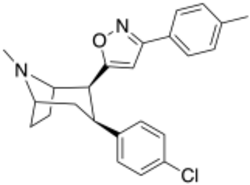
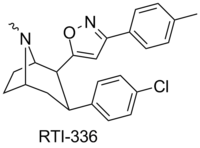
Chemistry:RTI-336
 | |
| Identifiers | |
|---|---|
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| Chemical and physical data | |
| Formula | C24H25ClN2O |
| Molar mass | 392.93 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
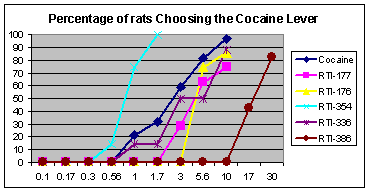
RTI(-4229)-336, (LS-193,309, (−)-2β-(3-(4-methylphenyl)isoxazol-5-yl)-3β-(4-chlorophenyl)tropane) is a phenyltropane derivative which acts as a potent and selective dopamine reuptake inhibitor and stimulant drug. It binds to the dopamine transporter with around 20x the affinity of cocaine,[1] however it produces relatively mild stimulant effects, with a slow onset and long duration of action.[2] (however other sources class it as having among the faster onsets of action from among phenyltropanes[3]) These characteristics make it a potential candidate for treatment of cocaine addiction, as a possible substitute drug analogous to how methadone is used for treating heroin abuse.[4][5] RTI-336 fully substitutes for cocaine in addicted monkeys and supports self-administration,[6][7] and significantly reduces rates of cocaine use, especially when combined with SSRIs,[8] and research is ongoing to determine whether it could be a viable substitute drug in human cocaine addicts.
Update
Pharmacotherapy for Cocaine Abuse
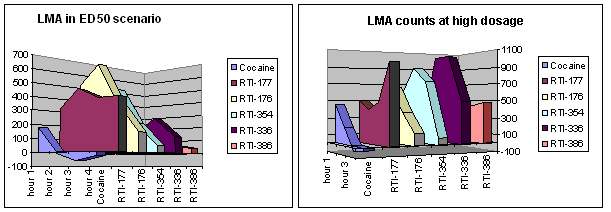
| RTI | X | R | [3H]CFT | [3H]Nisoxetine | [3H]Paroxetine | N ÷ D | S ÷ D |
|---|---|---|---|---|---|---|---|
| Coc | — | — | 89.1 | 3298 (1986) | 1045 (45) | 37.01 | 11.79 |
| 177 | Cl | phenyl | 1.28 | 504 (304) | 2420 (220) | 393.8 | 1891 |
| 176 | Me | phenyl | 1.58 | 398 (239) | 5110 (465) | 251.9 | 3234 |
| 354 | Me | ethyl | 1.62 | 299 (180) | 6400 (582) | 184.6 | 3951 |
| 336 | Cl | p-tolyl | 4.09 | 1714 (1033) | 5741 (522) | 419.1 | 1404 |
| 386 | Me | p-anisyl | 3.93 | 756 (450) | 4027 (380) | 192.4 | 1025 |
See also
References
- ↑ "Synthesis, monoamine transporter binding properties, and behavioral pharmacology of a series of 3beta-(substituted phenyl)-2beta-(3'-substituted isoxazol-5-yl)tropanes". Journal of Medicinal Chemistry 47 (2): 296–302. January 2004. doi:10.1021/jm030453p. PMID 14711303.
- ↑ "Effects of dopamine transporter selective 3-phenyltropane analogs on locomotor activity, drug discrimination, and cocaine self-administration after oral administration". European Journal of Pharmacology 553 (1–3): 149–56. December 2006. doi:10.1016/j.ejphar.2006.09.024. PMID 17067572.
- ↑ "Faster onset and dopamine transporter selectivity predict stimulant and reinforcing effects of cocaine analogs in squirrel monkeys". Pharmacology, Biochemistry, and Behavior 86 (1): 45–54. January 2007. doi:10.1016/j.pbb.2006.12.006. PMID 17258302.
- ↑ "Development of the dopamine transporter selective RTI-336 as a pharmacotherapy for cocaine abuse". The AAPS Journal 8 (1): E196-203. March 2006. doi:10.1208/aapsj080124. PMID 16584128.
- ↑ "Emerging pharmacological strategies in the fight against cocaine addiction". Expert Opinion on Emerging Drugs 11 (1): 91–8. March 2006. doi:10.1517/14728214.11.1.91. PMID 16503828.
- ↑ "Faster onset and dopamine transporter selectivity predict stimulant and reinforcing effects of cocaine analogs in squirrel monkeys". Pharmacology, Biochemistry, and Behavior 86 (1): 45–54. January 2007. doi:10.1016/j.pbb.2006.12.006. PMID 17258302.
- ↑ "Relationship between rate of drug uptake in brain and behavioral pharmacology of monoamine transporter inhibitors in rhesus monkeys". Pharmacology, Biochemistry, and Behavior 90 (3): 453–62. September 2008. doi:10.1016/j.pbb.2008.03.032. PMID 18468667.
- ↑ "Effects of combined dopamine and serotonin transporter inhibitors on cocaine self-administration in rhesus monkeys". The Journal of Pharmacology and Experimental Therapeutics 320 (2): 757–65. February 2007. doi:10.1124/jpet.106.108324. PMID 17105829.
- ↑ "Positive allosteric modulation of the human cannabinoid (CB) receptor by RTI-371, a selective inhibitor of the dopamine transporter". British Journal of Pharmacology 156 (7): 1178–84. April 2009. doi:10.1111/j.1476-5381.2009.00124.x. PMID 19226282.
 |