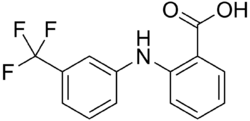
Chemistry:Flufenamic acid
 | |
 | |
| Clinical data | |
|---|---|
| AHFS/Drugs.com | International Drug Names |
| Routes of administration | By mouth, topical |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Protein binding | extensively |
| Metabolism | Hydroxylation, glucuronidation |
| Elimination half-life | ~3 h |
| Excretion | 50% urine, 36% feces |
| Identifiers | |
| |
| CAS Number |
|
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank |
|
| ChemSpider |
|
| UNII | |
| KEGG |
|
| ChEBI | |
| ChEMBL | |
| Chemical and physical data | |
| Formula | C14H10F3NO2 |
| Molar mass | 281.234 g·mol−1 |
| 3D model (JSmol) | |
| Melting point | 124 to 125 °C (255 to 257 °F) resolidification and remelting at 134°C to 136°C |
| Solubility in water | Practically insoluble in water; soluble in ethanol, chloroform and diethyl ether mg/mL (20 °C) |
| |
| |
| (verify) | |
Flufenamic acid (FFA) is a member of the anthranilic acid derivatives (or fenamate) class of nonsteroidal anti-inflammatory drugs (NSAIDs).[1]:718 Like other members of the class, it is a cyclooxygenase (COX) inhibitor, preventing the formation of prostaglandins.[2] FFA is known to bind to and reduce the activity of prostaglandin F synthase and activate TRPC6.[3]
It is not widely used in humans as it has a high rate (30–60%) of gastrointestinal side effects.[4]:310 It is generally not available in the US.[2] It is available in some Asian and European countries as a generic drug.[5]
Scientists led by Claude Winder from Parke-Davis invented FFA in 1963, along with fellow members of the class, mefenamic acid in 1961 and meclofenamic acid in 1964.[1]:718
Although flufenamic acid was at one time informally referred to as "Fluffy" (see history cache), this pet name could also refer to flufenoxine.
References
- ↑ 1.0 1.1 "Drugs to treat inflammation: a historical introduction". Current Medicinal Chemistry 12 (25): 2931–42. 2005. doi:10.2174/092986705774462879. ISBN 9781608052073. PMID 16378496.
- ↑ 2.0 2.1 NIH LiverTox Database Mefenamic Acid Last updated June 23, 2015. Page accessed July 3, 2015. Quote: "(fenamates generally not available in the United States, such as tolfenamic acid and flufenamic acid)"
- ↑ "Chemical–Gene Interaction Query: Flufenamic Acid (Homo sapiens)". North Carolina State University. http://ctdbase.org/query.go?type=ixn&chemqt=equals&chem=name%3AFlufenamic+Acid&actionDegreeTypes=increases&actionDegreeTypes=decreases&actionDegreeTypes=affects&actionTypes=activity&actionTypes=binding&geneqt=equals&gene=&pathwayqt=equals&pathway=&taxonqt=equals&taxon=TAXON%3A9606&goqt=equals&go=&sort=chemNmSort&perPage=500&action=Search.
- ↑ Aronson, Jeffrey K. (2009). Meyler's Side Effects of Analgesics and Anti-inflammatory Drugs. Elsevier. ISBN 978-0-08-093294-1.
- ↑ "International listings for flufenamic acid". Drugs.com. https://www.drugs.com/international/flufenamic-acid.html.
| Pyrazolones / Pyrazolidines | |
|---|---|
| Salicylates | |
| Acetic acid derivatives and related substances | |
| Oxicams | |
| Propionic acid derivatives (profens) |
|
| N-Arylanthranilic acids (fenamates) | |
| Coxibs | |
| Other | |
Items listed in bold indicate initially developed compounds of specific groups. #WHO-EM †Withdrawn drugs. ‡Veterinary use medications. | |
 | 0.00      (0 votes) (0 votes) |
