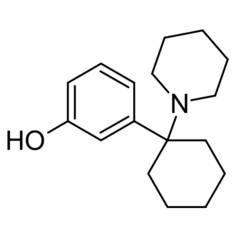
Chemistry:3-HO-PCP
 | |
| Clinical data | |
|---|---|
| Other names | 3-Hydroxyphencyclidine; 3-OH-PCP; PCP-3-OH |
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number |
|
| PubChem CID | |
| ChemSpider | |
| UNII | |
| Chemical and physical data | |
| Formula | C17H25NO |
| Molar mass | 259.393 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
3-Hydroxyphencyclidine (3-HO-PCP) is a dissociative of the arylcyclohexylamine class related to phencyclidine (PCP) that has been sold online as a designer drug.[1][2]
Pharmacology
3-HO-PCP acts as a high-affinity uncompetitive antagonist of the NMDA receptor via the dizocilpine (MK-801) site (Ki = 30 nM).[1][3] It has much higher affinity than PCP for this site (Ki = 250 nM, for comparison; 8-fold difference).[3] The drug also has high affinity for the μ-opioid receptor (MOR) (Ki = 39–60 nM) in animal test subjects,[1][3][4][5] the κ-opioid receptor (KOR) (Ki = 140 nM),[4] and the sigma σ1 receptor (Ki = 42 nM; IC50 = 19 nM),[4][6][7][8] whereas it has only low affinity for the δ-opioid receptor (Ki = 2,300 nM).[4] The high affinity of 3-HO-PCP for opioid receptors is unique among arylcyclohexylamines and is in contrast to PCP, which has only very low affinity for the MOR (Ki = 11,000–26,000 nM; 282- to 433-fold difference) and the other opioid receptors (Ki = 4,100 nM for the KOR and 73,000 nM for the DOR).[3][4]
Although it was hypothesized that 3-HO-PCP might be a metabolite of PCP in humans, there is no evidence that this is the case.[9][10]
Chemistry
3-HO-PCP is an arylcyclohexylamine.[1] Close analogues of 3-HO-PCP include PCP, 3-MeO-PCP, 4-MeO-PCP, 3-MeO-PCMo, and somewhat more distantly ketamine, methoxyketamine, 3-MeO-PCE, methoxetamine and dimetamine.[1]
Society and culture
Legal status
On October 18, 2012, the Advisory Council on the Misuse of Drugs in the United Kingdom released a report about methoxetamine, saying that the "harms of methoxetamine are commensurate with Class B of the Misuse of Drugs Act (1971)", despite the fact that the act does not classify drugs based on harm. The report went on to suggest that all analogues of MXE should also become class B drugs and suggested a catch-all clause covering both existing and unresearched arylcyclohexamines, including 3-HO-PCP.[11]
3-HO-PCP is banned in Sweden[12][13] and Switzerland.[14]
See also
References
- ↑ 1.0 1.1 1.2 1.3 1.4 "From PCP to MXE: a comprehensive review of the non-medical use of dissociative drugs". Drug Testing and Analysis 6 (7–8): 614–632. 2014. doi:10.1002/dta.1620. PMID 24678061.
- ↑ "In vitro and in vivo metabolism and detection of 3-HO-PCP, a synthetic phencyclidine, in human samples and pooled human hepatocytes using high resolution mass spectrometry". Drug Testing and Analysis 12 (7): 987–993. July 2020. doi:10.1002/dta.2807. PMID 32311838.
- ↑ 3.0 3.1 3.2 3.3 "Chemical synthesis and molecular pharmacology of hydroxylated 1-(1-phenylcyclohexyl-piperidine derivatives". Journal of Medicinal Chemistry 25 (4): 431–435. April 1982. doi:10.1021/jm00346a019. PMID 6279847.
- ↑ 4.0 4.1 4.2 4.3 4.4 "Interaction of two phencyclidine opiate-like derivatives with 3H-opioid binding sites". European Journal of Pharmacology 101 (3–4): 281–284. June 1984. doi:10.1016/0014-2999(84)90171-7. PMID 6088255.
- ↑ "On the opioid nature of phencyclidine and its 3-hydroxy derivative". European Journal of Pharmacology 73 (2–3): 229–233. July 1981. doi:10.1016/0014-2999(81)90097-2. PMID 6273187.
- ↑ "Characterization of specific binding sites for [3H](d)-N-allylnormetazocine in rat brain membranes". Molecular Pharmacology 27 (1): 46–52. January 1985. PMID 3965930.
- ↑ "[3H]PCP-3-OH and (+)[3H]SKF 10047 binding sites in rat brain membranes: evidence of multiplicity". European Journal of Pharmacology 136 (2): 231–234. April 1987. doi:10.1016/0014-2999(87)90715-1. PMID 3036548.
- ↑ "Pharmacological specificity of some psychotomimetic and antipsychotic agents for the sigma and PCP binding sites". Life Sciences 42 (7): 745–752. 1988. doi:10.1016/0024-3205(88)90646-7. PMID 2893238.
- ↑ "Biotransformation of phencyclidine". Drug Metabolism Reviews 16 (3): 285–320. 1985. doi:10.3109/03602538508991437. PMID 3914938.
- ↑ "Quantitation of phencyclidine, its metabolites, and derivatives by gas chromatography with nitrogen-phosphorus detection: application for in vivo and in vitro biotransformation studies". Journal of Analytical Toxicology 10 (3): 107–115. 1986. doi:10.1093/jat/10.3.107. PMID 3724069.
- ↑ "(ACMD) Methoxetamine Report (2012)" (PDF). UK Home Office. 2012-10-18. pp. 14. https://www.gov.uk/government/publications/advisory-council-on-the-misuse-of-drugs-acmd-methoxetamine-report-2012.
- ↑ "Elva nya ämnen klassas som narkotika eller hälsofarlig vara" (in sv). Folkhälsomyndigheten. 28 June 2018. https://www.folkhalsomyndigheten.se/nyheter-och-press/nyhetsarkiv/2018/juni/elva-nya-amnen-klassas-som-narkotika-eller-halsofarlig-vara/.
- ↑ "Förordning (1992:1554) om kontroll av narkotika" (in sv). Riksdagsförvaltningen. https://riksdagen.se/sv/dokument-lagar/dokument/svensk-forfattningssamling/forordning-19921554-om-kontroll-av-narkotika_sfs-1992-155.
- ↑ "Verordnung des EDI vom 30. Mai 2011 über die Verzeichnisse der Betäubungsmittel, psychotropen Stoffe, Vorläuferstoffe und Hilfschemikalien (Betäubungsmittelverzeichnisverordnung, BetmVV-EDI)" (in de). Der Bundesrat. https://www.admin.ch/opc/de/classified-compilation/20101220/index.html.
 |

