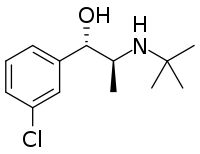
Chemistry:Threohydrobupropion
 | |
| Clinical data | |
|---|---|
| Other names | threo-Hydrobupropion; Threohydroxybupropion; BW 494; BW A494U; threo-3-Chloro-N-tert-butyl-β-hydroxy-α-methylphenethylamine; threo-3-Chloro-N-tert-butyl-β-hydroxyamphetamine |
| Pharmacokinetic data | |
| Protein binding | 42%[1] |
| Metabolism | Hydroxylation (CYP2B6, CYP2C19), glucuronidation (UGTs)[1] |
| Elimination half-life | 37 hours[1][2] |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| ChEMBL | |
| Chemical and physical data | |
| Formula | C13H20ClNO |
| Molar mass | 241.76 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Threohydrobupropion (developmental code names BW 494, BW A494U) is a substituted amphetamine derivative—specifically a β-hydroxyamphetamine—and a major active metabolite of the antidepressant drug bupropion (Wellbutrin).[1][2] Bupropion is a norepinephrine–dopamine reuptake inhibitor and nicotinic acetylcholine receptor negative allosteric modulator, with its metabolites contributing substantially to its activities.[1] Threohydrobupropion exists as two isomers, (1R,2R)-threohydrobupropion and (1S,2S)-threohydrobupropion.[3][1] Other metabolites of bupropion include hydroxybupropion and erythrohydrobupropion.[1][2]
Information on the pharmacological actions of threohydrobupropion is scarce.[1] In any case, it is about 20% as pharmacologically potent as bupropion and in the range of 20 to 50% as potent as bupropion in mouse models of depression.[1][2] Moreover, threohydrobupropion has been reported to weakly inhibit the reuptake of norepinephrine, dopamine, and serotonin with rat IC50 or Ki values of 16 μM, 47 μM, and 67 μM, respectively.[4] These values can be compared to rat values with bupropion of 1,400 nM, 570 nM, and 19,000 nM, respectively.[4] Besides monoamine reuptake inhibition, threohydrobupropion has also been reported to inhibit α3β4 nicotinic acetylcholine receptors, with an IC50 value of 14 μM.[5] Threohydrobupropion circulates at higher concentrations than bupropion during bupropion therapy, similarly to hydroxybupropion but in contrast to erythrohydrobupropion—which circulates at similar concentrations as bupropion.[1][2]
The plasma protein binding of threohydrobupropion is 42%.[1] Threohydrobupropion is formed from bupropion via reduction of the ketone group by 11β-hydroxysteroid dehydrogenase-1 and aldo-keto reductases.[1] It can also be formed from bupropion by carbonyl reductases.[1][2] The compound is metabolized by the cytochrome P450 enzymes CYP2B6 and CYP2C19 into threo-4'-hydroxy-hydrobupropion and by various glucuronosyltransferase enzymes into glucuronide conjugates.[1] Its elimination half-life is approximately 37 hours.[1][2]
Dry mouth during bupropion therapy has been associated with threohydrobupropion concentrations.[1] Administration of threohydrobupropion in mice produces seizures at sufficiently high doses similarly to bupropion and other metabolites.[1] Threohydrobupropion is a CYP2D6 inhibitor and accounts for about 21% of CYP2D6 inhibition during bupropion therapy, with hydroxybupropion accounting for 65% and erythrohydrobupropion accounting for 9%.[1]
References
- ↑ 1.00 1.01 1.02 1.03 1.04 1.05 1.06 1.07 1.08 1.09 1.10 1.11 1.12 1.13 1.14 1.15 1.16 1.17 "Pharmacokinetic and pharmacodynamic of bupropion: integrative overview of relevant clinical and forensic aspects". Drug Metab Rev 51 (3): 293–313. August 2019. doi:10.1080/03602532.2019.1620763. PMID 31124380.
- ↑ 2.0 2.1 2.2 2.3 2.4 2.5 2.6 "Bupropion for major depressive disorder: Pharmacokinetic and formulation considerations". Clin Ther 27 (11): 1685–95. November 2005. doi:10.1016/j.clinthera.2005.11.011. PMID 16368442.
- ↑ "Chiral Plasma Pharmacokinetics and Urinary Excretion of Bupropion and Metabolites in Healthy Volunteers". J Pharmacol Exp Ther 358 (2): 230–8. August 2016. doi:10.1124/jpet.116.232876. PMID 27255113.
- ↑ 4.0 4.1 "Comparison of the effects of antidepressants and their metabolites on reuptake of biogenic amines and on receptor binding". Cell Mol Neurobiol 19 (4): 467–89. August 1999. doi:10.1023/a:1006986824213. PMID 10379421.
- ↑ "Behavioral and biochemical investigations of bupropion metabolites". Eur J Pharmacol 474 (1): 85–93. August 2003. doi:10.1016/s0014-2999(03)02010-7. PMID 12909199.
 |

