prescription".[132]
United States
Diazepam is controlled as a Schedule IV substance under the Controlled Substances Act of 1970.
Judicial executions
The states of California and Florida offer diazepam to condemned inmates as a pre-execution sedative as part of their lethal injection program, although the state of California has not executed a prisoner since 2006.[133][134] In August 2018, Nebraska used diazepam as part of the drug combination used to execute Carey Dean Moore, the first death row inmate executed in Nebraska in over 21 years.[135]
Veterinary uses
Diazepam is used as a short-term sedative and anxiolytic for cats and dogs,[136] sometimes used as an appetite stimulant.[136][137] It can also be used to stop seizures in dogs and cats.[138]
References
- ↑ 1.0 1.1 1.2 1.3 1.4 "National Highway Traffic Safety Administration Drugs and Human Performance Fact Sheet- Diazepam". https://one.nhtsa.gov/people/injury/research/job185drugs/diazepam.htm.
- ↑ 2.0 2.1 "Valium". 31 January 2020. https://www.nps.org.au/medicine-finder/valium-tablets#full-pi.
- ↑ Pharmacology for the Primary Care Provider (4th ed.). Mosby. 2013. p. 545. ISBN 9780323087902. https://books.google.com/books?id=8FwdEsvnw8oC&pg=PA545.
- ↑ Clinical Addiction Psychiatry. Cambridge University Press. 2010. p. 156. ISBN 9781139491693. https://books.google.com/books?id=xD-R7Z27UBEC&pg=PA156.
- ↑ Principles of addiction medicine (4 ed.). Philadelphia: Wolters Kluwer/Lippincott Williams & Wilkins. 2009. p. 106. ISBN 9780781774772. https://books.google.com/books?id=j6GGBud8DXcC&pg=PA106.
- ↑ 6.0 6.1 6.2 "Valtoco – diazepam spray". 13 January 2020. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=1a8bcc90-68fa-474d-832c-0df01e825f39.
- ↑ 7.0 7.1 7.2 "Valium – diazepam tablet". 8 November 2019. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=554baee5-b171-4452-a50a-41a0946f956c.
- ↑ "Bioavailability of diazepam after intravenous, oral and rectal administration in adult epileptic patients". British Journal of Clinical Pharmacology 13 (3): 427–32. March 1982. doi:10.1111/j.1365-2125.1982.tb01397.x. PMID 7059446.
- ↑ "Diazepam Tablets BP 10 mg – Summary of Product Characteristics (SmPC)". 16 September 2019. https://www.medicines.org.uk/emc/product/4522/smpc.
- ↑ 10.0 10.1 10.2 10.3 10.4 10.5 10.6 10.7 10.8 10.9 "Classics in chemical neuroscience: diazepam (valium)". ACS Chemical Neuroscience 5 (4): 253–60. April 2014. doi:10.1021/cn5000056. PMID 24552479.
- ↑ "Diazepam". PubChem. National Institute of Health: National Library of Medicine. 2006. https://pubchem.ncbi.nlm.nih.gov/compound/3016.
- ↑ 12.0 12.1 12.2 12.3 12.4 12.5 "Diazepam". The American Society of Health-System Pharmacists. https://www.drugs.com/monograph/diazepam.html.
- ↑ "Diazepam". StatPearls [Internet]. StatPearls Publishing. September 2022. http://www.ncbi.nlm.nih.gov/books/NBK537022/. Retrieved 13 October 2019.
- ↑ "Prescribed Benzodiazepines and Suicide Risk: A Review of the Literature". The Primary Care Companion for CNS Disorders 19 (2). March 2017. doi:10.4088/PCC.16r02037. PMID 28257172.
- ↑ 15.00 15.01 15.02 15.03 15.04 15.05 15.06 15.07 15.08 15.09 15.10 15.11 15.12 15.13 15.14 15.15 15.16 15.17 15.18 15.19 "Benzodiazepines in epilepsy: pharmacology and pharmacokinetics". Acta Neurologica Scandinavica 118 (2): 69–86. August 2008. doi:10.1111/j.1600-0404.2008.01004.x. PMID 18384456.
- ↑ Pediatric hospital medicine : textbook of inpatient management (2nd ed.). Philadelphia: Wolters Kluwer Health/Lippincott Williams & Wilkins. 2008. p. 862. ISBN 9780781770323. https://books.google.com/books?id=sV6-ifUGoMYC&pg=PA862.
- ↑ Analogue-based Drug Discovery. John Wiley & Sons. 2006. p. 535. ISBN 9783527607495. https://books.google.com/books?id=FjKfqkaKkAAC&pg=PA535.
- ↑ & Earl Reeder"5-ARYL-3H-1,4-BENZODIAZEPIN-2(1H)-ONES" US patent 3371085, published 1968-02-27, issued 1968-02-27, assigned to Hoffmann La Roche AG
- ↑ "The Top 300 of 2020". https://clincalc.com/DrugStats/Top300Drugs.aspx.
- ↑ "Diazepam – Drug Usage Statistics". https://clincalc.com/DrugStats/Drugs/Diazepam.
- ↑ World Health Organization model list of essential medicines: 22nd list (2021). Geneva: World Health Organization. 2021. WHO/MHP/HPS/EML/2021.02.
- ↑ 22.0 22.1 22.2 22.3 "Diazepam". Drug Bank. http://redpoll.pharmacy.ualberta.ca/drugbank/cgi-bin/getCard.cgi?CARD=APRD00642.txt.
- ↑ "EFNS guideline on the diagnosis and management of alcohol-related seizures: report of an EFNS task force". European Journal of Neurology 12 (8): 575–81. August 2005. doi:10.1111/j.1468-1331.2005.01247.x. PMID 16053464.
- ↑ "FDA approves Valtoco". Drugs.com. 13 January 2020. https://www.drugs.com/newdrugs/fda-approves-valtoco-diazepam-nasal-seizure-rescue-5141.html.
- ↑ "Valium Tablets". https://www.nps.org.au/medicine-finder/valium-tablets.
- ↑ "The treatment of acute vertigo". Neurological Sciences 25 (Suppl 1): S26-30. March 2004. doi:10.1007/s10072-004-0213-8. PMID 15045617.
- ↑ "Withdrawing benzodiazepines in primary care". CNS Drugs 23 (1): 19–34. 2009. doi:10.2165/0023210-200923010-00002. PMID 19062773.
- ↑ "Diazepam for treating tetanus". The Cochrane Database of Systematic Reviews 2010 (1): CD003954. 2004. doi:10.1002/14651858.CD003954.pub2. PMID 14974046.
- ↑ 29.0 29.1 29.2 "Diazepam: indications". Rxlist.com. RxList Inc.. 24 January 2005. http://www.rxlist.com/cgi/generic/diazepam_ids.htm.
- ↑ 30.0 30.1 30.2 30.3 30.4 30.5 30.6 30.7 30.8 "Diazepam". Prescription Drug Information. Thomson Healthcare (Micromedex). March 2000. https://www.drugs.com/pdr/diazepam.html.
- ↑ 31.00 31.01 31.02 31.03 31.04 31.05 31.06 31.07 31.08 31.09 31.10 31.11 31.12 31.13 31.14 31.15 "Diazepam". Inchem.org. 1998. http://www.inchem.org/documents/pims/pharm/pim181.htm.
- ↑ Hyperbaric Medicine Practice (2nd ed.). Best Publishing Company. 1999. ISBN 978-0-941332-78-1.
- ↑ "Status epilepticus: an evidence based guide". BMJ 331 (7518): 673–7. September 2005. doi:10.1136/bmj.331.7518.673. PMID 16179702.
- ↑ "Anticonvulsant therapy for status epilepticus". The Cochrane Database of Systematic Reviews 2014 (9): CD003723. September 2014. doi:10.1002/14651858.CD003723.pub3. PMID 25207925.
- ↑ "Benzodiazepines in the treatment of epilepsy in people with intellectual disability". Journal of Intellectual Disability Research 42 (Suppl 1): 80–92. December 1998. PMID 10030438.
- ↑ Organophosphates/nerve agent poisoning: mechanism of action, diagnosis, prophylaxis, and treatment. Advances in Clinical Chemistry. 38. 2004. pp. 151–216. doi:10.1016/S0065-2423(04)38006-6. ISBN 978-0-12-010338-6.
- ↑ "Prophylactic drug management for febrile seizures in children". The Cochrane Database of Systematic Reviews 2021 (6): CD003031. June 2021. doi:10.1002/14651858.CD003031.pub4. PMID 34131913.
- ↑ 38.0 38.1 38.2 "Diazepam in the Treatment of Moderate to Severe Alcohol Withdrawal". CNS Drugs 31 (2): 87–95. February 2017. doi:10.1007/s40263-016-0403-y. PMID 28101764.
- ↑ "Neurologic aspects of eclampsia". Neurologic Clinics 22 (4): 841–61. November 2004. doi:10.1016/j.ncl.2004.07.005. PMID 15474770.
- ↑ "Evidence and practice: the magnesium sulphate story". Best Practice & Research. Clinical Obstetrics & Gynaecology 19 (1): 57–74. February 2005. doi:10.1016/j.bpobgyn.2004.10.010. PMID 15749066.
- ↑ "Subtype-selective GABAA receptor mimetics—novel antihyperalgesic agents?". Journal of Molecular Medicine 87 (5): 465–9. May 2009. doi:10.1007/s00109-009-0454-3. PMID 19259638. https://www.zora.uzh.ch/id/eprint/17933/10/ZORA_NL_17933.pdf.
- ↑ "[Therapy of dystonia in Japan]" (in ja). Rinsho Shinkeigaku = Clinical Neurology 45 (9): 634–42. September 2005. PMID 16248394.
- ↑ "[Medical treatment of dystonia]" (in ja). Rinsho Shinkeigaku = Clinical Neurology 41 (12): 1181–2. December 2001. PMID 12235832.
- ↑ "The diagnosis and management of benzodiazepine dependence". Current Opinion in Psychiatry 18 (3): 249–55. May 2005. doi:10.1097/01.yco.0000165594.60434.84. PMID 16639148. http://www.benzo.org.uk/amisc/ashdiag.pdf.
- ↑ "Diagnostic Algorithms for Neuromuscular Diseases". Clinics in Podiatric Medicine and Surgery 16 (1): 67–79. January 1999. doi:10.1016/S0891-8422(23)00935-7. PMID 9929772.
- ↑ "International AED Database". ILAE. http://www.ilae.org/Visitors/Centre/AEDs/index.cfm.
- ↑ 47.0 47.1 47.2 47.3 "Diazepam". The Elephant Formulary. Elephant Care International. 2005. http://www.elephantcare.org/Drugs/diazepam.htm.
- ↑ "Delivery of diazepam through an inhalation route". US Patent 6,805,853. PharmCast.com. 19 October 2004. http://www.pharmcast.com/Patents100/Yr2004/Oct2004/101904/6805853_Diazepam101904.htm.
- ↑ U.S. Army Medical Research Institute of Chemical Defense, Medical Management of Chemical Casualties Handbook, Third Edition (June 2000), Aberdeen Proving Ground, MD, pp. 118–126.
- ↑ Epocrates. "Diazepam Contraindications and Cautions". US: Epocrates Online. https://online.epocrates.com/u/103193/diazepam/Contraindications+Cautions.
- ↑ "Intra-uterine diazepam exposure decreases the number of catecholaminergic and serotoninergic neurons of neonate rats". Neuroscience Letters 795: 137014. December 2022. doi:10.1016/j.neulet.2022.137014. PMID 36521643.
- ↑ "Benzodiazepine dependence: focus on withdrawal syndrome". Annales Pharmaceutiques Françaises 67 (6): 408–13. November 2009. doi:10.1016/j.pharma.2009.07.001. PMID 19900604.
- ↑ 53.0 53.1 53.2 53.3 53.4 53.5 53.6 "Diazepam". PDRHealth.com. 2006. http://www.pdrhealth.com/drug_info/rxdrugprofiles/drugs/val1473.shtml.
- ↑ 54.0 54.1 "Diazepam: precautions". Rxlist.com. RxList Inc.. 24 January 2005. http://www.rxlist.com/cgi/generic/diazepam_wcp.htm.
- ↑ "[Falls in the geriatric department: responsibility of the care-giver and the hospital]" (in he). Harefuah 128 (11): 690–3, 743. June 1995. PMID 7557666.
- ↑ "Use of benzodiazepines during pregnancy, labour and lactation, with particular reference to pharmacokinetic considerations". Drugs 23 (5): 354–80. May 1982. doi:10.2165/00003495-198223050-00002. PMID 6124415.
- ↑ "The effects of benzodiazepine use during pregnancy and lactation". Reproductive Toxicology 8 (6): 461–75. 1994. doi:10.1016/0890-6238(94)90029-9. PMID 7881198.
- ↑ The American Psychiatric Publishing Textbook of Neuropsychiatry and Behavioral Neurosciences, Fifth Edition (American Psychiatric Press Textbook of Neuropsychiatry). US: American Psychiatric Publishing, Inc.. 1 December 2007. pp. 583–584. ISBN 978-1-58562-239-9. https://books.google.com/books?id=f5BEk-6yO_4C&pg=PA583.
- ↑ "Mortality Risk of Hypnotics: Strengths and Limits of Evidence". Drug Safety 39 (2): 93–107. February 2016. doi:10.1007/s40264-015-0362-0. PMID 26563222. https://escholarship.org/content/qt08d9f3d5/qt08d9f3d5.pdf?t=nz1gjv.
- ↑ "FDA expands Boxed Warning to improve safe use of benzodiazepine drug". 23 September 2020. https://www.fda.gov/drugs/drug-safety-and-availability/fda-requiring-boxed-warning-updated-improve-safe-use-benzodiazepine-drug-class.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ↑ 61.0 61.1 "Diazepam (Valium and Others)". Brooklyn College (Eilat.sci.Brooklyn.CUNY.edu). http://eilat.sci.brooklyn.cuny.edu/newnyc/DRUGS/Diazepam.htm.
- ↑ "Paradoxical reactions elicited by diazepam in children with classic autism". Functional Neurology 2 (3): 355–61. 1987. PMID 2826308.
- ↑ "Diazepam: Side Effects". RxList.com. http://www.rxlist.com/cgi/generic/diazepam_ad.htm.
- ↑ "[Benzodiazepines and forensic aspects"] (in fr). L'Encéphale 29 (6): 479–85. 2003. PMID 15029082. http://www.masson.fr/masson/portal/bookmark?Global=1&Page=18&MenuIdSelected=106&MenuItemSelected=0&MenuSupportSelected=0&CodeProduct4=539&CodeRevue4=ENC&Path=REVUE/ENC/2003/29/6/ARTICLE11106200473.xml&Locations=.
- ↑ "The effects of diazepam on human self-aggressive behavior". Psychopharmacology 178 (1): 100–6. February 2005. doi:10.1007/s00213-004-1966-8. PMID 15316710.
- ↑ "Bromazepam-induced dystonia". Biomedicine & Pharmacotherapy 46 (8): 375–6. 1992. doi:10.1016/0753-3322(92)90306-R. PMID 1292648.
- ↑ "[An experimental study of tolerance to benzodiazepines]". Revista Medico-Chirurgicala a Societatii de Medici Si Naturalisti Din Iasi 106 (4): 806–11. October–December 2002. PMID 14974234.
- ↑ "Sleep in chronic respiratory disease: COPD and hypoventilation disorders". European Respiratory Review 28 (153): 190064. September 2019. doi:10.1183/16000617.0064-2019. PMID 31554703.
- ↑ "Vigilance impairment after a single dose of benzodiazepines". Psychopharmacology 119 (1): 39–45. May 1995. doi:10.1007/BF02246052. PMID 7675948.
- ↑ "Risk factors associated with iatrogenic opioid and benzodiazepine withdrawal in critically ill pediatric patients: a systematic review and conceptual model". Pediatric Critical Care Medicine 16 (2): 175–83. February 2015. doi:10.1097/PCC.0000000000000306. PMID 25560429.
- ↑ "Benzodiazepine withdrawal syndrome: a literature review and evaluation". The American Journal of Drug and Alcohol Abuse 9 (1): 19–33. 1982. doi:10.3109/00952998209002608. PMID 6133446.
- ↑ "The benzodiazepine withdrawal syndrome and its management". The Journal of the Royal College of General Practitioners 39 (321): 160–3. April 1989. PMID 2576073.
- ↑ "New concepts in benzodiazepine therapy: rebound anxiety and new indications for the more potent benzodiazepines". Progress in Neuro-Psychopharmacology & Biological Psychiatry 7 (4–6): 669–73. 1983. doi:10.1016/0278-5846(83)90043-X. PMID 6141609.
- ↑ "Long-term anxiolytic therapy: the issue of drug withdrawal". The Journal of Clinical Psychiatry 48 (Suppl): 12–6. December 1987. PMID 2891684.
- ↑ "Comparative assessment of efficacy and withdrawal symptoms after 6 and 12 weeks' treatment with diazepam or buspirone". The British Journal of Psychiatry 154 (4): 529–34. April 1989. doi:10.1192/bjp.154.4.529. PMID 2686797.
- ↑ "[Benzodiazepines in the treatment of epilepsy]". L'Encéphale 9 (4 Suppl 2): 287B–292B. 1983. PMID 6373234.
- ↑ 77.0 77.1 "Diazepam: abuse and dependence". Rxlist.com. RxList Inc.. 24 January 2005. http://www.rxlist.com/cgi/generic/diazepam_ad.htm#DA.
- ↑ "Low-dose diazepam primes motivation for alcohol and alcohol-related semantic networks in problem drinkers". Behavioural Pharmacology 15 (7): 503–12. November 2004. doi:10.1097/00008877-200411000-00006. PMID 15472572.
- ↑ "Predictors of benzodiazepine discontinuation in subjects manifesting complicated dependence". Substance Use & Misuse 40 (4): 499–510. 2005. doi:10.1081/JA-200052433. PMID 15830732.
- ↑ "Effectiveness of current treatment approaches for benzodiazepine discontinuation: a meta-analysis". Addiction 104 (1): 13–24. January 2009. doi:10.1111/j.1360-0443.2008.02364.x. PMID 18983627.
- ↑ "Consider this slow-taper program for benzodiazepines". Current Psychiatry. July 2013. https://www.mdedge.com/psychiatry/article/77248/addiction-medicine/consider-slow-taper-program-benzodiazepines.
- ↑ "Tapering Benzodiazepines". https://www.smchealth.org/sites/main/files/file-attachments/benzodiazepinetaper08-12-13.pdf.
- ↑ 83.0 83.1 "Diazepam: overdose". Rxlist.com. RxList Inc.. 24 January 2005. http://www.rxlist.com/cgi/generic/diazepam_od.htm.
- ↑ 84.0 84.1 84.2 Better Than Prozac. New York: Oxford University Press. 2003. pp. 47–59. ISBN 978-0-19-515130-5. https://archive.org/details/betterthanprozac00baro/page/47.
- ↑ "Rapid recovery from massive diazepam overdose". JAMA 240 (17): 1872–4. October 1978. doi:10.1001/jama.1978.03290170054026. PMID 357765.
- ↑ "A survey of buprenorphine related deaths in Singapore". Forensic Science International 162 (1–3): 80–6. October 2006. doi:10.1016/j.forsciint.2006.03.037. PMID 16879940.
- ↑ 87.0 87.1 87.2 87.3 Food and Drug Interactions: A Guide for Consumers. Chicago: Precept Press. 1998. pp. 90–91. ISBN 978-0-944496-59-6.
- ↑ "Cardiovascular effects of diazepam and nitrazepam in combination with ethanol". Die Pharmazie 37 (12): 853–6. December 1982. PMID 7163374.
- ↑ "Pharmacokinetic drug interactions with oral contraceptives". Clinical Pharmacokinetics 18 (6): 472–84. June 1990. doi:10.2165/00003088-199018060-00004. PMID 2191822.
- ↑ "[Relationship between blood serum luminal and diphenylhydantoin level and the results of treatment and other clinical data in drug-resistant epilepsy]". Neurologia I Neurochirurgia Polska 14 (1): 39–45. January–February 1980. PMID 7374896.
- ↑ "The action of cisapride on gastric emptying and the pharmacodynamics and pharmacokinetics of oral diazepam". European Journal of Clinical Pharmacology 30 (2): 205–8. 1986. doi:10.1007/BF00614304. PMID 3709647.
- ↑ "Caffeine and theophylline counteract diazepam effects in man". Medical Biology 61 (6): 337–43. 1983. PMID 6374311.
- ↑ "Possible Interactions with: Valerian". University of Maryland Medical Center. 13 May 2013. http://www.umm.edu/altmed/articles/valerian-000934.htm.
- ↑ "Pharmacological characterization of benzodiazepine receptors in the brain". European Journal of Pharmacology 48 (3): 263–70. April 1978. doi:10.1016/0014-2999(78)90085-7. PMID 639854.
- ↑ "Effect of GABA agonists on the neurotoxicity and anticonvulsant activity of benzodiazepines". Life Sciences 36 (8): 737–44. February 1985. doi:10.1016/0024-3205(85)90193-6. PMID 2983169.
- ↑ "Micromolar-affinity benzodiazepine receptors regulate voltage-sensitive calcium channels in nerve terminal preparations". Proceedings of the National Academy of Sciences of the United States of America 81 (10): 3118–22. May 1984. doi:10.1073/pnas.81.10.3118. PMID 6328498. PMC 345232. Bibcode: 1984PNAS...81.3118T. http://www.pnas.org/cgi/reprint/81/10/3118.pdf.
- ↑ "Effects of anticonvulsants in vivo on high affinity choline uptake in vitro in mouse hippocampal synaptosomes". British Journal of Pharmacology 84 (1): 19–25. January 1985. doi:10.1111/j.1476-5381.1985.tb17368.x. PMID 3978310.
- ↑ "[3HDiazepam binding in mammalian central nervous system: a pharmacological characterization"]. The Journal of Neuroscience 1 (2): 218–25. February 1981. doi:10.1523/JNEUROSCI.01-02-00218.1981. PMID 6267221.
- ↑ "Diazepam-induced decrease in histamine turnover in mouse brain". European Journal of Pharmacology 124 (3): 337–42. May 1986. doi:10.1016/0014-2999(86)90236-0. PMID 3089825.
- ↑ "Suppression of prolactin secretion by benzodiazepines in vivo". Neuroendocrinology 34 (5): 369–73. 1982. doi:10.1159/000123330. PMID 6979001.
- ↑ "Hooked on benzodiazepines: GABAA receptor subtypes and addiction". University of Geneva. 2011. http://www.addictionscience.unige.ch/LabPublications-1/TrendsNeurosciKT2011.pdf.
- ↑ "Pharmacogenomics knowledge for personalized medicine". Clinical Pharmacology and Therapeutics 92 (4): 414–7. October 2012. doi:10.1038/clpt.2012.96. PMID 22992668.
- ↑ "Further evidence for GABA-ergic mechanisms in the action of benzodiazepines". Archives Internationales de Pharmacodynamie et de Thérapie 229 (2): 313–26. October 1977. PMID 23084.
- ↑ "Benzodiazepines, but not beta-carbolines, limit high-frequency repetitive firing of action potentials of spinal cord neurons in cell culture". The Journal of Pharmacology and Experimental Therapeutics 244 (2): 789–95. February 1988. PMID 2450203.
- ↑ "Investigation of the muscle relaxant activity of nitrazepam". Archives Internationales de Pharmacodynamie et de Thérapie 272 (1): 129–39. November 1984. PMID 6517646.
- ↑ "[Pharmacologic bases of use of benzodiazepines in peréinatal medicine]". Archives Françaises de Pédiatrie 34 (1): 74–89. January 1977. PMID 851373.
- ↑ "[Pharmacokinetic of benzodiazepines in old age]". Schweizerische Medizinische Wochenschrift 111 (47): 1789–93. November 1981. PMID 6118950.
- ↑ Plumb's, 6th edition page 372
- ↑ "New and mild method for the synthesis of alprazolam and diazepam and computational study of their binding mode to GABAA receptor". Medicinal Chemistry Research 25 (8): 1538–1550. August 2016. doi:10.1007/s00044-016-1585-z. http://link.springer.com/10.1007/s00044-016-1585-z.
- ↑ 110.0 110.1 "Concentrations of scheduled prescription drugs in blood of impaired drivers: considerations for interpreting the results". Therapeutic Drug Monitoring 29 (2): 248–60. April 2007. doi:10.1097/FTD.0b013e31803d3c04. PMID 17417081.
- ↑ "Evaluation of the Abbott ADx and TDx serum benzodiazepine immunoassays for analysis of alprazolam". Journal of Analytical Toxicology 15 (2): 63–5. 1991. doi:10.1093/jat/15.2.63. PMID 1675703.
- ↑ Disposition of Toxic Drugs and Chemicals in Man (9th ed.). Seal Beach, CA: Biomedical Publications. 2011. pp. 471–473. ISBN 978-0-9626523-8-7.
- ↑ "Susceptibility of phytoplankton to the increasing presence of active pharmaceutical ingredients (APIs) in the aquatic environment: A review". Aquatic Toxicology 234: 105809. May 2021. doi:10.1016/j.aquatox.2021.105809. PMID 33780670. Bibcode: 2021AqTox.23405809C.
- ↑ "Roche to Shut Former U.S. Headquarters". New York Times. 26 June 2012. https://www.nytimes.com/2012/06/27/business/roche-to-shut-down-former-us-headquarters-after-83-years.html.
- ↑ 115.0 115.1 "Leo Sternbach's Obituary". The Guardian (Guardian Unlimited). 3 October 2005. https://www.theguardian.com/medicine/story/0,,1583671,00.html.
- ↑ "Social reactions to Valium and Prozac: a cultural lag perspective of drug diffusion and adoption". Research in Social & Administrative Pharmacy 5 (2): 94–107. June 2009. doi:10.1016/j.sapharm.2008.06.005. PMID 19524858.
- ↑ "How the American opiate epidemic was started by one pharmaceutical company". 4 March 2015. https://theweek.com/articles/541564/how-american-opiate-epidemic-started-by-pharmaceutical-company.
- ↑ "Benzodiazepine utilisation in Australia: report from a new pharmacoepidemiological database". Australian Journal of Public Health 17 (4): 345–9. December 1993. doi:10.1111/j.1753-6405.1993.tb00167.x. PMID 7911332.
- ↑ "The benzodiazepine binding site of GABA(A) receptors as a target for the development of novel anxiolytics". Expert Opinion on Investigational Drugs 14 (5): 601–18. May 2005. doi:10.1517/13543784.14.5.601. PMID 15926867.
- ↑ "Benzodiazepines prescription in Dakar: a study about prescribing habits and knowledge in general practitioners, neurologists and psychiatrists". Fundamental & Clinical Pharmacology 20 (3): 235–8. June 2006. doi:10.1111/j.1472-8206.2006.00400.x. PMID 16671957.
- ↑ "New Evidence on Addiction To Medicines Diazepam Has Effect on Nerve Cells in the Brain Reward System". Medical News Today. August 2008. http://www.medicalnewstoday.com/articles/119284.php.
- ↑ "[Pharmacological studies on drug dependence. (III): Intravenous self-administration of some CNS-affecting drugs and a new sleep-inducer, 1H-1, 2, 4-triazolyl benzophenone derivative (450191-S), in rats]". Nihon Yakurigaku Zasshi. Folia Pharmacologica Japonica 83 (1): 39–67. January 1984. doi:10.1254/fpj.83.39. PMID 6538866.
- ↑ "Effects of several benzodiazepines, alone and in combination with flumazenil, in rhesus monkeys trained to discriminate pentobarbital from saline". Psychopharmacology 122 (3): 230–6. December 1995. doi:10.1007/BF02246544. PMID 8748392.
- ↑ "Report of the International Narcotics Control Board for 1996" (PDF). United Nations. International Narcotics Control Board. 1996. p. 27. http://www.incb.org/documents/Publications/AnnualReports/AR1996/AR_1996_E.pdf#37. "Phenobarbital was identified as the psychotropic substance most frequently used as an adulterant in seized heroin; it was followed by diazepam and flunitrazepam."
- ↑ "Relative abuse liability of hypnotic drugs: a conceptual framework and algorithm for differentiating among compounds". The Journal of Clinical Psychiatry 66 (Suppl 9): 31–41. 2005. PMID 16336040.
- ↑ "Methamphetamine and Benzodiazepines: Methamphetamine & Benzodiazepines". Erowid Experience Vaults. http://de1.erowid.org/experiences/exp.phpquery=ID=9402.html.
- ↑ "Drug Abuse Warning Network, 2011: National Estimates of Drug-Related Emergency Department Visits". U.S. Department of Health and Human Services, Substance Abuse and Mental Health Services Administration. 2011. http://www.samhsa.gov/data/2k13/DAWN2k11ED/DAWN2k11ED.htm#6.1.
- ↑ "Drugs and the impaired driver in Northern Ireland: an analytical survey". Forensic Science International 32 (4): 245–58. December 1986. doi:10.1016/0379-0738(86)90201-X. PMID 3804143.
- ↑ "List of psychotropic substances under international control". Green list. International Narcotics Control Board. 2003. http://infoespai.org/wp-content/uploads/2014/12/green.pdf.
- ↑ "List of controlled Drugs". http://www.homeoffice.gov.uk/publications/alcohol-drugs/drugs/drug-licences/controlled-drugs-list?view=Binary.
- ↑ "Anlage III (zu § 1 Abs. 1) verkehrsfähige und verschreibungsfähige Betäubungsmittel". Betäubungsmittelgesetz. 2001. http://bundesrecht.juris.de/btmg_1981/anlage_iii_61.html.
- ↑ 132.0 132.1 Poisons Standard June 2018 "Poisons Standard June 2018". https://www.legislation.gov.au/Details/F2018L00625.
- ↑ San Quentin State Prison Operational Procedure 0–770, Execution By Lethal Injection (pp. 44 & 92) Accessed 10 January 2014.
- ↑ "Execution by lethal injection procedures". Florida Department of Corrections. 9 September 2013. http://www.dc.state.fl.us/oth/deathrow/lethal-injection-procedures-as-of_9-9-2013.pdf.
- ↑ "Nebraska executes Carey Dean Moore for murders of Omaha cab drivers Maynard Helgeland, Reuel Van Ness Jr.". Lincoln Journal Star. 14 August 2018. https://journalstar.com/news/local/911/nebraska-executes-carey-dean-moore-for-murders-of-omaha-cab/article_c2332b24-145e-51b8-9bd3-d4b13e64b70c.html#1.
- ↑ 136.0 136.1 "Drugs Affecting Appetite (Monogastric)". The Merck Veterinary Manual. http://www.merckvetmanual.com/mvm/index.jsp?cfile=htm/bc/190302.htm.
- ↑ "Effects of delta 9-tetrahydrocannabinol and diazepam on feeding behavior in mice". The Journal of Veterinary Medical Science 61 (4): 351–5. April 1999. doi:10.1292/jvms.61.351. PMID 10342284.
- ↑ "Anticonvulsants Used to Stop Ongoing Seizure Activity". March 2012. http://www.merckmanuals.com/vet/pharmacology/systemic_pharmacotherapeutics_of_the_nervous_system/anticonvulsants_used_to_stop_ongoing_seizure_activity.html#v3331734. "Dogs and Cats:
A variety of drugs can be used to stop seizures in dogs and cats.
Benzodiazepines:
Diazepam is the most common benzodiazepine used in dogs and cats to reduce motor activity and permit placement of an IV catheter."
Further reading
- "Diazepam Therapy and CYP2C19 Genotype". Medical Genetics Summaries. National Center for Biotechnology Information (NCBI). 2016. Bookshelf ID: NBK379740. https://www.ncbi.nlm.nih.gov/books/NBK379740/.
External links
- "Diazepam Nasal Spray". https://medlineplus.gov/druginfo/meds/a620026.html.
- "Diazepam". Resource Center. Chemwatch. https://www.chemwatch.net/resource-center/diazepam/.
 |