Chemistry:3',4'-Methylenedioxy-α-pyrrolidinopropiophenone
From HandWiki
Short description: Stimulant drug
 | |
 | |
| Clinical data | |
|---|---|
| Routes of administration | By mouth, insufflation, vaporization, IV, rectal, sublingual |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Metabolism | Hepatic[1][2] |
| Excretion | Primarily urine (renal) |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| Chemical and physical data | |
| Formula | C14H17NO3 |
| Molar mass | 247.294 g·mol−1 |
| 3D model (JSmol) | |
| Chirality | Racemic mixture |
| |
| |
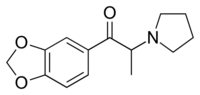
3',4'-Methylenedioxy-α-pyrrolidinopropiophenone (MDPPP) is a stimulant designer drug. It was sold in Germany in the late 1990s and early 2000s as an ingredient in imitation ecstasy (MDMA) pills.[3] It shares a similar chemical structure with α-PPP and MDPV,[4][1][2] and has been shown to have reinforcing effects in rats.[5]
Metabolism
MDPPP appears to have a similar metabolic fate as MDPV.[2]
Legal Status
(As of October 2015) MDPPP is a controlled substance in China.[6]
See also
- α-Pyrrolidinopropiophenone (α-PPP)
- 4'-Methyl-α-pyrrolidinopropiophenone (MPPP)
- 4'-Methoxy-α-pyrrolidinopropiophenone (MOPPP)
- 3',4'-Methylenedioxy-α-pyrrolidinobutiophenone (MDPBP)
- Dimethylone
References
- ↑ 1.0 1.1 "Metabolism of designer drugs of abuse". Current Drug Metabolism 6 (3): 259–74. June 2005. doi:10.2174/1389200054021825. PMID 15975043.
- ↑ 2.0 2.1 2.2 "Identification of cytochrome P450 enzymes involved in the metabolism of 3',4'-methylenedioxy-alpha-pyrrolidinopropiophenone (MDPPP), a designer drug, in human liver microsomes". Xenobiotica; the Fate of Foreign Compounds in Biological Systems 35 (3): 227–37. March 2005. doi:10.1080/00498250400028239. PMID 16019948.
- ↑ "Metabolism and toxicological detection of the new designer drug 3',4'-methylenedioxy-alpha-pyrrolidinopropiophenone studied in urine using gas chromatography-mass spectrometry". Journal of Chromatography. B, Analytical Technologies in the Biomedical and Life Sciences 793 (2): 377–88. August 2003. doi:10.1016/S1570-0232(03)00350-7. PMID 12906913.
- ↑ "Chemistry, pharmacology, toxicology, and hepatic metabolism of designer drugs of the amphetamine (ecstasy), piperazine, and pyrrolidinophenone types: a synopsis". Therapeutic Drug Monitoring 26 (2): 127–31. April 2004. doi:10.1097/00007691-200404000-00007. PMID 15228152.
- ↑ "Relative reinforcing effects of second-generation synthetic cathinones: Acquisition of self-administration and fixed ratio dose-response curves in rats". Neuropharmacology 134 (Pt A): 28–35. May 2018. doi:10.1016/j.neuropharm.2017.08.018. PMID 28811192.
- ↑ "关于印发《非药用类麻醉药品和精神药品列管办法》的通知" (in Chinese). China Food and Drug Administration. 27 September 2015. http://www.sfda.gov.cn/WS01/CL0056/130753.html.
 |

