Biology:Eomesodermin
 Generic protein structure example |
Eomesodermin also known as T-box brain protein 2 (Tbr2) is a protein that in humans is encoded by the EOMES gene.[1]
The Eomesodermin/Tbr2 gene, EOMES, encodes a member of a conserved protein family that shares a common DNA-binding domain, the T-box.[2] T-box genes encode transcription factors, which control gene expression, involved in the regulation of developmental processes. Eomesodermin/Tbr2 itself controls regulation of radial glia, as well as other related cells.[2] Eomesodermin/Tbr2 has also been found to have a role in immune response, and there exists some loose evidence for its connections in other systems.[3]
Nervous system development
Neurogenesis
Eomesodermin/Tbr2 is expressed highly in the intermediate progenitor stage of the developing neuron.[4] Neurons, the primary functional cells of the brain, are developed from radial glia cells. This process of cells developing into other types of cells is called differentiation. Radial glia are present in the ventricular zone of the brain, which are on the lateral walls of the lateral ventricles.[5] Radial glia divide and migrate towards the surface of the brain, the cerebral cortex. During this migration, there are three stages of cellular development: radial glia, intermediate progenitors, and postmitotic projection neurons.[4] Radial glia express Pax6, while intermediate progenitor cells express Eomesodermin/Tbr2, and postmitotic projection neurons express Tbr1.[4] This process, known as neurogenesis, occurs mainly in the developing cortex before the organism has fully developed, and thus Eomesodermin/Tbr2 has been implicated in neurodevelopment.
Tbr2 has been observed in a transcription factor cascade to enable to development of glutamatergic neurons. Pax6, as expressed by radial glia cells, activates the transcription of Neurogenin-2 which then activates the generation of intermediate progenitor cells (IPC) expressing Tbr2. These cells are localized within the subventricular zone. The IPCs then undergo symmetric division to produce NeuroD expressing cells that can differentiate in TBR1 neurons. Similar mechanisms have been observed in both embryonic and adult neurogenesis. [6]
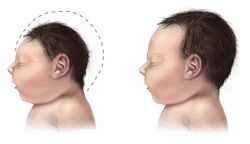
Tbr2 inactivation has also been tied to deficiencies in cortical neurogenesis further suggesting the importance of the cascade in activating and maintaining neuron production.[7] It has been found experimentally through knockout studies that mice lacking Eomesodermin/Tbr2 during early development have a reduced number of actively dividing cells, called proliferating cells, in the subventricular zone.[8] This, may lead to the microcephaly (small head size due to improper brain development) seen in Eomesodermin/Tbr2 deficient mice.[8] Eomesodermin/Tbr2 lacking mice have smaller upper cortical layers and a smaller sub ventricular zone in the brain, and have an absence of a mitral cell (neurons involved in the olfactory pathway) layer, with mitral cells instead being scattered about.[8] Phenotypically, Eomesodermin/Tbr2 lacking mice show high anger levels and perform infanticide.[2] Eomesodermin/Tbr2 lacking mice also seem to have problems with long axon connections.[8] Axons are projections from neurons that connect with other cells in what is called a synapse and send neurotransmitters. In this way, they can communicate with other cells, and form the processing that allows are brains to function. Eomesodermin/Tbr2 lacking mice seem to lack fully formed commissural fibers, which connect the two hemispheres of the brain, and lack the corpus callosum, another region of the brain involved in hemisphere connections.[8]
Role in adult development
There are locations within the brain that have been discovered to perform neurogenesis into adulthood,[3] [4] including the ventricular zone.[9] The hippocampus, which is involved in memory formation, shows decreased neurogenesis when Eomesodermin/Tbr2 is removed.[10] It was also found that Eomesodermin/Tbr2 functions by reducing amounts of Sox2, which is associated with radial glia.[10] Another study found that mice without Eomesodermin/Tbr2 lacked long term memory formation, which may relate to Eomesodermin/Tbr2's effects on the hippocampus.[11]
Cardiac development
Early in development, Eomesodermin/Tbr2 controls early differentiation of the cardiac mesoderm.[12] Lack of Eomesodermin/Tbr2 appears to be correlated with failure to differentiate into cardiomyocytes. Eomesodermin/Tbr2 controls the expression of cardiac specific genes Mesp1, Myl7, Myl2, Myocardin, Nkx2.5 and Mef2c.[12]
Immune response
Eomesodermin/Tbr2 is highly expressed in CD8+ T cells, but not CD4+ T cells.[3] CD4+ T cells are the helper T cells which detect foreign particles in the body, and call CD8+ T cells to facilitate death of the foreign particles. Eomesodermin/Tbr2 was found to play a role in the anti cancer properties of CD8+ T cells.[3] Lack of Eomesodermin/Tbr2, alongside T bet, another T box protein, caused CD8+ T cells to not penetrate tumors so they could perform their anti cancer duties.[3] Eomesodermin/Tbr2 prevents CD8+ cells from differentiating into other types of T cells, but does not play a role in the production of CD8+ T cells itself.[3]
See also
References
- ↑ "Entrez Gene: Eomesodermin". https://www.ncbi.nlm.nih.gov/sites/entrez?db=gene&cmd=retrieve&list_uids=8320.
- ↑ 2.0 2.1 2.2 "The T-box transcription factor Eomes/Tbr2 regulates neurogenesis in the cortical subventricular zone". Genes & Development 22 (18): 2479–84. 2008. doi:10.1101/gad.475408. PMID 18794345.
- ↑ 3.0 3.1 3.2 3.3 3.4 3.5 "T-bet and eomesodermin are required for T cell-mediated antitumor immune responses". Journal of Immunology 185 (6): 3174–83. Sep 2010. doi:10.4049/jimmunol.1000749. PMID 20713880.
- ↑ 4.0 4.1 4.2 4.3 "Pax6, Tbr2, and Tbr1 are expressed sequentially by radial glia, intermediate progenitor cells, and postmitotic neurons in developing neocortex". The Journal of Neuroscience 25 (1): 247–51. Jan 2005. doi:10.1523/JNEUROSCI.2899-04.2005. PMID 15634788.
- ↑ "Cellular composition and cytoarchitecture of the adult human subventricular zone: a niche of neural stem cells". The Journal of Comparative Neurology 494 (3): 415–34. Jan 2006. doi:10.1002/cne.20798. PMID 16320258.
- ↑ Hodge, Rebecca D.; Kowalczyk, Thomas D.; Wolf, Susanne A.; Encinas, Juan M.; Rippey, Caitlin; Enikolopov, Grigori; Kempermann, Gerd; Hevner, Robert F. (2008-04-02). "Intermediate Progenitors in Adult Hippocampal Neurogenesis: Tbr2 Expression and Coordinate Regulation of Neuronal Output". The Journal of Neuroscience 28 (14): 3707–3717. doi:10.1523/JNEUROSCI.4280-07.2008. PMID 18385329.
- ↑ Kowalczyk, Tom; Pontious, Adria; Englund, Chris; Daza, Ray A. M.; Bedogni, Francesco; Hodge, Rebecca; Attardo, Alessio; Bell, Chris et al. (2009-10-01). "Intermediate Neuronal Progenitors (Basal Progenitors) Produce Pyramidal–Projection Neurons for All Layers of Cerebral Cortex" (in en). Cerebral Cortex 19 (10): 2439–2450. doi:10.1093/cercor/bhn260. ISSN 1047-3211. PMID 19168665.
- ↑ 8.0 8.1 8.2 8.3 8.4 "Tbr2 directs conversion of radial glia into basal precursors and guides neuronal amplification by indirect neurogenesis in the developing neocortex glish". Neuron 60 (1): 56–69. Oct 2008. doi:10.1016/j.neuron.2008.09.028. PMID 18940588.
- ↑ "Adult neurogenesis in the mammalian brain: significant answers and significant questions". Neuron 70 (4): 687–702. May 2011. doi:10.1016/j.neuron.2011.05.001. PMID 21609825.
- ↑ 10.0 10.1 "Tbr2 is essential for hippocampal lineage progression from neural stem cells to intermediate progenitors and neurons". The Journal of Neuroscience 32 (18): 6275–87. May 2012. doi:10.1523/JNEUROSCI.0532-12.2012. PMID 22553033.
- ↑ "Characterization of T-bet and eomes in peripheral human immune cells". Frontiers in Immunology 5: 217. 2014-05-14. doi:10.3389/fimmu.2014.00217. PMID 24860576.
- ↑ 12.0 12.1 "The T-box transcription factor Eomesodermin acts upstream of Mesp1 to specify cardiac mesoderm during mouse gastrulation". Nature Cell Biology 13 (9): 1084–91. Sep 2011. doi:10.1038/ncb2304. PMID 21822279.
Further reading
- "A novel mammalian T-box-containing gene, Tbr2, expressed in mouse developing brain". Brain Research. Developmental Brain Research 115 (2): 183–93. Jun 1999. doi:10.1016/s0165-3806(99)00064-4. PMID 10407135.
- "Effector and memory CD8+ T cell fate coupled by T-bet and eomesodermin". Nature Immunology 6 (12): 1236–44. Dec 2005. doi:10.1038/ni1268. PMID 16273099.
- "Homozygous silencing of T-box transcription factor EOMES leads to microcephaly with polymicrogyria and corpus callosum agenesis". Nature Genetics 39 (4): 454–6. Apr 2007. doi:10.1038/ng1993. PMID 17353897.
- "The T-box transcription factor eomesodermin controls CD8 T cell activity and lymph node metastasis in human colorectal cancer". Gut 56 (11): 1572–8. Nov 2007. doi:10.1136/gut.2006.117812. PMID 17566017.
- "Histone acetylation facilitates rapid and robust memory CD8 T cell response through differential expression of effector molecules (eomesodermin and its targets: perforin and granzyme B)". Journal of Immunology 180 (12): 8102–8. Jun 2008. doi:10.4049/jimmunol.180.12.8102. PMID 18523274.
- "Tbr2 directs conversion of radial glia into basal precursors and guides neuronal amplification by indirect neurogenesis in the developing neocortex". Neuron 60 (1): 56–69. Oct 2008. doi:10.1016/j.neuron.2008.09.028. PMID 18940588.
- "Surviving the crash: transitioning from effector to memory CD8+ T cell". Seminars in Immunology 21 (2): 92–8. Apr 2009. doi:10.1016/j.smim.2009.02.002. PMID 19269192.
- "[Role of transcription factor T-bet and Eomes in IFN-gamma secretion of different human T cell subsets]". Xi Bao Yu Fen Zi Mian Yi Xue Za Zhi = Chinese Journal of Cellular and Molecular Immunology 26 (1): 31–4. Jan 2010. PMID 20056084.
- Unutmaz, Derya, ed (2010). "Human Th1 cells that express CD300a are polyfunctional and after stimulation up-regulate the T-box transcription factor eomesodermin". PLOS ONE 5 (5): e10636. doi:10.1371/journal.pone.0010636. PMID 20498708. Bibcode: 2010PLoSO...510636N.
- "Cutting edge: Lymphoproliferation caused by Fas deficiency is dependent on the transcription factor eomesodermin". Journal of Immunology 185 (12): 7151–5. Dec 2010. doi:10.4049/jimmunol.1003193. PMID 21076068.
This article incorporates text from the United States National Library of Medicine, which is in the public domain.
 |
