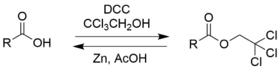(diff) ← Older revision | Latest revision (diff) | Newer revision → (diff)
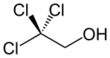
2,2,2-Trichloroethanol
|
|
| Names
|
Preferred IUPAC name
2,2,2-Trichloroethan-1-ol |
| Identifiers
|
|
|
|
|
|
|
|
|
1697495
|
| ChEBI
|
|
| ChEMBL
|
|
| ChemSpider
|
|
| EC Number
|
|
|
|
2407
|
|
|
|
| KEGG
|
|
|
|
|
| UNII
|
|
InChI=1S/C2H3Cl3O/c3-2(4,5)1-6/h6H,1H2  Y YKey: KPWDGTGXUYRARH-UHFFFAOYSA-N  Y YInChI=1/C2H3Cl3O/c3-2(4,5)1-6/h6H,1H2 Key: KPWDGTGXUYRARH-UHFFFAOYAW
|
|
|
| Properties
|
|
|
C2H3Cl3O
|
| Molar mass
|
149.40 g/mol
|
| Density
|
1.55 g/cm3
|
| Melting point
|
17.8[1] °C (64.0 °F; 290.9 K)
|
| Boiling point
|
151[1] °C (304 °F; 424 K)
|
| Hazards
|
| Safety data sheet
|
[1]
|
| GHS pictograms
|
 
|
| GHS Signal word
|
Danger
|
|
|
H302, H315, H318, H336
|
|
|
P261, P264, P270, P271, P280, P301+312, P302+352, P304+340, P305+351+338, P310, P312, P321, P330, P332+313, P362, P403+233, P405, P501
|
| Flash point
|
88 °C (190 °F; 361 K)[1]
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). |
 Y verify (what is Y verify (what is  Y Y N ?) N ?)
|
| Infobox references
|
|
|
|
2,2,2-Trichloroethanol is the chemical compound with formula Cl
3C–CH
2OH. Its molecule can be described as that of ethanol, with the three hydrogen atoms at position 2 (the methyl group) replaced by chlorine atoms. It is a clear flammable liquid at room temperature, colorless when pure but often with a light yellow color.[1][2]
The pharmacological effects of this compound in humans are similar to those of its prodrug chloral hydrate, and of chlorobutanol. Historically, it has been used as a sedative hypnotic.[3] The hypnotic drug triclofos (2,2,2-trichloroethyl phosphate) is metabolized in vivo to 2,2,2-trichloroethanol. Chronic exposure may result in kidney and liver damage.[4]
2,2,2-Trichloroethanol can be added to SDS-PAGE gels in order to enable fluorescent detection of proteins without a staining step, for immunoblotting or other analysis methods.[5]
Use in organic synthesis
2,2,2-Trichloroethanol is an effective protecting group for carboxylic acids due to its ease in addition and removal.[6]
See also
References
- ↑ 1.0 1.1 1.2 1.3 1.4 "Material Safety Data Sheet- 2,2,2-Trichloroethanol, 98%" Online document at the Cole-Parmer website. Accessed on 2020-07-11.
- ↑ "2,2,2-Trichloroethanol ≥99%". Online product catalog page at Merck website. Accessed on 2020-07-11.
- ↑ The Merck Index, 13th Edition.
- ↑ S. Budavari; M. O'Neil; Ann Smith; P. Heckelman; J. Obenchain (15 March 1996). The Merck Index (12th print ed.). Taylor & Francis. ISBN 978-0-911910-12-4.
- ↑ Ladner, Carol (March 2004). "Visible fluorescent detection of proteins in polyacrylamide gels without staining". Analytical Biochemistry 326 (1): 13–20. doi:10.1016/j.ab.2003.10.047. PMID 14769330.
- ↑ Lowder, Patrick D. (2001-04-15), "2,2,2-Trichloroethanol" (in en), Encyclopedia of Reagents for Organic Synthesis, John Wiley & Sons, Ltd, doi:10.1002/047084289x.rt203, ISBN 978-0471936237
|
|---|
|
|---|
| Alcohols | |
|---|
| Barbiturates | |
|---|
| Benzodiazepines | |
|---|
| Carbamates | |
|---|
| Flavonoids | |
|---|
| Imidazoles | |
|---|
| Kava constituents | |
|---|
| Monoureides | |
|---|
| Neuroactive steroids | |
|---|
| Nonbenzodiazepines | |
|---|
| Phenols | |
|---|
| Piperidinediones | |
|---|
| Pyrazolopyridines | |
|---|
| Quinazolinones | |
|---|
| Volatiles/gases | |
|---|
| Others/unsorted |
- 3-Hydroxybutanal
- α-EMTBL
- AA-29504
- Avermectins (e.g., ivermectin)
- Bromide compounds (e.g., lithium bromide, potassium bromide, sodium bromide)
- Carbamazepine
- Chloralose
- Chlormezanone
- Clomethiazole
- DEABL
- Dihydroergolines (e.g., dihydroergocryptine, dihydroergosine, dihydroergotamine, ergoloid (dihydroergotoxine))
- DS2
- Efavirenz
- Etazepine
- Etifoxine
- Fenamates (e.g., flufenamic acid, mefenamic acid, niflumic acid, tolfenamic acid)
- Fluoxetine
- Flupirtine
- Hopantenic acid
- Lanthanum
- Lavender oil
- Lignans (e.g., 4-O-methylhonokiol, honokiol, magnolol, obovatol)
- Loreclezole
- Menthyl isovalerate (validolum)
- Monastrol
- Niacin
- Nicotinamide (niacinamide)
- Org 25,435
- Phenytoin
- Propanidid
- Retigabine (ezogabine)
- Safranal
- Seproxetine
- Stiripentol
- Sulfonylalkanes (e.g., sulfonmethane (sulfonal), tetronal, trional)
- Terpenoids (e.g., borneol)
- Topiramate
- Valerian constituents (e.g., isovaleric acid, isovaleramide, valerenic acid, valerenol)
|
|---|
|
|
|---|
Receptor
(ligands) | | GlyR |
- Positive modulators: Alcohols (e.g., brometone, chlorobutanol (chloretone), ethanol (alcohol), tert-butanol (2M2P), tribromoethanol, trichloroethanol, trifluoroethanol)
- Alkylbenzene sulfonate
- Anandamide
- Barbiturates (e.g., pentobarbital, sodium thiopental)
- Chlormethiazole
- D12-116
- Dihydropyridines (e.g., nicardipine)
- Etomidate
- Ginseng constituents (e.g., ginsenosides (e.g., ginsenoside-Rf))
- Glutamic acid (glutamate)
- Ivermectin
- Ketamine
- Neuroactive steroids (e.g., alfaxolone, pregnenolone (eltanolone), pregnenolone acetate, minaxolone, ORG-20599)
- Nitrous oxide
- Penicillin G
- Propofol
- Tamoxifen
- Tetrahydrocannabinol
- Triclofos
- Tropeines (e.g., atropine, bemesetron, cocaine, LY-278584, tropisetron, zatosetron)
- Volatiles/gases (e.g., chloral hydrate, chloroform, desflurane, diethyl ether (ether), enflurane, halothane, isoflurane, methoxyflurane, sevoflurane, toluene, trichloroethane (methyl chloroform), trichloroethylene)
- Xenon
- Zinc
- Antagonists: 2-Aminostrychnine
- 2-Nitrostrychnine
- 4-Phenyl-4-formyl-N-methylpiperidine
- αEMBTL
- Bicuculline
- Brucine
- Cacotheline
- Caffeine
- Colchicine
- Colubrine
- Cyanotriphenylborate
- Dendrobine
- Diaboline
- Endocannabinoids (e.g., 2-AG, anandamide (AEA))
- Gaboxadol (THIP)
- Gelsemine
- iso-THAZ
- Isobutyric acid
- Isonipecotic acid
- Isostrychnine
- Laudanosine
- N-Methylbicuculline
- N-Methylstrychnine
- N,N-Dimethylmuscimol
- Nipecotic acid
- Pitrazepin
- Pseudostrychnine
- Quinolines (e.g., 4-hydroxyquinoline, 4-hydroxyquinoline-3-carboxylic acid, 5,7-CIQA, 7-CIQ, 7-TFQ, 7-TFQA)
- RU-5135
- Sinomenine
- Strychnine
- Thiocolchicoside
- Tutin
|
|---|
| NMDAR | |
|---|
|
|---|
Transporter
(blockers) | |
|---|
|
|
 | Original source: https://en.wikipedia.org/wiki/2,2,2-Trichloroethanol. Read more |