Biology:Protein c-Fos
 Generic protein structure example |
Protein c-Fos is a proto-oncogene and transcription factor that plays an important role in cellular function and human disease including cancer. It is encoded in humans by the FOS gene and frequently forms both homodimers and heterdimers.
When c-fos heterodimerizes with the transcription factor c-jun (from the Jun family), they form an AP-1 (Activator Protein-1) complex which binds DNA at specific sites to modulate gene expression.[1]
c-Fos is a part of a bigger Fos family of transcription factors which includes c-Fos, FosB, Fra-1 and Fra-2.[2] It plays an important role in many cellular functions and has been found to be overexpressed in a variety of cancers.
History
It was first discovered in rat fibroblasts as the transforming gene of the FBJ MSV (Finkel–Biskis–Jinkins murine osteogenic sarcoma virus).[3]
Structure and function
c-Fos is a 380 amino acid (62 kDa) protein with a basic leucine zipper region for dimerisation and DNA-binding and a transactivation domain at C-terminus, and, like Jun proteins, it can form homodimers.[4] In vitro studies have shown that Jun–Fos heterodimers are more stable and have stronger DNA-binding activity than Jun–Jun homodimers.[5]
C-Fos the human homolog of the retroviral oncogene v-fos.[6] In humans, it has been mapped to chromosome region 14q21→q31.
A variety of stimuli, including serum, growth factors, tumor promoters, cytokines, and UV radiation induce their expression. The c-fos mRNA and protein is generally among the first to be expressed and hence referred to as an immediate early gene. It is rapidly and transiently induced, within 15 minutes of stimulation.[7] Its activity is also regulated by posttranslational modification caused by phosphorylation by different kinases, like MAPK, CDC2, PKA or PKC which influence protein stability, DNA-binding activity and the trans-activating potential of the transcription factors.[8][9][10] It can cause gene repression as well as gene activation, although different domains are believed to be involved in both processes.
It is involved in important cellular events, including cell proliferation, differentiation and survival; genes associated with hypoxia; and angiogenesis;[11] which makes its dysregulation an important factor for cancer development. It can also induce a loss of cell polarity and epithelial-mesenchymal transition, leading to invasive and metastatic growth in mammary epithelial cells.[12]
The importance of c-fos in biological context has been determined by eliminating endogenous function by using anti-sense mRNA, anti-c-fos antibodies, a ribozyme that cleaves c-fos mRNA or a dominant negative mutant of c-fos. The transgenic mice thus generated are viable, demonstrating that there are c-fos dependent and independent pathways of cell proliferation, but display a range of tissue-specific developmental defects, including osteoporosis, delayed gametogenesis, lymphopenia and behavioral abnormalities.
Clinical significance
The AP-1 complex has been implicated in transformation and progression of cancer. In osteosarcoma and endometrial carcinoma, c-Fos overexpression was associated with high-grade lesions and poor prognosis. Also, in a comparison between precancerous lesion of the cervix uteri and invasive cervical cancer, c-Fos expression was significantly lower in precancerous lesions. c-Fos has also been identified as independent predictor of decreased survival in breast cancer.[20]
It was found that overexpression of c-fos from class I MHC promoter in transgenic mice leads to the formation of osteosarcomas due to increased proliferation of osteoblasts whereas ectopic expression of the other Jun and Fos proteins does not induce any malignant tumors. Activation of the c-Fos transgene in mice results in overexpression of cyclin D1, A and E in osteoblasts and chondrocytes, both in vitro and in vivo, which might contribute to the uncontrolled growth leading to tumor. Human osteosarcomas analyzed for c-fos expression have given positive results in more than half the cases and c-fos expression has been associated with higher frequency of relapse and poor response to chemotherapy.
Several studies have raised the idea that c-Fos may also have tumor-suppressor activity, that it might be able to promote as well as suppress tumorigenesis. Supporting this is the observation that in ovarian carcinomas, loss of c-Fos expression correlates with disease progression. This double action could be enabled by differential protein composition of tumour cells and their environment, for example, dimerisation partners, co-activators and promoter architecture. It is possible that the tumor suppressing activity is due to a proapoptotic function. The exact mechanism by which c-Fos contributes to apoptosis is not clearly understood, but observations in human hepatocellular carcinoma cells indicate that c-Fos is a mediator of c-myc-induced cell death and might induce apoptosis through the p38 MAP kinase pathway. Fas ligand (FASLG or FasL) and the tumour necrosis factor-related apoptosis-inducing ligand (TNFSF10 or TRAIL) might reflect an additional apoptotic mechanism induced by c-Fos, as observed in a human T-cell leukaemia cell line. Another possible mechanism of c-Fos involvement in tumour suppression could be the direct regulation of BRCA1, a well established factor in familial breast and ovarian cancer.
In addition, the role of c-fos and other Fos family proteins has also been studied in endometrial carcinoma, cervical cancer, mesotheliomas, colorectal cancer, lung cancer, melanomas, thyroid carcinomas, esophageal cancer, hepatocellular carcinomas, etc.
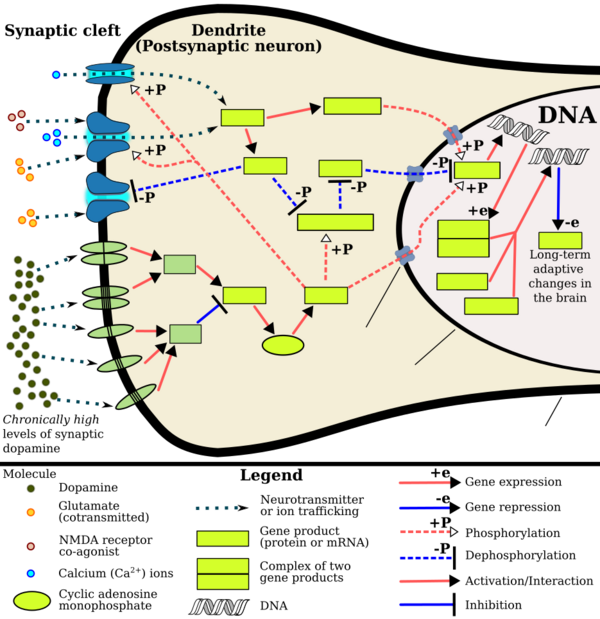
Cocaine, methamphetamine,[21] morphine,[22] and other psychoactive drugs[23][24] have been shown to increase c-Fos production in the mesocortical pathway (prefrontal cortex) as well as in the mesolimbic reward pathway (nucleus accumbens), as well as display variability depending on prior sensitization.[24] c-Fos repression by ΔFosB's AP-1 complex within the D1-type medium spiny neurons of the nucleus accumbens acts as a molecular switch that enables the chronic induction of ΔFosB, thus allowing it to accumulate more rapidly. As such, the c-Fos promoter finds utilization in drug addiction research in general, as well as with context-induced relapse to drug-seeking and other behavioral changes associated with chronic drug taking.
Applications
Expression of c-fos is an indirect marker of neuronal activity because c-fos is often expressed when neurons fire action potentials.[25][26][27] Up-regulation of c-fos mRNA in a neuron is considered a marker for activity.[28]
The c-fos promoter has also been utilized for drug abuse research. Scientists use this promoter to turn on transgenes in rats, allowing them to manipulate specific neuronal ensembles to assess their role in drug-related memories and behavior.[29] TetTag mice have been created to reactivate or silence cFos-expressing neurons with optogenetic tools or with DREADDs.[30]
Interactions
c-Fos has been shown to interact with:
- BCL3,[31]
- COBRA1,[32]
- CSNK2A1,[33]
- CSNK2A2,[33]
- DDIT3,[34]
- JUN[35][36][37][38][39][40][41]
- NCOA1,[42][43]
- NCOR2,[44]
- RELA,[35]
- RUNX1,[45][46]
- RUNX2,[45][46]
- SMAD3,[47] and
- TBP.[48]
See also
- Leptomycin
- c-Jun
- Egr-1
References
- ↑ (Text color) Transcription factors
- ↑ "The c-Fos protein interacts with c-Jun/AP-1 to stimulate transcription of AP-1 responsive genes". Cell 54 (4): 541–52. August 1988. doi:10.1016/0092-8674(88)90076-1. PMID 3135940.
- ↑ Milde-Langosch K (November 2005). "The Fos family of transcription factors and their role in tumourigenesis". Eur. J. Cancer 41 (16): 2449–61. doi:10.1016/j.ejca.2005.08.008. PMID 16199154.
- ↑ Curran, T; Teich, N M (1982-04-01). "Candidate product of the FBJ murine osteosarcoma virus oncogene: characterization of a 55,000-dalton phosphoprotein". Journal of Virology 42 (1): 114–122. doi:10.1128/jvi.42.1.114-122.1982. ISSN 0022-538X. https://doi.org/10.1128/jvi.42.1.114-122.1982.
- ↑ "Evidence for homodimerization of the c-Fos transcription factor in live cells revealed by fluorescence microscopy and computer modeling". Mol. Cell. Biol. 35 (21): 3785–98. November 2015. doi:10.1128/MCB.00346-15. PMID 26303532.
- ↑ "c-Jun dimerizes with itself and with c-Fos, forming complexes of different DNA binding affinities". Cell 55 (5): 917–24. December 1988. doi:10.1016/0092-8674(88)90147-X. PMID 3142692. http://genesdev.cshlp.org/content/2/12b/1687.full.pdf.
- ↑ Curran, T: The c-fos proto-oncogene. In: Reddy EP, Skalka AM, Curran T (eds.). The Oncogene Handbook 1988 Elsevier, New York, pp 307–327
- ↑ "Targeted disruption of the c-fos gene demonstrates c-fos-dependent and -independent pathways for gene expression stimulated by growth factors or oncogenes". EMBO J. 13 (13): 3094–103. July 1994. doi:10.1002/j.1460-2075.1994.tb06608.x. PMID 8039503.
- ↑ "Regulation of Fra-1 and Fra-2 phosphorylation differs during the cell cycle of fibroblasts and phosphorylation in vitro by MAP kinase affects DNA binding activity". Oncogene 9 (9): 2537–47. September 1994. PMID 8058317.
- ↑ "Dual role for mitogen-activated protein kinase (Erk) in insulin-dependent regulation of Fra-1 (fos-related antigen-1) transcription and phosphorylation". Biochem. J. 368 (Pt 2): 573–80. December 2002. doi:10.1042/BJ20020579. PMID 12197835.
- ↑ "Extracellular signal-regulated kinase 1/2-mediated phosphorylation of JunD and FosB is required for okadaic acid-induced activator protein 1 activation". J. Biol. Chem. 274 (2): 1124–30. January 1999. doi:10.1074/jbc.274.2.1124. PMID 9873060.
- ↑ Tulchinsky E (July 2000). "Fos family members: regulation, structure and role in oncogenic transformation". Histol. Histopathol. 15 (3): 921–8. PMID 10963134.
- ↑ "The estrogen-dependent c-JunER protein causes a reversible loss of mammary epithelial cell polarity involving a destabilization of adherens junctions". J. Cell Biol. 132 (6): 1115–32. March 1996. doi:10.1083/jcb.132.6.1115. PMID 8601589.
- ↑ 13.0 13.1 13.2 Renthal W, Nestler EJ (September 2009). "Chromatin regulation in drug addiction and depression". Dialogues in Clinical Neuroscience 11 (3): 257–268. PMID 19877494. "[Psychostimulants] increase cAMP levels in striatum, which activates protein kinase A (PKA) and leads to phosphorylation of its targets. This includes the cAMP response element binding protein (CREB), the phosphorylation of which induces its association with the histone acetyltransferase, CREB binding protein (CBP) to acetylate histones and facilitate gene activation. This is known to occur on many genes including fosB and c-fos in response to psychostimulant exposure. ΔFosB is also upregulated by chronic psychostimulant treatments, and is known to activate certain genes (eg, cdk5) and repress others (eg, c-fos) where it recruits HDAC1 as a corepressor. ... Chronic exposure to psychostimulants increases glutamatergic [signaling] from the prefrontal cortex to the NAc. Glutamatergic signaling elevates Ca2+ levels in NAc postsynaptic elements where it activates CaMK (calcium/calmodulin protein kinases) signaling, which, in addition to phosphorylating CREB, also phosphorylates HDAC5.".
Figure 2: Psychostimulant-induced signaling events - ↑ "Co-transmission of dopamine and glutamate". The Journal of General Physiology 139 (1): 93–96. January 2012. doi:10.1085/jgp.201110659. PMID 22200950. "Coincident and convergent input often induces plasticity on a postsynaptic neuron. The NAc integrates processed information about the environment from basolateral amygdala, hippocampus, and prefrontal cortex (PFC), as well as projections from midbrain dopamine neurons. Previous studies have demonstrated how dopamine modulates this integrative process. For example, high frequency stimulation potentiates hippocampal inputs to the NAc while simultaneously depressing PFC synapses (Goto and Grace, 2005). The converse was also shown to be true; stimulation at PFC potentiates PFC–NAc synapses but depresses hippocampal–NAc synapses. In light of the new functional evidence of midbrain dopamine/glutamate co-transmission (references above), new experiments of NAc function will have to test whether midbrain glutamatergic inputs bias or filter either limbic or cortical inputs to guide goal-directed behavior.".
- ↑ Kanehisa Laboratories (10 October 2014). "Amphetamine – Homo sapiens (human)". KEGG Pathway. http://www.genome.jp/kegg-bin/show_pathway?hsa05031+2354. Retrieved 31 October 2014. "Most addictive drugs increase extracellular concentrations of dopamine (DA) in nucleus accumbens (NAc) and medial prefrontal cortex (mPFC), projection areas of mesocorticolimbic DA neurons and key components of the "brain reward circuit". Amphetamine achieves this elevation in extracellular levels of DA by promoting efflux from synaptic terminals. ... Chronic exposure to amphetamine induces a unique transcription factor delta FosB, which plays an essential role in long-term adaptive changes in the brain."
- ↑ "Transcriptional and epigenetic substrates of methamphetamine addiction and withdrawal: evidence from a long-access self-administration model in the rat". Molecular Neurobiology 51 (2): 696–717. 2015. doi:10.1007/s12035-014-8776-8. PMID 24939695. "Figure 1".
- ↑ 17.0 17.1 17.2 Robison AJ, Nestler EJ (November 2011). "Transcriptional and epigenetic mechanisms of addiction". Nature Reviews Neuroscience 12 (11): 623–637. doi:10.1038/nrn3111. PMID 21989194. "ΔFosB serves as one of the master control proteins governing this structural plasticity. ... ΔFosB also represses G9a expression, leading to reduced repressive histone methylation at the cdk5 gene. The net result is gene activation and increased CDK5 expression. ... In contrast, ΔFosB binds to the c-fos gene and recruits several co-repressors, including HDAC1 (histone deacetylase 1) and SIRT 1 (sirtuin 1). ... The net result is c-fos gene repression.".
Figure 4: Epigenetic basis of drug regulation of gene expression - ↑ 18.0 18.1 18.2 Nestler EJ (December 2012). "Transcriptional mechanisms of drug addiction". Clinical Psychopharmacology and Neuroscience 10 (3): 136–143. doi:10.9758/cpn.2012.10.3.136. PMID 23430970. "The 35-37 kD ΔFosB isoforms accumulate with chronic drug exposure due to their extraordinarily long half-lives. ... As a result of its stability, the ΔFosB protein persists in neurons for at least several weeks after cessation of drug exposure. ... ΔFosB overexpression in nucleus accumbens induces NFκB ... In contrast, the ability of ΔFosB to repress the c-Fos gene occurs in concert with the recruitment of a histone deacetylase and presumably several other repressive proteins such as a repressive histone methyltransferase".
- ↑ Nestler EJ (October 2008). "Transcriptional mechanisms of addiction: Role of ΔFosB". Philosophical Transactions of the Royal Society B: Biological Sciences 363 (1507): 3245–3255. doi:10.1098/rstb.2008.0067. PMID 18640924. "Recent evidence has shown that ΔFosB also represses the c-fos gene that helps create the molecular switch—from the induction of several short-lived Fos family proteins after acute drug exposure to the predominant accumulation of ΔFosB after chronic drug exposure".
- ↑ "C-Fos expression is a molecular predictor of progression and survival in epithelial ovarian carcinoma". Br. J. Cancer 99 (8): 1269–75. October 2008. doi:10.1038/sj.bjc.6604650. PMID 18854825.
- ↑ "Amphetamine and cocaine induce drug-specific activation of the c-fos gene in striosome-matrix compartments and limbic subdivisions of the striatum". Proc. Natl. Acad. Sci. U.S.A. 87 (17): 6912–6. September 1990. doi:10.1073/pnas.87.17.6912. PMID 2118661. Bibcode: 1990PNAS...87.6912G.
- ↑ "Psychomotor stimulant- and opiate-induced c-fos mRNA expression patterns in the rat forebrain: comparisons between acute drug treatment and a drug challenge in sensitized animals". Neurochem. Res. 21 (11): 1425–35. November 1996. doi:10.1007/BF02532384. PMID 8947933.
- ↑ "A single dose of lysergic acid diethylamide influences gene expression patterns within the mammalian brain". Neuropsychopharmacology 26 (5): 634–42. May 2002. doi:10.1016/S0893-133X(01)00405-5. PMID 11927188.
- ↑ 24.0 24.1 "Induction of c-Fos expression in specific areas of the fear circuitry in rat forebrain by anxiogenic drugs". Biol. Psychiatry 53 (4): 275–83. February 2003. doi:10.1016/S0006-3223(02)01574-3. PMID 12586446.
- ↑ "Comparing the effects of transcranial alternating current and temporal interference (tTIS) electric stimulation through whole-brain mapping of c-Fos immunoreactivity". Frontiers in Neuroanatomy 17. 2023-03-13. doi:10.3389/fnana.2023.1128193. PMID 36992795.
- ↑ "Environmental novelty is associated with a selective increase in c-fos expression in the output elements of the hippocampal formation and the perirhinal cortex". Learn. Mem. 15 (12): 899–908. 2008. doi:10.1101/lm.1196508. PMID 19050162.
- ↑ "The use of c-fos as a metabolic marker in neuronal pathway tracing". Journal of Neuroscience Methods 29 (3): 261–265. 1989. doi:10.1016/0165-0270(89)90150-7. PMID 2507830.
- ↑ "Conditioned Fear Inhibits c-fos mRNA Expression in the Central Extended Amygdala". Brain Res. 1229: 137–46. September 2008. doi:10.1016/j.brainres.2008.06.085. PMID 18634767.
- ↑ "Targeted disruption of cocaine-activated nucleus accumbens neurons prevents context-specific sensitization". Nat. Neurosci. 12 (8): 1069–73. August 2009. doi:10.1038/nn.2364. PMID 19620976.
- ↑ "Generation of a synthetic memory trace". Science 335 (6075): 1513–1516. March 2012. doi:10.1126/science.1214985. PMID 22442487. Bibcode: 2012Sci...335.1513G.
- ↑ "Bcl3, an IkappaB protein, stimulates activating protein-1 transactivation and cellular proliferation". J. Biol. Chem. 274 (40): 28491–6. October 1999. doi:10.1074/jbc.274.40.28491. PMID 10497212.
- ↑ "COBRA1 inhibits AP-1 transcriptional activity in transfected cells". Biochem. Biophys. Res. Commun. 325 (2): 568–73. December 2004. doi:10.1016/j.bbrc.2004.10.079. PMID 15530430. Bibcode: 2004BBRC..325..568Z.
- ↑ 33.0 33.1 "Casein kinase II interacts with the bZIP domains of several transcription factors". Nucleic Acids Res. 26 (16): 3854–61. August 1998. doi:10.1093/nar/26.16.3854. PMID 9685505.
- ↑ "CHOP enhancement of gene transcription by interactions with Jun/Fos AP-1 complex proteins". Mol. Cell. Biol. 19 (11): 7589–99. November 1999. doi:10.1128/MCB.19.11.7589. PMID 10523647.
- ↑ 35.0 35.1 "ERK MAP kinase links cytokine signals to activation of latent HIV-1 infection by stimulating a cooperative interaction of AP-1 and NF-kappaB". J. Biol. Chem. 274 (39): 27981–8. September 1999. doi:10.1074/jbc.274.39.27981. PMID 10488148.
- ↑ "Identification of SWI.SNF complex subunit BAF60a as a determinant of the transactivation potential of Fos/Jun dimers". J. Biol. Chem. 276 (4): 2852–7. January 2001. doi:10.1074/jbc.M009633200. PMID 11053448.
- ↑ "Cross-family interaction between the bHLHZip USF and bZip Fra1 proteins results in down-regulation of AP1 activity". Oncogene 14 (17): 2091–8. May 1997. doi:10.1038/sj.onc.1201046. PMID 9160889.
- ↑ "Crystal structure of the heterodimeric bZIP transcription factor c-Fos-c-Jun bound to DNA". Nature 373 (6511): 257–61. January 1995. doi:10.1038/373257a0. PMID 7816143. Bibcode: 1995Natur.373..257G.
- ↑ "Isolation and characterization of a novel member of the gene family encoding the cAMP response element-binding protein CRE-BP1". J. Biol. Chem. 268 (6): 4259–66. February 1993. doi:10.1016/S0021-9258(18)53604-8. PMID 8440710.
- ↑ "Detection and modulation in vivo of helix-loop-helix protein-protein interactions". J. Biol. Chem. 268 (1): 5–8. January 1993. doi:10.1016/S0021-9258(18)54105-3. PMID 8380166.
- ↑ "Nrf2 and Nrf1 in association with Jun proteins regulate antioxidant response element-mediated expression and coordinated induction of genes encoding detoxifying enzymes". Oncogene 17 (24): 3145–56. December 1998. doi:10.1038/sj.onc.1202237. PMID 9872330.
- ↑ "Activating protein-1, nuclear factor-kappaB, and serum response factor as novel target molecules of the cancer-amplified transcription coactivator ASC-2". Mol. Endocrinol. 14 (6): 915–25. June 2000. doi:10.1210/mend.14.6.0471. PMID 10847592.
- ↑ "Steroid receptor coactivator-1 coactivates activating protein-1-mediated transactivations through interaction with the c-Jun and c-Fos subunits". J. Biol. Chem. 273 (27): 16651–4. July 1998. doi:10.1074/jbc.273.27.16651. PMID 9642216.
- ↑ "Silencing mediator of retinoic acid and thyroid hormone receptors, as a novel transcriptional corepressor molecule of activating protein-1, nuclear factor-kappaB, and serum response factor". J. Biol. Chem. 275 (17): 12470–4. April 2000. doi:10.1074/jbc.275.17.12470. PMID 10777532.
- ↑ 45.0 45.1 "AP-1 and Cbfa/runt physically interact and regulate parathyroid hormone-dependent MMP13 expression in osteoblasts through a new osteoblast-specific element 2/AP-1 composite element". J. Biol. Chem. 276 (23): 20029–38. June 2001. doi:10.1074/jbc.M010601200. PMID 11274169.
- ↑ 46.0 46.1 "Physical interaction of the activator protein-1 factors c-Fos and c-Jun with Cbfa1 for collagenase-3 promoter activation". J. Biol. Chem. 277 (1): 816–22. January 2002. doi:10.1074/jbc.M107082200. PMID 11641401.
- ↑ "Smad3 and Smad4 cooperate with c-Jun/c-Fos to mediate TGF-beta-induced transcription". Nature 394 (6696): 909–13. August 1998. doi:10.1038/29814. PMID 9732876. Bibcode: 1998Natur.394..909Z.
- ↑ "c-Fos-induced activation of a TATA-box-containing promoter involves direct contact with TATA-box-binding protein". Mol. Cell. Biol. 14 (9): 6021–9. September 1994. doi:10.1128/MCB.14.9.6021. PMID 8065335.
Further reading
- "Regulation of gene expression in T-47D human breast cancer cells by progestins and antiprogestins". Hum. Reprod. 9 (Suppl 1): 174–80. June 1994. doi:10.1093/humrep/9.suppl_1.174. PMID 7962462.
- "c-Fos expression during wakefulness and sleep". Neurophysiol Clin 25 (6): 329–41. 1995. doi:10.1016/0987-7053(96)84906-9. PMID 8904195.
- "Activation of c-fos in the brain". Prog. Neurobiol. 50 (2–3): 83–107. October 1996. doi:10.1016/S0301-0082(96)00021-4. PMID 8971979.
- "[Possible role of transcription factor AP1 in the tissue-specific regulation of human papillomavirus]" (in es). Rev. Invest. Clin. 54 (3): 231–42. 2002. PMID 12183893.
External links
- c-fos+Proteins at the US National Library of Medicine Medical Subject Headings (MeSH)
- c-fos+Genes at the US National Library of Medicine Medical Subject Headings (MeSH)
- FactorBook c-Fos
- Drosophila kayak - The Interactive Fly
- Human FOS genome location and FOS gene details page in the UCSC Genome Browser.
- Overview of all the structural information available in the PDB for UniProt: P01100 (Human Proto-oncogene c-Fos) at the PDBe-KB.
- Overview of all the structural information available in the PDB for UniProt: P01101 (Mouse Proto-oncogene c-Fos) at the PDBe-KB.
 |